Abstract
The influence of environmental factors like smoking and alcohol on myopia and astigmatism is controversial. However, due to ethical concerns, alternative study designs are urgently needed to assess causal inference, as mandatory exposure to cigarettes and alcohol is unethical. Following comprehensive screenings, 326 single nucleotide polymorphisms (SNPs) related to myopia and astigmatism were included in the dataset. To validate the causal association between exposures such as cigarette smoking, alcohol consumption, and coffee intake, and outcomes namely astigmatism and myopia, five regression models were employed. These models encompassed MR-Egger regression, random-effects inverse-variance weighted (IVW), weighted median estimator (WME), weighted model, and simple model. The instrumental variables utilized in these analyses were the aforementioned SNPs. Apply Cochran’s Q test to determine heterogeneity of SNPs; if heterogeneity exists, focus on IVW model results. The IVW model showed a 1.379-fold increase in the risk of astigmatism (OR = 1.379, 95%CI 0.822~2.313, P = 0.224) and a 0.963-fold increase in the risk of myopia (OR = 0.963, 95%CI 0.666~1.393, P = 0.841) for each unit increase in smoking. For each unit increase in coffee intake, the risk of astigmatism increased 1.610-fold (OR = 1.610, 95%CI 0.444~5.835, P = 0.469) and the risk of myopia increased 0.788-fold (OR = 0.788, 95%CI 0.340~1.824, P = 0.578). For each additional unit of alcohol consumption, the risk of astigmatism increased by 0.763-fold (OR = 0.763, 95%CI 0.380~1.530, P = 0.446), and none of the differences were statistically significant. However, for each unit of alcohol consumption, the risk of myopia increased by 1.597 times, and the difference was statistically significant (OR = 1.597, 95%CI 1.023~2.493, P = 0.039). The findings indicate that alcohol consumption is a risk factor for myopia but smoking and coffee intake do not affect its development. Additionally, there is no association between smoking, alcohol consumption, coffee intake, and the risk of astigmatism.
Similar content being viewed by others
Introduction
Myopia's global prevalence exhibits a consistent upward trajectory, particularly in East and Southeast Asian countries where its prevalence surpasses the global average1,2,3. More than 60% of adults, meanwhile, are afflicted by astigmatism4. For a long time, it has been widely postulated that astigmatism and myopia share a close association5,6,7. Furthermore, myopia is associated with many eye diseases such as retinal detachment, cataract, glaucoma, and macular degeneration, and even permanent vision loss in severe cases. A study in Hong Kong China finds higher incidence rate of glaucoma of myopic individuals seeking corneal refractive surgery8. The World Health Organization has listed uncorrected refractive error as one of the leading causes of visual impairment worldwide9, and elevated vision health to the level of an international public health issue. An important review summarize several risk factors for myopia including age, genetic or acquired, and environmental factors10. For example, many researches agree that increased TSO and reduced NW are protective against myopia development among nonmyopes even the strength of evidence is less because of high heterogeneity and lack of clinical trials with clear definition11. The underlying mechanism is that when we engage in outdoor activities, we are exposed to an average outdoor light intensity that is approximately 20 times higher than typical indoor lighting. This strong light exposure increases retinal illumination and stimulates dopamine release, which in turn suppresses the axial growth of the eye12. Therefore, the early identification of potential risk factors for myopia and astigmatism and the discovery of their specific pathogenesis are of great practical significance to effectively prevent or delay myopia and other refractive errors13,14,15,16.
Recent epidemiologic studies suggest that, smoking, alcohol consumption and beverage intake may also play a significant role influencing refractive errors such as myopia. A British study from birth to adult observational study found that myopia was positively associated with maternal smoking in early pregnancy and a similar study was done for astigmatism17. There is a similar study on astigmatism 18. Nevertheless, a study conducted on the UK ALSPAC database presents a contrasting perspective, suggesting that the act of smoking during pregnancy by grandmothers actually diminishes the prevalence of early-onset myopia19,20,21. The Singapore Myopia Risk Factor Cohort Study (STARS) also suggests that maternal smoking may affect ocular development via nicotinic acetylcholine receptors, thereby reducing myopia in children22,23. The cross-sectional reports from different regions and age groups in China also support the view that smoking is beneficial to reducing myopia24,25,26. The Spanish prospective cohort study (SUN project) followed the relationship between alcohol intake and myopia over a 10-year period after graduation, and found that alcohol consumption was significantly associated with the prevalence of myopia27. More than half of adults with refractive error in India have a drinking habit28. Children with Fetal Alcohol Spectrum Disorder (FASD) are more likely to have visual impairment29. In conclusion, potential environmental factors like smoking and alcohol consumption can impact the development of myopia and astigmatism, but the exact causal relationship and relative importance are still subject to controversy. However, most of this evidence comes from traditional observational studies, which are susceptible to confounding, reverse causation, and measurement error. Considering the unethical nature of forced exposure to cigarettes and alcohol, randomized controlled trials, are not applicable in the present issue.
Mendelian randomization (MR) is one of the study designs in genetic epidemiology. It utilizes the random splitting and combining of genetically variant gametes to re-randomize populations, and uses genotype as an instrumental variable to assess causality between risk factors and disease. In theory, MR can minimize the influence of confounding factors, circumvent reverse causality, and provide sufficient statistical validity. In recent times, MR has gained widespread utilization in investigating the causality of risk factors in recurrent diseases, primarily relying on the valuable data obtained from genome-wide association studies (GWAS). This approach has enabled researchers to delve into the causal relationship of these risk factors with greater depth and precision [PNS]30,31.
Mendelian randomization (MR) has not been widely employed to comprehensively examine the causal connections between potential risk factors and myopia. In a pioneering manner, we have employed MR to investigate the potential impacts of alcohol consumption, coffee intake, and smoking on myopia. Utilizing data from the Genome-Wide Association Studies (GWAS) database, we have made predictions regarding the prevalence risk of myopia and astigmatism. Our findings indicate a lack of evidence supporting a causal relationship between smoking, coffee intake, and astigmatism. However, it is noteworthy that alcohol consumption emerges as a significant risk factor for the development of myopia.
Data sources.
The information on smoking alcohol consumption and coffee intake was obtained through the IEU Open GWAS project (https://gwas.mrcieu.ac.uk/), UK Biobank (http://www.nealelab.is/uk-biobank), FinnGen (https://www.finngen.fi/fi) websites for smoking, alcohol intake frequency, coffee intake, astigmatism and myopia in GWAS data. Site accessed on 2023-08-20.
The final population sources of the data obtained were all European populations, both sexes, whose summary information is presented in Table 1. Relevant data were obtained from five different published GWAS databases, and the original studies had obtained the study Informed consent was obtained from the subjects for the original study and therefore this part of the study did not involve the need for ethics committee approval.
Methods
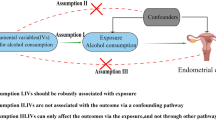
The following criteria were used to screen instrumental variables in this study (Fig. 1):
-
①Instrumental variables were highly correlated with exposure, with P < 5 × 10−8 as the criterion for strong correlation (assumption of association).
-
② The instrumental variables were not directly related to the outcome, but only affected the outcome through exposure, i.e., there was no genetic pleiotropy (exclusivity hypothesis). The absence of genetic pleiotropy is now indicated by a non-zero intercept term in the MR-Egger regression model (P > 0.05).
-
③ Instrumental variables must be independent of confounders (independence assumption). Since the SNPs selected by the MR method follow the genetic principle of random assignment of parental alleles to offspring, they are subjected to very little environmental and acquired life, i.e., theoretically, the instrumental variables can be assumed to be independent of environmental factors, such as socio-economic and cultural factors. In addition, the F-statistic greater than 10 was used as an indicator for evaluating weak instrumental variables32.
Schematic of Mendelian randomization of the association between smoking and astigmatism. Hypothesis 1: Genotypes must be associated with the exposure factor to be studied; Hypothesis 2: Genotypes must be independent of confounders; Hypothesis 3: Genotypes can only be associated with outcomes by influencing the exposure factor to be studied. ieu-b-4877, ukb-a-25, ukb-b-5237, finn-b-H7_ASTIGMATISM and finn-b-H7_MYOPIA of GWAS were each five published GWAS studies.
SNP screening
Significant SNPs were screened from the smoking GWAS pooled data (with P < 5 × 10−8 as the screening condition, Hypothesis 1); the chain disequilibrium coefficient r 2 was set to be 0.001, and the width of chain disequilibrium region was set to be 10,000 kb to ensure that individual SNPs were independent of each other and to exclude the influence of gene pleiotropy on the results; SNPs associated with confounding factors and endpoints were excluded by the Pheno Scanner associated SNPs (hypotheses 2 and 3). The relevant SNPs screened above were extracted from the GWAS pooled data of astigmatism; a minimum r 2 > 0.8 was set. Information of the above dataset was summarized, while SNPs directly related to astigmatism were excluded (P < 5 × 10−8).Including the results of the main analysis, heterogeneity analysis and sensitivity analysis.
Five regression models, MR-Egger regression, random-effects inverse-variance weighted (IVW), weighted median estimator (WME), weighted model, and simple model, were used to validate the causal relationship between exposures (smoking, alcohol consumption, and coffee intake) and outcomes (astigmatism, myopia) using SNP as an instrumental variable. We used SNP as an instrumental variable to verify the causal relationship between exposure (smoking, alcohol consumption, coffee intake) and outcome (astigmatism, myopia). The IVW method does not require individual-level data and can directly calculate causal effect values using pooled data, while the MR-Egger regression calculates the correlation between each SNP and astigmatism and myopia (Y) and the correlation between each SNP and smoking, alcohol, and coffee intake (X), and then fits a linear function. The WME method calculates the causal effect estimate of the exposure-endpoint for the jth SNP (βj). Cochran's Q test was applied to determine the heterogeneity of the SNPs; if heterogeneity existed, the IVW model results were focused on; multivariate analyses were performed using the intercept term of the MR-Egger method and the Leave-one-out test; and finally, multivariate MR analyses were used for corrective analyses to adjust for potential confounders. All of the above methods were implemented using the TwoSample MR package in the R 4.2.2 software with a test level of α = 0.05.
Results
After several screenings, 326 SNPs were finally included in the myopia and astigmatism dataset, and the basic information of some SNPs is shown in Table 2, and the unlisted parts are similar to them. The distribution of F-statistics corresponding to single SNPs ranged from 29.75 to 646.73 (mean 54.29), indicating that causal associations were less likely to be affected by weak instrumental variables.
Results
Causality verification
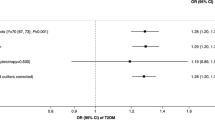
Regression results (Table 3), the results suggest that alcohol consumption is a risk factor for the development of myopia, while smoking and coffee intake do not affect the development of myopia, and smoking, alcohol consumption, and coffee intake are not associated with the risk of astigmatism (P > 0.05). The IVW model showed that the risk of astigmatism increased by 1.379 times for each unit increase in smoking, but the difference was not statistically significant (OR = 1.379, 95%CI 0.822–2.313, P = 0.224), and the results of the MR-Egger regression, WME analysis, weighted model, and simple modeling analysis also showed that there was no direction of causality effect, and the direction of the above five methods was consistent. However, the results of IVW method suggested that the risk of myopia increased by 1.597 times for each unit increase in alcohol consumption, and the difference was statistically significant (OR = 1.597, 95%CI 1.023–2.493, P = 0.039), whereas the results of MR-Egger regression, WME analysis, weighted model, and simple model showed no statistically significant direction, and the direction of causality was inconsistent, taking the results of IVW method as a whole. The results of IVW method were taken in synthesis (Fig. 2).
Heterogeneity test
The statistic Q shown by MR-Egger regression and Cochran Q test of IVW method, both P > 0.05, suggested that there was no heterogeneity among SNPs (Table 4). And the difference between the MR-Egger regression intercept term and 0 was not statistically significant (P > 0.05), so we concluded that there was no genetic pleiotropy among the SNPs (Table 5). The scatterplot and funnel plot showed that the distributions of all the included SNPs were largely symmetrical, suggesting that causal associations were less likely to be affected by potential bias (Fig. 3).
Sensitivity analysis
After the Leave-one-out test, the analysis results of the remaining SNPs were similar to those of the inclusion of all SNPs after removing each SNP of smoking in turn, and no SNPs were found to have a large impact on the causal association estimates, indicating that the MR results of this study were robust (Fig. 4).
Discussion
In this study, we used a sample of studies from the GWAS database to predict the prevalence risk of myopia and astigmatism. We found no evidence of a causal relationship between smoking, coffee intake and astigmatism. However, the results of IVW method suggested that the risk of myopia prevalence increased by 1.597 times for each unit increase in alcohol consumption, and the difference was statistically significant (OR = 1.597, 95%CI 1.023~2.493, P = 0.039), while in the SUN cohort study Alcohol intake was linearly and significantly associated with a higher risk of myopia development or progression with the OR for 10-year incidence/progression of myopia was 1.05, 95% CI 1.01–1.09 per each 10-g increase in alcohol intake27.
Mendelian randomized causal inference is commonly used in MR-Egger regression, inverse-variance weighted (IVW), weighted median estimator (WME), weighted model, simple model, etc. Among them, IVW method does not require individual-level data and can use pooled data to calculate causal effect values directly, which is the main analysis method. Among them, the IVW method does not require individual-level data and can directly calculate causal effect values using aggregated data, which is the main analysis method, while the MR-Egger regression and the WME method are supplementary analysis methods. In the causal relationship between alcohol intake and myopia, the IVW method suggested that the difference between the results was statistically significant, and subsequent sensitivity analyses suggested that the present results were robust, i.e., the conclusion that alcohol intake is a risk factor for myopia was valid although the results of the MR-Egger regression, the WME analysis, the weighted model, and the simple model showed no statistically significant difference.
We speculate that there may be the following mechanisms: (1) Dopamine regulation: Alcohol consumption has been shown to decrease dopamine secretion in the brain. As dopamine plays a role in regulating eye growth and preventing myopia, the reduction in dopamine levels caused by alcohol may disrupt this regulatory process. (2) Visual processing impairment: Alcohol can impair visual processing and perception. This may lead to difficulties in focusing and strain on the eyes, potentially contributing to the development or progression of myopia. (3) Lifestyle factors: Alcohol consumption is often associated with certain lifestyle habits, such as spending less time outdoors and engaging in more sedentary activities. These factors have been linked to an increased risk of myopia. (4) Nutritional deficiencies: Excessive alcohol consumption can lead to nutritional deficiencies, such as deficiencies in vitamins and minerals that are essential for eye health. These deficiencies may contribute to the development of myopia. However, it is important to note that these are speculative mechanisms and further research is needed to confirm their validity.
In summary, this study further illustrates the causal relationship between alcohol consumption and myopia, which is useful for the promotion of myopia prevention and control. However, the results of the analysis show that the results of alcohol consumption and myopia are not completely unified, and the results of the IVW method, as well as the individual differences between people and other living habits, need to be further investigated.
Data availability
The datasets analyzed during the current study are available in the IEU Open GWAS project (https://gwas.mrcieu.ac.uk/), UK Biobank (http://www.nealelab.is/uk-biobank), FinnGen (https://www.finngen.fi/fi).
References
Dolgin, E. The myopia boom. Nature 519, 276–278. https://doi.org/10.1038/519276a (2015).
Holden, B. A. et al. Global prevalence of myopia and high myopia and temporal trends from 2000 through 2050. Ophthalmology 123, 1036–1042. https://doi.org/10.1016/j.ophtha.2016.01.006 (2016).
Wu, P. C., Huang, H. M., Yu, H. J., Fang, P. C. & Chen, C. T. Epidemiology of myopia. Asia-Pacific J. Ophthalmol. (Philadelphia Pa.) 5, 386–393. https://doi.org/10.1097/apo.0000000000000236 (2016).
Attebo, K., Ivers, R. Q. & Mitchell, P. Refractive errors in an older population: The Blue Mountains eye study. Ophthalmology 106, 1066–1072. https://doi.org/10.1016/s0161-6420(99)90251-8 (1999).
Green, J. On astigmatism as an active cause of myopia. Trans. Am. Ophthalmol. Soc. 1, 105–107 (1871).
Gwiazda, J., Grice, K., Held, R., McLellan, J. & Thorn, F. Astigmatism and the development of myopia in children. Vision research 40, 1019–1026. https://doi.org/10.1016/s0042-6989(99)00237-0 (2000).
Linke, S. J., Richard, G. & Katz, T. Prevalence and associations of anisometropia with spherical ametropia, cylindrical power, age, and sex in refractive surgery candidates. Investig. Ophthalmol. Vis. Sci. 52, 7538–7547. https://doi.org/10.1167/iovs.11-7620 (2011).
Biswas, S., Jhanji, V. & Leung, C. K. Prevalence of glaucoma in myopic corneal refractive surgery candidates in Hong Kong China. J. Refract. Surg. 32(5), 298–304. https://doi.org/10.3928/1081597X-20160229-01 (2016).
Harb, E. N. & Wildsoet, C. F. Origins of refractive errors: Environmental and genetic factors. Ann. Rev. Vis. Sci. 5, 47–72. https://doi.org/10.1146/annurev-vision-091718-015027 (2019).
Ohno-Matsui, K. et al. IMI pathologic myopia [published correction appears in Invest Ophthalmol Vis Sci. 2021 Jun 1;62(7):17]. Invest. Ophthalmol. Vis. Sci. 62(5), 5. https://doi.org/10.1167/iovs.62.5.5 (2021).
Karthikeyan, S. K., Ashwini, D. L., Priyanka, M., Nayak, A. & Biswas, S. Physical activity, time spent outdoors, and near work in relation to myopia prevalence, incidence, and progression: An overview of systematic reviews and meta-analyses. Indian J. Ophthalmol. 70(3), 728–739. https://doi.org/10.4103/ijo.IJO_1564_21 (2022).
Muralidharan, A. R. et al. Light and myopia: From epidemiological studies to neurobiological mechanisms. Ther. Adv. Ophthalmol. https://doi.org/10.1177/25158414211059246 (2021).
Backhouse, S., Collins, A. V. & Phillips, J. R. Influence of periodic versus continuous daily bright light exposure on development of experimental myopia in the chick. Ophthalmic Physiol. Opt. J. Br. Coll. Ophthalmic Opt. (Optom.) 33, 563–572. https://doi.org/10.1111/opo.12069 (2013).
Ding, B. Y., Shih, Y. F., Lin, L. L. K., Hsiao, C. K. & Wang, I. J. Myopia among schoolchildren in East Asia and Singapore. Surv. Ophthalmol. 62, 677–697. https://doi.org/10.1016/j.survophthal.2017.03.006 (2017).
Morgan, I. G. & Rose, K. A. Myopia: Is the nature-nurture debate finally over?. Clin. Exp. Optom. 102, 3–17. https://doi.org/10.1111/cxo.12845 (2019).
Smith, E. L. 3rd., Hung, L. F., Arumugam, B. & Huang, J. Negative lens-induced myopia in infant monkeys: Effects of high ambient lighting. Investig. Ophthalmol. Vis. Sci. 54, 2959–2969. https://doi.org/10.1167/iovs.13-11713 (2013).
Rahi, J. S., Cumberland, P. M. & Peckham, C. S. Myopia over the lifecourse: Prevalence and early life influences in the 1958 British birth cohort. Ophthalmology 118, 797–804. https://doi.org/10.1016/j.ophtha.2010.09.025 (2011).
Wang, Z. et al. Risk factors for astigmatic components and internal compensation: The Nanjing Eye Study. Eye (Lond., Engl.) 35, 499–507. https://doi.org/10.1038/s41433-020-0881-5 (2021).
Autti-Rämö, I. et al. Fetal alcohol spectrum disorders in Finland: Clinical delineation of 77 older children and adolescents. Am. J Med. Genet. Part A 140, 137–143. https://doi.org/10.1002/ajmg.a.31037 (2006).
Fernandes, M., Yang, X., Li, J. Y. & CheikhIsmail, L. Smoking during pregnancy and vision difficulties in children: A systematic review. Acta Ophthalmol. 93, 213–223. https://doi.org/10.1111/aos.12627 (2015).
McKean-Cowdin, R. et al. Risk factors for astigmatism in preschool children: The multi-ethnic pediatric eye disease and Baltimore pediatric eye disease studies. Ophthalmology 118, 1974–1981. https://doi.org/10.1016/j.ophtha.2011.06.031 (2011).
Hin, S. A., Reginald, L. K. & Lim, T. C. Acute renal failure in Reye’s syndrome. J. Singap. Paediatr. Soc. 32, 36–39 (1990).
Iyer, J. V., Low, W. C., Dirani, M. & Saw, S. M. Parental smoking and childhood refractive error: The STARS study. Eye (Lond., Engl.) 26, 1324–1328. https://doi.org/10.1038/eye.2012.160 (2012).
Saw, S. M., Chia, K. S., Lindstrom, J. M., Tan, D. T. & Stone, R. A. Childhood myopia and parental smoking. Br. J. Ophthalmol. 88, 934–937. https://doi.org/10.1136/bjo.2003.033175 (2004).
Shi, Y. et al. Ethnic disparities in risk factors for myopia among Han and minority schoolchildren in Shawan, Xinjiang, China. Optom. Vis. Sci. Off. Publ. Am. Acad. Optom. 100, 82–90. https://doi.org/10.1097/opx.0000000000001949 (2023).
Wang, X. et al. Prevalence and risk factors of myopia in Han and Yugur older adults in Gansu, China: A cross-sectional study. Sci. Rep. 10, 8249. https://doi.org/10.1038/s41598-020-65078-x (2020).
Menéndez-Acebal, C. et al. The influence of alcohol intake in myopia development or progression: The SUN cohort study. Drug Alcohol Depend. 229, 109149. https://doi.org/10.1016/j.drugalcdep.2021.109149 (2021).
Majhi, D. et al. Prevalence and causes of visual impairment in Dongaria indigenous (tribal) community. Tribal Odisha eye disease study # 12. Indian J. Ophthalmol. 71, 2850–2855. https://doi.org/10.4103/ijo.Ijo_2788_22 (2023).
Lyubasyuk, V., Jones, K. L., Caesar, M. A. & Chambers, C. Vision outcomes in children with fetal alcohol spectrum disorders. Birth Defects Res. 115, 1208–1215. https://doi.org/10.1002/bdr2.2223 (2023).
Mountjoy, E. et al. Education and myopia: Assessing the direction of causality by Mendelian Randomisation. BMJ (Clin. Res. ed.) 361, k2022. https://doi.org/10.1136/bmj.k2022 (2018).
Zhou, H. et al. Education and lung cancer: A Mendelian randomization study. Int. J. Epidemiol. 48, 743–750. https://doi.org/10.1093/ije/dyz121 (2019).
Sanderson, E. et al. Mendelian randomization. Nat. Rev. Methods Primers 2, 6. https://doi.org/10.1038/s43586-021-00092-5 (2022).
Author information
Authors and Affiliations
Contributions
Author 1.3.4 (Diyao Wei,Ling Huang and Minghui Hou):participated in data curation and drafting of the manuscript. Author 2 (Huangyan Wang): corresponding author Author 5.6.7(Hong-Gang Liang,Xiang Shi and Xianghui Wei):prepared figures 1-4 Author 8.9.10.11.12(Jingrong Li,Liuzhu Gan,Bi Lv,Jiabi Deng and,Lulu Qing):prepared Table 1-5.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wei, D., Wang, H., Huang, L. et al. A Mendelian randomization study on the causal relationship between smoking, alcohol consumption, and the development of myopia and astigmatism. Sci Rep 14, 1868 (2024). https://doi.org/10.1038/s41598-024-52316-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-52316-9
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.