Abstract
Adrenal cortex autotransplantation with ACTH stimulation may be an alternative therapy for patients with bilateral adrenalectomy to avoid adrenal crisis, but its underlying mechanism has not been elucidated. Previously, we detected Dhh upregulation in rat adrenocortical autografts after transplantation. Here, we investigated potential regulators such as Gata4, Gata6, Sry and Sox9 which affect Dhh transcription in adrenocortical autografts with or without ACTH stimulation. In ACTH-stimulated autografts, Gata4 and Gata6 were downregulated compared to control autografts. This response was linked to rDhh repression. A reporter assay using the upstream region of rDhh and a GATA binding motif revealed that rDhh promoters were significantly upregulated by co-transfection with Gata4 or Gata6 or both. Sry and Sox9 expression in autografts with or without ACTH stimulation were verified by PCR and RNAscope analyses. The ovarian differentiation factors Foxl2 and Rspo1 were also upregulated in the autografts. Gata4 and Gata6 were found to be significant factors in the regulation of rDhh expression and could be associated with adrenocortical autograft maintenance. Gonadal primordia with bipotential testicular and ovarian functions may also be present in these autografts.
Similar content being viewed by others
Introduction
Pheochromocytomas arise from the adrenal medulla and are catecholamine-producing tumours. Hereditary pheochromocytoma can be treated with bilateral adrenalectomy and lifelong glucocorticoid replacement therapy1. Autotransplantation and allotransplantation of the adrenal cortex are potential alternatives that allow bilateral adrenalectomy patients to avoid adrenal crises1,2. However, adrenal autotransplantation has not been established in humans and its success rate is only 20–35%3,4. Possible reasons for this poor performance include ACTH suppression by negative feedback from excessive postoperative glucocorticoid replacement therapy. This response causes autograft regression. According to previous reports, adrenal autotransplantation has been highly successful in the management of Cushing’s disease (ACTH hypersecretion from the pars distalis)5,6,7. Four patients who underwent bilateral adrenalectomy and ACTH replacement were able to withdraw from glucocorticoid replacement immediately after adrenal autotransplantation8. Dexamethasone-induced adrenal atrophy in mice was restored with daily ACTH stimulation9. ACTH stimulation after autotransplantation preserves autografts and may involve an unidentified pathway which promotes adrenal cortical regeneration and recovers endocrine function.
In the search for factors affecting post-transplant adrenocortical autograft remodelling and regeneration, we found that Dhh was upregulated and Shh was downregulated in the regeneration step of rat adrenocortical autograft8. The HH signalling pathway may participate in adrenocortical autograft regeneration as well as adrenal cortex development. The regulation of Dhh transcription during gonadal development involves transcription factors such as Wt1, Gata4, Gata6, Sox9 and Sry9,10. In this study, we examined whether they affect Dhh transcription in adrenocortical autografts. Although ACTH stimulation is important, to the best of our knowledge, no studies have evaluated the influence of transient ACTH stimulation on adrenal autografts. Therefore, we also assessed the effects of transient ACTH and rDhh transcription-associated factors.
Results and Discussion
ACTH stimulation induced angiogenic factor in adrenal glands
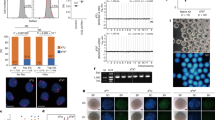
After ACTH stimulation for 2 h, Vegfa showed a 1.8-fold increase in the adrenal gland compared with that in the intact group (Fig. 1). Angpt1 in the adrenal gland after ACTH stimulation showed a 0.5-fold decrease compared with that in the intact group (Fig. 1). There was no difference between the intact and saline groups in terms of Vegfa and Angpt1 expression (Fig. 1). Further, no differences in Angpt2 expression among the three groups were observed (Fig. 1). These results corroborated those of previous reports11,12,13. Even the 2-h ACTH stimulation of the adrenal gland in the present study suggested that ACTH regulates angiogenic factors which could affect adrenal autograft conditions.
Relative expression of angiogenesis factors in adrenal glands. Rats without injection (Intact group), injected with natural saline (Saline group) or injected with ACTH (ACTH group) were euthanized after 2 h. Relative expression levels in the adrenal glands were evaluated by RT-qPCR. Changes in transcription level were analysed by ANOVA with a Steel multiple comparisons test *P < 0.05 vs. Intact group. Vegfa: vascular endothelial growth factor a; Angpt: Angiopoietin.
ACTH suppressed the HH signal in the adrenal autografts
RNAscope analysis in our previous study confirmed that Shh was downregulated and Dhh was upregulated in the autografts 2–3 wks after surgery10. Similarly, Shh expression showed a 0.05-fold decrease in the control autografts compared to that in the sham (Fig. 2). Dhh expression was 5-fold higher in the autografts than in the sham (Fig. 2). In the ACTH-stimulated autograft, neither Shh nor Dhh was upregulated in the adrenocortical autografts relative to the sham. Similar results were observed for Gil1 expression (4.2-fold increase in the control autografts, no difference in the ACTH-stimulated autograft) (Fig. 2). Therefore, 2-h ACTH stimulation dysregulated HH signal-related genes in the autografts 2 wks after surgery.
Relative expression of HH signalling molecules in adrenal tissues. Rats with Sham operation (Sham), adrenocortical autotransplantation (Control group) or adrenocortical autotransplantation plus ACTH (ACTH group) were sacrificed at POD14. Relative expression levels in the adrenal tissues were evaluated by RT-qPCR. Changes in transcription level were analysed by ANOVA with a Steel multiple comparisons test; *P < 0.05 vs. Sham rats. For non-parametric factors such as Shh and Dhh, the Mann-Whitney U test with a Bonferroni correction was used; †P < 0.0167 vs. Sham rats. Kif7: kinesin family member 7; Ptch1: human patched-1; Shh: sonic hedgehog; Smo: Smoothened; Sufu: Sufu negative regulator of hedgehog signalling; Dhh: desert hedgehog; Gli1: GLI family zinc finger 1; Disp1: Dispatched RND Transporter Family Member 1.
Disp1 encodes HH ligand secretion receptors and is co-localised with the HH ligand in the same cells14. Disp1 showed a 2.1-fold upregulation in the control autograft but a 0.6-fold downregulation by ACTH stimulation (Fig. 2). Therefore, both DHH synthesis and release were suppressed in the DHH-producing cells of the ACTH-stimulated autograft. On postoperative day (POD)14, there might be slight HH ligand binding in the autograft HH target cells.
Transcriptional Dhh regulator and effect of ACTH stimulation
The expression of certain Dhh transcriptional regulators may be linked to Dhh expression in the 2–3 wks after surgery, during which time Dhh was upregulated. Therefore, we measured the expression levels of candidate transcription factors in adrenal autografts at POD14.
Wt1 was upregulated in the control- (4.0-fold) and ACTH-stimulated (4.5-fold) adrenal autografts compared with that in the sham adrenal gland (Fig. 3). We were, then, the first to identify Sry and Sox9 expression in adult adrenal cortex using their cDNA in qRT-PCR. Both were elevated in autografts independently of ACTH stimulation (Sry: 4.3-fold change, Sox9: 3.4-fold change) (Fig. 3). Cycle sequence analysis disclosed that these PCR products were indeed Sry and Sox9. RNAscope analysis also confirmed that Sry and Sox9 were localised in the adrenal gland and the autograft. Sry and Sox9 were detected in the zona glomerulosa (ZG) and the estimated undifferentiated zone (ZU), respectively (Fig. 4A,B; Supplementary Fig. 1). Sry was detected at low levels in the capsule and the zona fasciculate (ZF). In the adrenocortical autograft, Sry was expressed in the stromal cells adjacent to the remnant adrenocortical cells. It was also found in the remnant adrenocortical cells around the capillary circumference at POD14. GATA4 and GATA6 are Shh transcriptional regulators in the limb bud15. Gata4 showed a 2.6-fold increase in the autograft (Fig. 3). In the ACTH-stimulated autograft, the transcription factors remained upregulated but both Gata4 (0.5-fold change) and Gata6 (0.6-fold change) were inhibited from linking to Dhh compared with that in the control autografts (Gata4: 0.5-fold change, Gata6: 0.6-fold change) (Fig. 3). Therefore, Gata4 and/or Gata6 were considered Dhh regulators in the adrenal autografts. To clarify this hypothesis, a luciferase reporter assay was conducted on the proximal upstream region of the rat Dhh gene including the GATA binding motif at −226/−220. Co-transfection of the rDhh-2000/+50 region (Dhh_−2000/+50_pGL4.10) with the pCMV6-Entry vector significantly increased luciferase activity by ~8× in the H295R cells, human Adrenal gland carcinoma cell line (Fig. 5). Therefore, there may be transcriptional activity in the Dhh upstream region even in adrenocortical cells. The rDhh promoter was significantly upregulated by co-transfection with Gata4 and/or Gata6 in H295R cells (Fig. 5). GATA binding motifs are conserved in the upstream region of mouse Dhh (−230/−224) and human DHH (−1024/−1027, −1031/−1034, −1075/−1081). For this reason, transcriptional regulation of adrenal Dhh expression by GATA4 and GATA6 might be important in both species. There is a TATA box-like sequence at −517/−514 and typical CCAAT boxes at −610/−606 and −758/−754. Nevertheless, in silico analysis suggested that they do not initiate Dhh transcription at their distance and that a different transcription initiation site may be present. The typical GC boxes at −49/−44, −27/−22 and −19/−15 were deemed potential promoter region sites. The GATA binding motif could affect this GC boxes activities in H295R cells (Supplementary Fig. 2).
Relative expression of transcriptional regulators in adrenal tissues. Rats with Sham operation (Sham), adrenocortical autotransplantation (Control group) or adrenocortical autotransplantation plus ACTH (ACTH group) were sacrificed at POD14. Relative expression levels in the adrenal tissues were evaluated by RT-qPCR. Changes in transcription level were analysed by ANOVA with a Steel multiple comparisons test *P < 0.05 vs. Sham rats. Wt1: Wilms tumour 1; Sry: Sex-determining region Y; Sox9: SRY-box 9; Gata: Gata-binding factor.
RNAscope in adrenal gland with sham. Sry expression was detected in the adrenal layers especially ZU and ZG. (A) 004Cow-power field; (B) High-power field. In situ hybridisation with RNAscope in the adrenocortical autograft at POD14. Sry expression persisted in the autograft at POD14. (C) Low-power field, (D) High-power field. Cap: capsule; ZG: zona glomerulosa; ZF: zona fasciculate; ZU: undifferentiated zone; RAC: renewal adrenocortical cells.
Luciferase activity in the rat Dhh upstream region of H295R cells. Four days after transfection, cells were lysed and luciferase activity was measured. Statistical comparisons between the pGL4.10 (pGL4.10) and the Dhh_−2000/+50-pGL4.10 (Dhh_upstream) were performed by Student’s t test; *P < 0.05 vs. the pGL4.10. Multiple comparisons of luciferase activity between the pCMV6-Entry (Mock) and the GATA4, GATA6 and GATA4 + GATA6 were performed using ANOVA with a Bonferroni correction; †P < 0.05 vs. Mock. Dhh, Desert Hedgehog; Gata: Gata-binding factor. N = 6 in each condition.
Effect of ACTH on adrenocortical regeneration
Foetal adrenocortical cells are derived from Gli1-expressing cells16. We examined the effects of ACTH on the factors determining adrenal development and steroidogenesis in autografts wherein ACTH repressed Gli1.
In ACTH-stimulated autografts, Nr5a1/Sf1 and Dax1 were not significantly upregulated relative to that in the sham group (Fig. 6). However, ACTH stimulation did not change the level of the stromal markers Nr2f2, Tcf21 and Pdgfra compared with that in the control autografts (Fig. 6). Dhh and Gli1 might be involved in adrenocortical cell differentiation and alter the levels of Nr5a1/Sf1 and Dax1 in adrenocortical autograft cells8. In contrast, the capsule cells could be under the control of other regulators such as WT1. Transient ACTH stimulation had a conflicting effect on the regenerations of autograft capsule and adrenocortical cells.
Relative expression of regeneration factors in adrenal tissues. Rats with Sham operation (Sham), adrenocortical autotransplantation (Control group) or adrenocortical autotransplantation plus ACTH (ACTH group) were sacrificed at POD14. Relative expression levels in the adrenal tissues were evaluated by RT-qPCR. Changes in transcription level were analysed by ANOVA with a Steel multiple comparisons test; *P < 0.05 vs. Sham rats. Nr5a1/Sf1: nuclear receptor subfamily 5 group A member 1/steroidogenic factor 1; Nr0b1/Dax1: nuclear receptor subfamily 0 group B member 1/dosage-sensitive sex reversal, adrenal hypoplasia critical region, on chromosome X, gene 1; Hoxb9: homeobox B9 Nr2f2: nuclear receptor subfamily 2 group f member 2; Tcf21: transcription factor 21; Pdgfra: platelet-derived growth factor receptor alpha; Cited2: Cbp/p300 interacting transactivator with Glu/Asp-rich carboxy terminal domain 2.
CITED2 co-ordinately controls Nr5a1/Sf1 mRNA accumulation in the adrenogenital primordium (AGP) along with WT117. We found that Cited2 was not upregulated in the autograft (0.4-fold change) (Fig. 6). Therefore, CITED2 might not participate in Nr5a1/Sf1 regulation there. Hoxb9 is an adrenal steroidogenic cell marker in the AGP18. In the autograft, Hoxb9 was also downregulated (0.1-fold change) (Fig. 6). For this reason, adrenal steroidogenic-like cells in the AGP might be absent in the autografts at POD14. In other words, Nr5a1/Sf1 found in the autografts at POD14 might be expressed in not newly generated adrenocortical cells, but in survival adrenocortical cells.
Adrenocortical autograft as a bipotential gonad?
Gonads and adrenal glands that originated from an AGP appeared in the form of a coelomic epithelium between the urogenital ridge and the dorsal mesentery. AGP can be detected at embryonic day (E) 9.0 in mice19. Adrenal- and gonad anlages progressively individualise from E9.5 to E10.5 and are distinct by E13. Primordial germ cells reach the sexually undetermined gonadal anlage stage by E10. After E11.5-E12, the bipotent gonad differentiates into the testis or ovary with or without Sry and by Sox9 upregulation or downregulation.
We used gonadal marker detection to investigate whether gonadal differentiation occurs in adrenocortical autografts. Normally, rodent Cyp17a1 is suppressed in the adrenal gland by a DNA methylation mechanism but is expressed in the gonad and placenta20. Even in the present study, we found no Cyp17a1 expression in the adrenal glands of sham-operated rats. However, the adrenocortical autografts at POD14 presented with relatively upregulated Cyp17a1 (Fig. 7). Autografts might show changes in their DNA methylation patterns only, but remain adrenal glands with remaining adrenocortical cells, without differentiating to gonadal tissues. We then examined other gonadal markers such as AMHR2 which is specific to AMH target tissues21. With or without ACTH stimulation, Amhr2 was upregulated in a 2.6-fold change in adrenal autografts relative to sham rats (Fig. 7). Amhr2 is expressed in the developing gonads and Mullerian ducts where it mediates AMH-induced regression22,23. In the gonad, AMH signalling promotes masculinisation by suppressing ovary-associated processes such as germ cell meiosis and aromatase and Lhcgr expression24. In the present study, Lhcgr showed a 4.8-fold upregulation in the autografts (Fig. 7) even though the gonad was intact in the adrenocortical autografted rat, unlike the Lhcgr upregulation found in post-gonadectomy-induced adrenal hyperplasia. Our histological analysis showed no obvious adrenal hyperplasia in the autografts8.
Relative expression of gonadal markers in adrenal tissues. Rats with Sham operation (Sham), adrenocortical autotransplantation (Control group) or adrenocortical autotransplantation plus ACTH (ACTH group) were sacrificed at POD14. Relative expression levels in adrenal tissues were evaluated by RT-qPCR. Changes in transcription level were analysed by ANOVA with a Steel multiple comparisons test *P < 0.05 vs. Sham rats. Cyp17a1: cytochrome P450 family 17 subfamily A member 1; Amhr2: anti-Mullerian hormone type 2 receptor; Lhcgr: luteinising hormone/choriogonadotropin receptor; Foxl2: forkhead box L2; Rspo1: R-spondin 1; Vnn1: vanin 1.
FOXL2 is a differentiation factor in the ovary and represses male-specific genes such as Sox9 there25. Foxl2 showed a 2.8-fold increase in the present study even though the male determinant factor Sox9 was elevated in the autografts (Fig. 7). Foxl2 is first detected by the end of the sex determination period26. Therefore, undifferentiated gonads occurred in the autografts. The ovarian determiner RSPO127 was also upregulated in the autografts (7.1-fold change) (Fig. 7). Here, feminisation occurred even in the presence of the male adrenal gland. On the other hand, Vnn1, a marker for steroidogenic Sertoli, Leydig and adrenocortical cells28,29 and a protectant against oxidative stress30, was downregulated in the autografts (0.4-fold decrease) (Fig. 7). Undifferentiated bipotential gonad-like tissues with few or no steroidogenic cells were found in the adrenocortical autograft at POD14. Therefore, gonadal primordia with bipotential testicular and ovarian functions31 could be present in autografts.
Conclusion
This report is the first to clearly demonstrate GATA4 and GATA6 were transcription factors in the regulation of Dhh expression in rat adrenocortical autografts. In the present study, only male rats were used and further examinations using female rats are needed to assess the difference of sex. Additionally, transient ACTH stimulation is not considered effective for maintaining the level of Dhh in autografts. Thus, future experiments with the long-term ACTH stimulation for autograft need to be performed.
Methods
Animals
Twenty-six male Wistar rats (age 7 wks, weight 210–270 g) were purchased from Shimizu Laboratory Supplies (Kyoto, Japan) and housed in a sound-attenuated, light-controlled room (12 h light-dark cycle: lights on at 8:00 and off at 20:00; constant 25 ± 1 °C and 50 ± 10% relative humidity) for 2 wks before the operation. Food and water were provided ad libitum. All animal experiments were approved by the Ethics Committee on Animal Experiments at Kansai Medical University (Approval No. 17–051) and conducted in accordance with the Guide for the Care and Use of Laboratory Animals of the Institute for Laboratory Animal Research.
Effects of 2 hours ACTH stimulation on adrenal glands
Four male Wistar rats (age 9 wks, weight 210–240 g) received subcutaneous injections of tetracosactide acetate (ACTH 1–24; 20 μg/200 g BW) dissolved in 0.9% w/v saline (the ACTH group). Four other rats received subcutaneous injections of 0.9% w/v saline (the saline group) between 8:00 and 9:00 were decapitated after 2 h. Four age-sex matched rats were used as the intact group. Between 10:00 and 11:00, their adrenal glands were rapidly excised and stored at −80 °C until RNA isolation.
Adrenocortical autotransplantation
Adrenocortical autotransplantation was performed in twelve 9-wk-old rats as previously described32. Briefly, bilateral adrenal glands were resected and divided into four pieces and the medullae were discarded. Adrenocortical autografts were autotransplanted with a pair of fine scissors into two abdominal muscle pockets or the right biceps femoris. Sham operations without adrenalectomy were performed simultaneously. Animals were maintained on saline without any glucocorticoid replacement following the adrenalectomy33. ACTH stimulation was performed by injecting tetracosactide acetate (20 μg/200 g BW) dissolved in 0.9% w/v saline 2 h before adrenocortical autotransplantation into abdominal muscle pockets. One rat with ACTH stimulation died within 2 wks after surgery. Two wks after surgery, the adrenal tissues were rapidly excised for gene expression and histological analyses.
Reverse transcription and quantitative (RT-q) PCR
Total RNA was isolated from each adrenal tissue sample with Sepasol-RNA I Super G reagent (Nacalai Tesque, Kyoto, Japan). Single-strand cDNA was synthesised with a PrimeScript RT reagent kit and gDNA Eraser (TaKaRa Bio Inc., Kyoto, Japan). The mRNA expression levels were determined by quantitative PCR on a Roter-Gene Q platform (Qiagen, Venlo, The Netherlands) using Thunderbird qPCR Mix (Toyobo, Fukui, Japan) and the gene-specific primers listed in Table 1. To test the amplification efficiencies for primer pairs, a 1:10 dilution was used to create a serial dilution series with the undiluted rat adrenal gland or testis cDNA as a starting point. We calculated the amplification efficiency for each primer pair (Table 1). Relative target gene expression levels were evaluated by the 2−ΔΔCt method34 using Hprt1 as an internal control according to previous our studies8,32. The 2−ΔΔCt method assumes that primer amplification efficiencies are similar (usually between 90–110%) among target genes and the internal control. All primers efficiencies except those for Angpt2 and Nr5a1/Sf1 ranged from 90 to 110%. We therefore applied the Pfaffl method35, which can account for any differences in efficiency, for Angpt2 and Nr5a1/Sf1 to confirm their reproducibility.
RNAscope
Adrenal glands and autografts were fixed by immersion in 4% formaldehyde with 0.1 M phosphate buffer (pH 7.4) at 4 °C overnight. The tissues were immersed in 30% w/v sucrose solution for cryoprotection. Fixed, frozen tissues were embedded in optimal cutting temperature compound, cut into 10-µm-thick sections and mounted on Superfrost Plus slides (Thermo Fisher Scientific, Waltham, MA, USA). The sections were air-dried at −20 °C and stored at −80 °C until use. When required, they were returned to 20–25 °C, washed once with distilled water and baked at 60 °C for 30 min. In situ hybridisation was conducted using the RNAscope 2.5HD Reagent Kit (Singleplex, RED; ACD LLC, Santa Ana, CA, USA), Probe-Rn-Sry (ACD LLC, Santa Ana, CA, USA), Probe-Rn-Sox9 (ACD LLC, Santa Ana, CA, USA) and Positive Control Probe-Rn-Polr2a (ACD LLC, Santa Ana, CA, USA) or Negative Control Probe-DapB (ACD LLC, Santa Ana, CA, USA) according to the manufacturer’s protocol. After the final amplification, fast-red chromogenic detection was performed. The slides were treated with DAPI Fluoromount-G (SouthernBiotech, Birmingham, AL, USA) and sealed with coverslips. The Sry and Sox9 signals were visualised under a confocal laser microscope (LMS700; Carl Zeiss AG, Oberkochen, Germany). Bright-field images of the fast-red staining were captured with an Eclipse E-1000M digital camera (Nikon, Tokyo, Japan).
Expression vector and reporter plasmid
The reporter plasmids pGL4.10[luc2] and pGL4.74[hRluc/TK] were purchased from Promega (Madison, WI, USA). The pGL4.74[hRluc/TK] which encodes Renilla luciferase was used as an internal control for transfection efficiency. The 2,000-bp upstream region and the 50-bp downstream region of the Dhh transcription start site10 were amplified with KOD Fx, rDhh_-2000F_SacI (5′-TCCGAGCTCctgagcaagccatgaggagca-3′; the SacI site is underlined) and rDhh_+50R_BglII (5′-GAAGATCTggtttctgctgcccagctccgg-3′; the BglII site is underlined). The PCR products were cloned into pGL4.10[luc2] (Dhh_−2000/+50_pGL4.10).
The expression, pCMV6-Entry, rGATA4-pCMV6 and rGATA6-pCMV6 vectors were purchased from Origen (Rockville, MD, USA). Rat Gata4 and Gata6 were subcloned into pCMV6-Entry (rGATA4-pCMV6) and pCMV6-Entry (rGATA6-pCMV6), respectively.
All constructs were confirmed to have no mutation, no insertion, and no deletion by sequencing analysis with a BigDye Terminator Cycle Sequencing Reaction Kit (Applied Biosystems, Foster City, CA, USA) and an ABI PRISM 3100 Genetic Analyzer (Applied Biosystems, Foster City, CA, USA). Both strands were read with sequence primers.
Luciferase reporter assay
H295R cells (human adrenal carcinoma) were grown in DMEM:F12 medium (Thermo Fisher Scientific, Tokyo, Japan) supplemented with 6.25 ng/mL each of insulin, transferrin and selenium, 1.25 mg/mL bovine serum albumin (BSA) and 5.35 ng/mL linoleic acid at 37 °C and 5% CO2. The final concentration was adjusted to 2.5% with Nu-Serum I (Corning Inc., Corning, NY, USA).
Cells were seeded in 24-well cell culture plates at a density of 2.5 × 105/well. Two types of luciferase plasmids and one expression vector were co-transfected with LipofectamineTM 3000 Transfection Reagent (Thermo Fisher Scientific, Tokyo, Japan) according to the manufacturer’s protocol. The following amounts of co-transfected plasmids and vectors were placed in each well: 200 ng firefly luciferase-encoding reporter plasmid (pGL4.10), 20 ng Renilla luciferase-encoding internal control plasmid (pGL4.74) and 200 ng expression vectors (pCMV6-Entry, rGata4-pCMV6, or rGata6-pCMV6). Approximately 96 h after transfection, luciferase activity was sequentially measured in duplicate using a PicaGene Dual Sea Pansy Luminescence Kit (TOYO INK CO. Ltd., Chuo-ku, Tokyo, Japan) and the 2030ARVO X multilabel reader (PerkinElmer Japan Co. Ltd., Yokohama, Japan) according to the manufacturer’s protocol. Briefly, 20 µL cell lysate per well in passive lysis buffer was transferred to an OptiPlate-96 plate (SUMILON, Tokyo, Japan). Firefly luciferase luminescence (FLU) from the pGL4.10 plasmids and Renilla luciferase luminescence (RLU) from the pGL4.74 plasmid were measured independently. Relative luciferase activity per well was calculated by dividing FLU by RLU. Relative luciferase activity was standardised by the corresponding control conditions, namely, either co-transfection of a pGL4.10 plasmid with a pCMV6-Entry, rGATA4-pCMV6, rGATA6-pCMV6 or rGATA4-pCMV6 with rGATA6-pCMV6 vectors. Activity levels were expressed as the mean of ≥6 independent experiments ± standard error (SE).
Statistical analysis
Normal distribution was analysed by the Shapiro-Wilk normality test. ANOVA with a Steel multiple comparisons test was used for the normally distributed factors. For Shh and Dhh data, which were not normally distributed, the Mann-Whitney U test with a Bonferroni correction was used. Comparisons between pGL4 and Dhh were performed by Student’s t test. Multiple comparisons between the Mock-Dhh and the other Dhh were performed using ANOVA with a Bonferroni correction. Statistical analyses were performed in IBM SPSS Statistics v. 21.0 (IBM Corp., Armonk, NY, USA). All values were two-sided with statistical significance set at 0.05.
References
Lenders, J. W. et al. Pheochromocytoma and paraganglioma: an endocrine society clinical practice guideline. The Journal of clinical endocrinology and metabolism 99, 1915–1942, https://doi.org/10.1210/jc.2014-1498 (2014).
Hahner, S. et al. High incidence of adrenal crisis in educated patients with chronic adrenal insufficiency: a prospective study. The Journal of clinical endocrinology and metabolism 100, 407–416, https://doi.org/10.1210/jc.2014-3191 (2015).
Erdogan, G. et al. Adrenal autotransplantation after total adrenalectomy: delayed determined function. Endocrine journal 41, 45–48 (1994).
Okamoto, T. et al. Bilateral adrenalectomy with autotransplantation of adrenocortical tissue or unilateral adrenalectomy: treatment options for pheochromocytomas in multiple endocrine neoplasia type 2A. Endocrine journal 43, 169–175 (1996).
Birke, G., Franksson, C., Moberger, G. & Plantin, L. O. Storage and autotransplantation of human adrenal tissue. Acta chirurgica Scandinavica 111, 113–123 (1956).
Ibbertson, H. K. & O’Brien, K. P. Adrenal autografts in treatment of Cushing’s disease. British medical journal 2, 703–706 (1962).
Xu, Y. M., Chen, Z. D., Qiao, Y. & Jin, N. T. The value of adrenal autotransplantation with attached blood vessels for the treatment of Cushing’s disease: a preliminary report. The Journal of urology 147, 1209–1211 (1992).
Takizawa, N. et al. Involvement of DHH and GLI1 in adrenocortical autograft regeneration in rats. Scientific reports 8, 14542, https://doi.org/10.1038/s41598-018-32870-9 (2018).
Rudigier, L. J., Dame, C., Scholz, H. & Kirschner, K. M. Ex vivo cultures combined with vivo-morpholino induced gene knockdown provide a system to assess the role of WT1 and GATA4 during gonad differentiation. PloS one 12, e0176296, https://doi.org/10.1371/journal.pone.0176296 (2017).
Li, Y., Zheng, M. & Lau, Y. F. The sex-determining factors SRY and SOX9 regulate similar target genes and promote testis cord formation during testicular differentiation. Cell reports 8, 723–733, https://doi.org/10.1016/j.celrep.2014.06.055 (2014).
Shifren, J. L., Mesiano, S., Taylor, R. N., Ferrara, N. & Jaffe, R. B. Corticotropin regulates vascular endothelial growth factor expression in human fetal adrenal cortical cells. The Journal of clinical endocrinology and metabolism 83, 1342–1347, https://doi.org/10.1210/jcem.83.4.4730 (1998).
Gaillard, I. et al. ACTH-regulated expression of vascular endothelial growth factor in the adult bovine adrenal cortex: a possible role in the maintenance of the microvasculature. Journal of cellular physiology 185, 226–234, 10.1002/1097-4652(200011)185:2<226::AID-JCP7>3.0.CO;2-E (2000).
Feraud, O., Mallet, C. & Vilgrain, I. Expressional regulation of the angiopoietin-1 and -2 and the endothelial-specific receptor tyrosine kinase Tie2 in adrenal atrophy: a study of adrenocorticotropin-induced repair. Endocrinology 144, 4607–4615, https://doi.org/10.1210/en.2003-0099 (2003).
Briscoe, J. & Therond, P. P. The mechanisms of Hedgehog signalling and its roles in development and disease. Nature reviews. Molecular cell biology 14, 416–429, https://doi.org/10.1038/nrm3598 (2013).
Kozhemyakina, E., Ionescu, A. & Lassar, A. B. GATA6 is a crucial regulator of Shh in the limb bud. PLoS genetics 10, e1004072, https://doi.org/10.1371/journal.pgen.1004072 (2014).
Wood, M. A. et al. Fetal adrenal capsular cells serve as progenitor cells for steroidogenic and stromal adrenocortical cell lineages in M. musculus. Development 140, 4522–4532, https://doi.org/10.1242/dev.092775 (2013).
Val, P., Martinez-Barbera, J. P. & Swain, A. Adrenal development is initiated by Cited2 and Wt1 through modulation of Sf-1 dosage. Development 134, 2349–2358, https://doi.org/10.1242/dev.004390 (2007).
Zubair, M., Ishihara, S., Oka, S., Okumura, K. & Morohashi, K. Two-step regulation of Ad4BP/SF-1 gene transcription during fetal adrenal development: initiation by a Hox-Pbx1-Prep1 complex and maintenance via autoregulation by Ad4BP/SF-1. Molecular and cellular biology 26, 4111–4121, https://doi.org/10.1128/MCB.00222-06 (2006).
Ikeda, Y., Shen, W. H., Ingraham, H. A. & Parker, K. L. Developmental expression of mouse steroidogenic factor-1, an essential regulator of the steroid hydroxylases. Mol Endocrinol 8, 654–662, https://doi.org/10.1210/mend.8.5.8058073 (1994).
Missaghian, E. et al. Role of DNA methylation in the tissue-specific expression of the CYP17A1 gene for steroidogenesis in rodents. The Journal of endocrinology 202, 99–109, https://doi.org/10.1677/JOE-08-0353 (2009).
Josso, N. & Clemente, N. Transduction pathway of anti-Mullerian hormone, a sex-specific member of the TGF-beta family. Trends in endocrinology and metabolism: TEM 14, 91–97 (2003).
Baarends, W. M. et al. A novel member of the transmembrane serine/threonine kinase receptor family is specifically expressed in the gonads and in mesenchymal cells adjacent to the mullerian duct. Development 120, 189–197 (1994).
Teixeira, J. & Donahoe, P. K. Molecular biology of MIS and its receptors. Journal of andrology 17, 336–341 (1996).
Josso, N., Racine, C., di Clemente, N., Rey, R. & Xavier, F. The role of anti-Mullerian hormone in gonadal development. Molecular and cellular endocrinology 145, 3–7 (1998).
Veitia, R. A. FOXL2 versus SOX9: a lifelong “battle of the sexes”. BioEssays: news and reviews in molecular, cellular and developmental biology 32, 375–380, https://doi.org/10.1002/bies.200900193 (2010).
Pannetier, M., Chassot, A. A., Chaboissier, M. C. & Pailhoux, E. Involvement of FOXL2 and RSPO1 in Ovarian Determination, Development, and Maintenance in Mammals. Sexual development: genetics, molecular biology, evolution. endocrinology, embryology, and pathology of sex determination and differentiation 10, 167–184, https://doi.org/10.1159/000448667 (2016).
Chassot, A. A. et al. Activation of beta-catenin signaling by Rspo1 controls differentiation of the mammalian ovary. Human molecular genetics 17, 1264–1277, https://doi.org/10.1093/hmg/ddn016 (2008).
Bowles, J., Bullejos, M. & Koopman, P. A subtractive gene expression screen suggests a role for vanin-1 in testis development in mice. Genesis 27, 124–135 (2000).
Wilson, M. J., Jeyasuria, P., Parker, K. L. & Koopman, P. The transcription factors steroidogenic factor-1 and SOX9 regulate expression of Vanin-1 during mouse testis development. The Journal of biological chemistry 280, 5917–5923, https://doi.org/10.1074/jbc.M412806200 (2005).
Latre de Late, P. et al. Vanin-1 inactivation antagonizes the development of adrenocortical neoplasia in Sf-1 transgenic mice. Endocrinology 155, 2349–2354, https://doi.org/10.1210/en.2014-1088 (2014).
Eggers, S., Ohnesorg, T. & Sinclair, A. Genetic regulation of mammalian gonad development. Nature reviews. Endocrinology 10, 673–683, https://doi.org/10.1038/nrendo.2014.163 (2014).
Takizawa, N. et al. Hypothalamohypophysial system in rats with autotransplantation of the adrenal cortex. Molecular medicine reports 15, 3215–3221, https://doi.org/10.3892/mmr.2017.6375 (2017).
Srougi, M., Gittes, R. F. & Underwood, R. H. Influence of exogenous glucocorticoids and ACTH on experimental adrenal autografts. Investigative urology 17, 265–268 (1980).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408, https://doi.org/10.1006/meth.2001.1262 (2001).
Pfaffl, M. W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 29, e45, https://doi.org/10.1093/nar/29.9.e45 (2001).
Acknowledgements
The present study was supported by the Japan Society for the Promotion of Science KAKENHI fund (Grants No. 15K08224 and 16K08533 to ST; Grant No. 16K10483 to TY), a research grant from the Takeda Science Foundation to ST, Yamaguchi Endocrine Research Foundation and Kansai Medical University grants to NT and a MEXT-Supported Program for the Strategic Research Foundation at Private Universities (Grants No. S1101034 and S1201038) to HY. The authors thank Prof. Kiyoshi Kurokawa (Osaka International University) and Drs. Souichi Oe, Taro Koike and Yukie Hirahara (Kansai Medical University) for their helpful comments. We also thank the Central Research Center and the Kansai Medical University research consortium for their support. Finally, we thank Ms. Ayako Nagata, Dr. Souichi Oe and Dr. Taro Koike for their technical assistance.
Author information
Authors and Affiliations
Contributions
Study concept and design: N.T., S.T. and H.Y.; data acquisition: T.Y., N.T. and S.T.; data processing: T.Y., N.T. and S.T.; data analysis and interpretation: T.Y. and S.T.; manuscript drafting: T.Y. and S.T.; critical manuscript revision for important intellectual content: T.Y., N.T., T.M., S.T., H.Y. and M.K.; statistical analysis: T.Y. and S.T.; materials: T.Y., N.T., T.M., S.T. and H.Y.; study supervision: S.T. and H.Y.; All authors approved the final draft of this manuscript for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yoshida, T., Takizawa, N., Matsuda, T. et al. GATA4/6 regulate DHH transcription in rat adrenocortical autografts. Sci Rep 10, 446 (2020). https://doi.org/10.1038/s41598-019-57351-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-57351-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.