Abstract
Endocrine disrupting compounds can interfere with androgen receptor (AR) signaling and disrupt steroidogenesis leading to reproductive failure. The brominated flame-retardant (BFR) 1, 2-dibromo-4-(1, 2-dibromoethyl) cyclohexane (TBECH), is an agonist to human, chicken and zebrafish AR. Recently another group of alternative BFRs, allyl 2, 4, 6-tribromophenyl ether (ATE), and 2, 3-dibromopropyl 2, 4, 6-tribromophenyl ether (DPTE) along with its metabolite 2-bromoallyl 2, 4, 6-tribromophenyl ether (BATE) were identified as potent human AR antagonists. These alternative BFRs are present in the environment. The aim of the present study was to determine the effect of mixed exposures to the AR agonist and the AR antagonists at environmentally relevant concentrations. In vitro reporter luciferase assay showed that the AR antagonists, when present at concentration higher than TBECH, were able to inhibit TBECH-mediated AR activity. These AR antagonists also promoted AR nuclear translocation. In vitro gene expression analysis in the non-tumorigenic human prostate epithelial cell RWPE1 showed that TBECH induced AR target genes whereas DPTE repressed these genes. Further analysis of steroidogenic genes showed that TBECH up-regulated most of the genes while DPTE down-regulated the same genes. The results indicate that when TBECH and DPTE are present together they will antagonize each other, thereby reducing their individual effects.
Similar content being viewed by others
Introduction
Endocrine-disrupting chemicals (EDCs) are either manmade or natural substances present in the environment as well as in products such as food, furniture, flame-retardants and other consumer products either as impurities or as constituents. According to the U.S. Environmental Protection Agency1, an endocrine-disrupting compound may be defined as “an exogenous agent that interferes with the synthesis, secretion, transport, binding, action, or elimination of natural hormones in the body that are responsible for the maintenance of homeostasis, reproduction, development, and/or behavior”. Recently, according to the Endocrine Society, EDCs may be defined as “an exogenous chemical or mixture of chemicals that interferes with any aspect of hormone action”2. Even though EDCs affect a wide range of endocrine functions in different tissues and organs, they were originally thought to mediate their actions entirely through the nuclear hormone receptors such as estrogen receptors, androgen receptors (ARs), progesterone receptors, thyroid receptors and retinoid receptors3. However, recent studies have shown that apart from alterations of nuclear receptor functions, EDCs also exert their effects via non-steroid receptors, transcriptional activators and different enzymatic pathways that are involved in steroid biosynthesis/metabolism, as well as through epigenetic mechanisms3,4. Some of these environmental chemicals have been associated with poor semen quality, testicular dysgenesis syndrome as well as an increased risk of testicular and prostate cancer5,6,7.
The endogenous androgens play a vital role in the normal development and function of the male reproductive system, and mediate their actions via AR signaling8,9. Although EDCs affect androgen-dependent signaling pathways through numerous mechanisms, modulation of AR function is a major mechanism. The increasing number of cases with masculinization of wildlife populations10,11 as well as the increasing incidences of prostate cancer development and progression6 indicate an increased presence of androgenic substances in the environment.
Several studies across different taxa have shown that 1,2-dibromo-4-(1,2-dibromoethyl)cyclohexane (TBECH) negatively impact endocrine and reproductive systems12,13,14,15,16 and that TBECH activates the chicken, human and zebrafish ARs12,13,14,16,17. TBECH has furthermore been shown to up-regulate the prostate specific antigen (PSA), which is a biomarker for prostate cancer13,14.
Although the presence of the two structurally similar BFRs allyl 2,4,6-tribromophenyl ether (ATE) and 2,3-dibromopropyl 2,4,6-tribromophenyl ether (DPTE), and the metabolite 2-bromoallyl 2,4,6-tribromophenyl ether (BATE) has been detected in animals, so far few studies have investigated the biological effects of these compounds. Recently, using molecular modeling and in vitro assays we showed that ATE and DPTE along with its metabolite BATE were able to bind and inhibit the activity of the human AR thereby acting as AR-antagonists18.
Previously it has been shown that anti-androgens such as flutamide, linuron, procymidone, p,p’-DDE, vinclozolin and prochloraz result in disturbed penile developments and disrupt sexual function and spermatogenesis in mammals5,19,20,21. Other BFRs acting as AR antagonists, including polybrominated diphenyl ethers (PBDEs) and their metabolites and hexabromocyclododecane (HBCDD)22 have been shown to alter the expression of genes involved in steroidogenesis23. It has also been shown that exposure of rats, from postnatal days 23 to 53, to DE-71 result in a significant delay in puberty and a decrease in seminal vesicle and ventral prostate weight24. Another study on adult rats reported a decrease in the size of the lateral and ventral prostate, seminal vesicles and bulbourethral gland25. Recently, in a study on chicken hepatocyte it was reported that exposure to ATE resulted in down-regulation of vitellogenin 2 (VTG2) and aromatase (CYP19A1) expression, suggesting that it may reduce the estrogen levels in chicken26.
ATE, BATE and DPTE have been detected in indoor dust samples from Vancouver, Canada with ATE being detected in 81% of the samples at levels up to 52 ng/g while DPTE was identified in 43% of the samples at concentrations up to 1200 µg/Kg27. The highest concentration of DPTE recorded was 1940 µg/Kg in sewer systems28 that corresponds to 3.65 µM. In a study of emerging flame-retardants in indoor environments in Norway the presence of TBECH was detected with indoor air concentrations of 77.9 pg/m3 in households, and 46.6 pg/m3 in schools29. Both TBECH and DPTE has been detected in Arctic mammals. TBECH was found at concentrations of 1.1 to 9.3 µg/kg (0.003–0.022 µM) in Arctic Beluga blubber30 while DPTE has been detected in seal blubber at much higher concentrations, up to 470 µg/kg (0.9 µM)31. TBECH was also the most common BFR detected in indoor and outdoor air samples in a study performed in Stockholm, Sweden32. DPTE was detected in 5% of the samples at 210 ng/g in household dust from Belgium33. It has been reported that DPTE is present in 95% of biota samples from Greenland34. Interestingly, studies on environmental samples such as sediment, soil and sludge as well as house dust and fire station dust samples have reported the concurrent presence of TBECH along with ATE, BATE and DPTE35,36,37. Currently, there is no information on the combined effect of these two different groups of BFRs, one exhibiting AR agonistic properties and the other with AR antagonistic properties12,13,14,16,17,18. The detection of these BFRs in household dust samples suggests chronic mixed exposures of people to these compounds.
The present study was aimed at analyzing the effects of mixed exposure to these BFRs. We determined if the AR antagonists ATE, BATE and DPTE could inhibit the AR activating potency of TBECH and if this would result in altered AR nuclear translocation. Gene expression analysis was performed to determine the effects of mixed exposure to TBECH and DPTE on the transcript levels of genes involved in steroidogenesis in the non-tumorigenic human prostate epithelial (RWPE1) cell lines.
Results
ATE, BATE and DPTE reduce TBECH-mediated luciferase activity
To determine whether ATE, and DPTE along with its metabolite BATE would reduce the AR activation by TBECH, transiently transfected HeLa cells were exposed to 100 nM TBECH-γδ (1:1) alone or in combination with different concentrations of ATE, BATE and DPTE. HeLa cells were chosen for these experiments as we have earlier observed that their slow metabolism (in contrast to HepG2 cells) of the endogenous steroids testosterone and DHT allows for co-exposure studies of TBECH, ATE, BATE and DPTE13,14. Exposure to ATE, BATE, and DPTE alone did not result in increased luciferase activity (Fig. 1A) confirming our earlier study showing that they are not AR agonists18. A significant decrease in TBECH-mediated luciferase activity was observed when cells were co-exposed to TBECH-γδ and ATE, BATE or DPTE. A 50% inhibition was observed with a 10,000-fold excess of the three BFRs (Fig. 1B). This demonstrates that all three BFRs are equivalent in potency at inhibiting TBECH-mediated luciferase activity.
ATE, BATE and DPTE inhibit TBECH-γδ mediated luciferase activity. HeLa cells transfected with human androgen receptor expression plasmid were exposed to ATE, BATE, DPTE or TBECH in 0.1% DMSO (control) (A), 100 nM TBECH-γδ (1:1 mixture) alone or in combination with ATE, BATE and DPTE (B) and 10 nM each of DHT or TBECH-γδ (1:1 mixture) alone or in combination with DPTE for 24 hr (C). All values were normalized against the controls and the maximum response of TBECH was set to 100%. n = 4 per exposure group.
To further determine the potency of DPTE at inhibiting DHT and TBECH-mediated AR activity, cells were exposed to 10 nM each of DHT and TBECH-γδ alone or in combination with different concentrations of DPTE. The results showed that DPTE was equally potent at inhibiting both DHT and TBECH-γδ mediated AR activity with 50% inhibition at 10.0 μM concentration (1000-fold excess) of DPTE (Fig. 1C).
ATE, BATE, DPTE induces AR nuclear translocation
ATE, BATE and DPTE inhibit DHT-mediated luciferase activity as well as TBECH-mediated luciferase activity in a dose-dependent manner. To further gain insight into the effect of ATE, BATE, and DPTE on the dynamics of AR regulation we studied the nuclear translocation of AR in HeLa cells transiently transfected with pEGFP-hAR. Initially, cells were exposed to 100 nM TBECH-γδ, and 10 μM each of the AR antagonists ATE, BATE, DPTE, bicalutamide and enzalutamide for 2 hr and sub-cellular localization was determined. Next, cells that were treated with the AR antagonists were co-exposed to 100 nM TBECH-γδ for an additional 3 hr and the sub-cellular localization was recorded. Most transfected cells exhibited exclusively cytoplasmic distribution of AR fused GFP protein in the absence of exposure (Fig. 2A). In cells treated with 10 nM DHT or 100 nM TBECH-γδ, the amount of AR fusion protein was almost exclusively located within the nucleus (Fig. 2B and H). Interestingly, exposure of cells to 10.0 μM each of the AR antagonists ATE, BATE, DPTE and bicalutamide alone resulted in distribution of AR to both the cytosol and the nucleus (Fig. 2C,D,E and F). However, following co-exposure of ATE, BATE, DPTE and bicalutamide (100-fold excess) with TBECH-γδ AR was exclusively localized within the nucleus (Fig. 2I,J,K and L). Exposure of cells to the second-generation AR antagonist enzalutamide alone resulted in predominantly cytoplasmic distribution of AR (Fig. 2G) that persisted in the presence of TBECH-γδ (Fig. 2M).
Subcellular localization of human AR following exposure to TBECH-γδ and different AR antagonists. HeLa cells transfected with plasmids pEGFP-hAR were exposed to 0.1% DMSO (control), 10 nM DHT, 100 nM of TBECH-γδ, and 10.0 µM each of ATE, BATE, DPTE, bicalutamide (Bic) and enzalutamide (Enz) alone for 2 hr. In addition, 10.0 µM each of ATE, BATE, DPTE, bicalutamide (Bic) and enzalutamide (Enz) was used in combination with 100 nM TBECH-γδ for another 3 hr. Cells were visualized by scanning confocal laser microscopy (60x magnification). Scale bar represents 20 µm.
TBECH-γδ and DPTE alter the transcription of androgen response genes in prostate epithelial cells
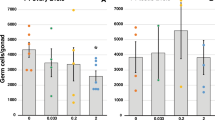
To analyze the effects of the BFRs on the expression of the AR target genes, RWPE1 cells were exposed to 0.1 and 1.0 μM of TBECH-γδ and DPTE, 20 nM DHT and 1.0 μM of hydroxyflutamide for 24 hr. The expression of the androgen response genes L-plastin (LCP1), microseminoprotein-β (MSMB) and prostate specific antigen (PSA) was up regulated by DHT and 1.0 μM of TBECH-γδ, while it was down regulated by 1.0 μM of DPTE and hydroxyflutamide (Fig. 3). These results were confirmed by analysis of PSA protein levels following exposure of RWPE1 cells to 100 nM DHT and TBECH that also resulted in significant increase of PSA protein levels (Fig. 4), a characteristic expected of androgenic compounds. Due to the low expression of PSA in the RWPE1 cells we used higher concentration of the compounds to follow the effects of co-exposure on PSA protein levels. Co-exposure of 100 nM DHT and TBECH with 20 µM of either DPTE or hydroxyflutamide resulted in significant decrease of PSA protein (Fig. 4).
TBECH-γδ and DPTE alter the expression of androgen response genes. RWPE1 cells were exposed to 0.1% DMSO (control), 20 nM DHT, 1.0 µM of hydroxyflutamide (HO-Flu), and 0.1 and 1.0 µM each of TBECH-γδ (γδ) and DPTE for 24 hr. One-way ANOVA followed by Dunnett’s post-test for multiple group comparison was performed to determine statistical significance (*p ≤ 0.05; **p ≤ 0.01; ***p ≤ 0.001). Error bars represent Mean ± SD, n = 4.
TBECH-γδ and DPTE alter the expression of prostate specific antigen protein. RWPE1 cells were exposed to 0.1% DMSO (control), 100 nM of DHT, TBECH-γδ and 20 µM of either hydroxyflutamide (HO-Flu) or DPTE alone for 6 days. For co-exposure experiments cells were exposed to 20 µM hydroxyflutamide and 20 µM DPTE together with 100 nM DHT or 100 nM TBECH-γδ for 6 days. PSA proteins were detected using PSA-specific antibody. β-actin was used as a loading control. The western blots results are represented by cropped gels and the complete western blots are shown in the supplementary material. One-way ANOVA followed by Dunnett’s post-test was used to determine statistical significance from (a) the control group, (b) DHT and (c) TBECH-γδ exposed cells (*p ≤ 0.05; **p ≤ 0.01; ***p ≤ 0.001). Mean ± SD of 3 independent experiments.
TBECH-γδ and DPTE alter the transcription of steroidogenesis genes in prostate epithelial cells
The effects of TBECH-γδ and DPTE on steroidogenic genes were analyzed. The expression of the steroidogenic factor 1 (SF-1) was down regulated following exposure to 20 nM DHT, 1.0 μM of DPTE and 1.0 μM hydroxyflutamide while it was not affected by TBECH-γδ exposure (Fig. 5A). The expression of the steroidogenic acute regulatory protein (StAR) was up regulated by 0.1 and 1.0 μM TBECH and 1.0 μM DPTE, but not altered following treatment with DHT and hydroxyflutamide (Fig. 5B). The expression of cytochrome P450 cholesterol side chain cleavage enzyme (P450scc, CYP11A1), which is involved in conversion of cholesterol to pregnenolone, was not altered by 20 nM DHT but significantly up-regulated by 1.0 μM of TBECH-γδ and down-regulated by 1.0 μM DPTE and hydroxyflutamide (Fig. 5C). Cytochrome P450 17α-hydroxylase/17, 20-lyase (CYP17A1) which is involved in the reactions converting pregnenolone to 17α-hydroxy-pregnenolone and then to dehydroepiandrosterone (DHEA), was not altered by DHT but up-regulated by 0.1 μM of TBECH-γδ, DPTE and hydroxyflutamide (Fig. 5D). In contrast, exposure to 1.0 μM TBECH-γδ resulted in a down regulation of CYP17A1 indicating a dual effect on steroidogenesis. 3β-hydroxysteroid dehydrogenase/Δ(5)-Δ(4)-isomerase type 2 (HSD3β2), which is involved in multiple conversion reactions (pregnenolone to progesterone, 17α-hydroxy-pregnenolone to 17α-hydroxy-pregesterone, DHEA to androstenedione, androstenediol to testosterone) and 17β-hydroxysteroid dehydrogenase type 3 (HSD17β3) were up regulated by DHT but not by the BFRs or hydroxyflutamide (Fig. 5E,F). The expression of Steroid 5 Alpha- Reductase 1 (SRD5A1) was up regulated by DHT and 1.0 μM of TBECH but down regulated by DPTE and hydroxyflutamide (Fig. 5G).
TBECH-γδ and DPTE alter expression of steroidogenesis genes. RWPE1 cells were exposed to 0.1% DMSO (control), 20 nM DHT, 1.0 µM of hydroxyflutamide (HO-Flu), 0.1 and 1.0 μM each of TBECH-γδ (γδ) and DPTE for 24 hr. One-way ANOVA followed by Dunnett’s post-test for multiple group comparison was performed to determine statistical significance (*p ≤ 0.05; **p ≤ 0.01; ***p ≤ 0.001). Error bars represent Mean ± SD, n = 4.
Discussion
Previously we have shown that the TBECH-γδ diastereomers are potent AR agonist for human13,14,17, chicken12 and zebrafish16 hence this mixture was selected for activation of AR-mediated luciferase activity in the present study. DPTE has been shown to be the most potent of the three BFRs at inhibiting DHT-induced PSA expression in LNCaP cells18. Thus, the presence of mixtures of these flame-retardants in the environment may result in a reduced response as they antagonize each other’s effects on AR mediated signaling in a dose dependent manner.
While being an AR antagonist, bicalutamide has been shown to exhibit AR agonistic properties at promoting AR nuclear translocation. However, it acts as an antagonist due to inefficient recruitment of co-activators resulting in an inactive transcription complex and inhibition of AR target genes38. Hydroxyflutamide also promote nuclear localization but retain its antagonistic properties by inhibiting AR dimerization and DNA binding39. The second-generation AR antagonist enzalutamide, which has been proposed to function as a pure AR antagonist inhibit AR nuclear translocation40. The present results suggest that ATE, BATE and DPTE, like bicalutamide or hydroxyflutamide, fail to block nuclear translocation of AR and therefore are weaker antagonists than enzalutamide.
To further analyze the effects of the studied BFRs we performed qRT-PCR analysis of genes regulated through AR. The LCP1, MSMB and PSA transcripts were analyzed. While DHT and TBECH-γδ up regulated all three genes, they were down regulated by the AR antagonists (ATE, BATE and DPTE). Human LCP1 is a leukocyte-specific actin-binding protein, was initially identified in transformed human fibroblasts, and later in normal hemopoietic cells and majority of human carcinoma cell lines41. LCP1 expression is regulated by DHT in human prostatic carcinoma LNCaP cells42, and by testosterone in breast and prostate cancer cells43. MSMB is secreted by the epithelial cells of the prostate, is considered a prostate cancer marker44, and is up regulated by DHT and down regulated by DPTE in ductal breast cancer T47D cells18. The presence of PSA transcript in RWPE1 cells has been previously reported45 and its induction by DHT observed in this study was comparatively lower than that observed in LNCaP cells18. In a previous study on LNCaP cells we have also shown that DPTE inhibits the expression of PSA transcripts18. Over-expression of PSA protein is used as a biomarker in prostate cancer diagnosis and is also considered an important biomarker for androgenic activity46. This suggest that the AR agonist TBECH-γδ like the natural ligand DHT could play a crucial role in normal prostate growth, while on the other hand DPTE would counteract this effect.
Regulation of AR requires the synthesis of endogenous androgens from cholesterol. Analysis of steroidogenic gene regulation demonstrated that the BFRs altered the transcription of all genes involved in the conversion of cholesterol to DHT. DHT, DPTE and hydroxyflutamide down regulated SF1. SF1 play a crucial role in regulating reproductive and endocrine functions including expression of steroidogenesis-related genes and is considered as a master regulator of steroid hormone production47. In contrast to DHT and hydroxyflutamide, TBECH-γδ and DPTE up regulated the StAR gene48. The transfer of cholesterol from the outer to the inner mitochondrial membrane is the first and rate-limiting step in steroidogenesis and is followed by conversion to pregnenolone by CYP11A149. HSD3β2 and HSD17β3 that are involved in multiple conversion reactions were only affected by DHT.
SRD5A1 is involved in conversion of androstenedione to 5α-androstenedione and testosterone to DHT50. While SRD5A1 was up regulated by DHT and TBECH-γδ, DPTE and hydroxyflutamide down regulated SRD5A1. It has been previously shown that SRD5A1 is an androgen response gene and that its expression is up regulated by androgen treatment in prostate cancer cells51. In T47D cells, DHT induced the expression of SRD5A1 while the AR antagonist DPTE repressed its expression18. The expression of cytochrome P450, family 19, subfamily A, polypeptide 1 (CYP19A1), involved in the conversion of testosterone to 17β-estradiol could not be analyzed due to low transcript levels (data not shown).
Previous studies have shown that exposure of animals to androgenic compounds result in masculinization and disruption of reproductive systems10,52 and this has also been suggested to increase the incidences of prostate cancer6. Studies with anti-androgens such as flutamide, linuron, procymidone, p,p’-DDE, vinclozolin and prochloraz have shown that exposure of mammals to these compounds result in induction of penile malformations as well as disruption of sexual function and spermatogenesis5,19,20. Prenatal exposure of female rats to testosterone has been shown to lead to permanently altered phenotype resulting in masculinization and presence of prostate tissue, usually not found in female rats53. In the present study, exposure of cells to the androgen DHT and AR agonist TBECH resulted in altered expression of genes involved in steroidogenesis. While TBECH-γδ up regulated StAR, CYP11A1 and SRD5A1, it down regulated CYP17A1. In a recent study on porcine ovarian follicles, it was observed that testosterone up regulated the expression of HSD3β2 while down-regulating CYP17A1 expression54 indicating that there may be cell type specific and species-specific differences in the regulation of steroidogenic gene transcription.
In the present study, we show that exposure to TBECH-γδ and DPTE alter the expression of steroidogenic genes. TBECH-γδ induced the expression of genes involved in steroidogenesis while DPTE repressed these genes. This shows that environmental co-exposures of TBECH-γδ and DPTE may result in opposing effects of the two compounds masking the exposure to these compounds. There is clearly a need for further studies as these compounds may affect other systems and not necessarily have opposing functions in all systems.
Materials and Methods
Chemicals
TBECH, ATE, BATE and DPTE were synthesized at 98% purity (Wellington Laboratories Inc., Canada) while bicalutamide, hydroxyflutamide (Sigma Aldrich, USA) and enzalutamide (Sellekchem, USA) were purchased. The studied compounds are described in Fig. 6 and Table 1. All the synthesized compounds, including bicalutamide and enzalutamide, were dissolved in dimethyl sulfoxide (DMSO). The amount of DMSO for exposure studies was maintained at a maximum of 0.1%.
Molecular structures of the studied compounds. (A) TBECH-γ, (B) TBECH-δ, (C) ATE, (D) BATE, (E) DPTE-R, (F) DPTE-S, (G) Bicalutamide, (H) Enzalutamide, (I) Hydroxyflutamide. The full names and functions of the compounds are given in Table 1.
Cell lines maintenance
Human cervical carcinoma (HeLa) and the non-tumorigenic human prostate epithelial cell (RWPE1) cell lines were obtained from ATCC. HeLa cells were grown and maintained in DMEM medium (Hyclone, UK) containing 4 mM L-glutamine and supplemented with 10% FBS (Hyclone, UK). RWPE1 cells were cultured in a keratinocyte serum free medium (K-SFM, Invitrogen, USA), supplemented with bovine pituitary extract (5 mg/ml) (BPE, Invitrogen, USA) and human recombinant epidermal growth factor (5 ng/ml) (EGF, Invitrogen, USA), penicillin (100 U/ml) and streptomycin (100 μg/ml) (Invitrogen, USA). The cell lines were incubated in a stable environment of 95% humidity, 5% CO2, and 37 °C.
Prior to seeding, HeLa cells were grown for 48 hr in MEM phenol-free medium (Invitrogen, USA) complemented with 10% charcoal-stripped FBS (PAA Laboratories, Australia) as previously described18.
Plasmids construction
The AR open reading frame (ORF) was PCR amplified using plasmid pCMVhAR17 as a template with primers containing Xho I and Bam HI sites (Table S1) and cloned into pZeoSV2 (Invitrogen, USA), a stable expression vector to generate the plasmid pZSVhAR. Using pEGFP-1 promoter reporter vector (Clontech, USA) as a template the EGFP ORF was PCR amplified with primers containing Nhe I and Bam HI sites (Table S1) then cloned into pZSVhAR and the resulting plasmid assigned as pEGFP-hAR. The plasmid constructs were confirmed by DNA sequencing (Eurofins, Germany).
Transient transfection and reporter assays
Prior to transfection, HeLa cells were seeded (8 × 104 cells/well) into 24 well plates (BD Falcon, USA) and grown until 90–95% confluence. Transient transfection was performed using Lipofectamine 2000 (Invitrogen, USA) according to the manufacturer’s instructions, using 270 ng of the human androgen receptor expression vector (pZSVhAR), 270 ng of the slp-ARE-Luc (sex-limiting protein–androgen response element–luciferase) reporter, and 60 ng Renilla luciferase (pRL; Promega, USA). At 24 h post-transfection, the media was replaced with phenol-free media complemented with charcoal-stripped FBS containing different concentrations of TBECH diastereomers TBECH-γδ, whereas for co-exposure experiments cells were exposed to ATE, BATE and DPTE (1.0–100 μM range) alone or in combination with 0.01 μM of DHT, or 0.01 and 0.1 μM of TBECH-γδ. After 24 hr of exposure, cells were then washed with PBS buffer (pH 7.4), lysed with passive lysis buffer and luciferase activity was measured using the Dual Luciferase Assay Kit (Promega, USA) in a TD 20/20 luminometer (Turner Designs, Sunnyvale, USA). The luciferase values thus obtained were normalized to the corresponding Renilla luciferase value.
Gene expression analysis
RWPE1 cells were seeded (2 × 105 cells/well) into 12-well plates (BD Falcon, USA). Cells were treated with 20 nM DHT, 0.1 and 1.0 µM of TBECH-γδ and DPTE, and 1.0 µM hydroxyflutamide for 24 hr. Total RNA isolation and qRT-PCR analysis were carried out as described previously18. The Ct values were normalized against elongation factor 1a (EF1A) and the data was presented as relative gene-expression as determined by the ΔΔCt method55. The primers used are listed in Table S1.
Confocal laser microscopy
HeLa cells were seeded (6 × 104 cells) in 24 well plates containing cover slips. Transient transfection was performed using Lipofectamine 2000 (Invitrogen, USA) as described above with 0.6 μg/well of pEGFP-hAR plasmid. At 24 hr post-transfection, the medium was replaced with fresh charcoal-stripped medium containing 10 nM DHT, 100 nM TBECH–γδ and 10 μM each of the AR antagonists ATE, BATE, DPTE, bicalutamide and enzalutamide. After 2 hr of exposure, cells that were pre-treated with AR antagonists were co-exposed with 100 nM of TBECH–γδ for an additional 3 hr. Following exposure, the coverslips were taken out and washed in PBS and mounted on microscope slides using Prolong Diamond Antifade Mountant containing DAPI (Invitrogen, USA). Cells were mounted on slides immediately after 2 hr of initial exposure and then at intervals of 60 min after addition of TBECH–γδ. Cells were viewed under FluoViewTM FV1000 Confocal Laser Scanning Biological Microscope (Olympus, Germany) and images were captured using 60x objective with oil immersion lens. The FV10-ASW viewer 2.0 software (Olympus, Germany) was used for processing the captured images.
Immunoblotting
RWPE1 cells were seeded (4 × 105 cells/well) into 6-well plates (BD Falcon, USA) and exposed to 100 nM of DHT and TBECH-γδ alone or in combination with 20.0 µM of either hydroxyflutamide or DPTE for 6 days. Cells were lysed in RIPA buffer and protein was extracted. Following quantification, the proteins were transferred onto PVDF membranes (Amersham Biosciences, UK) as described previously18. The membranes were rinsed for 30 min with Tris-buffered saline-Tween (0.1%) and incubated overnight at 4 °C with mouse anti-PSA antibody (Sigma, USA) at 1:500 dilutions. The secondary antibody, HRP-conjugated anti-mouse IgG (Amersham Biosciences, UK) was incubated for 1 hr at 1:5000 dilution at RT. Detection was performed using the enhanced chemiluminescent method (Amersham Biosciences, UK). The membrane was then stripped and probed for β-actin using mouse anti β-actin antibody (Sigma, USA). The bands were analyzed and quantified using ImageJ software (National Institute of Health, USA) and normalized with their respective β-actin level.
Statistical analysis
Statistical analysis was performed using GraphPad Prism 5 software (GraphPad Software, USA) using one-way analysis of variance (ANOVA) followed by Dunnett’s post-test for multiple group comparison. Statistically significant differences were considered if the p values were <0.05 (*p < 0.05, **p < 0.01 and ***p < 0.001).
References
Kavlock, R. J. et al. Research needs for the risk assessment of health and environmental effects of endocrine disruptors: A report of the U.S. EPA-sponsored workshop. Environ. Health Perspect. 104(4), 715–740 (1996).
Zoeller, R. T. et al. Endocrine-disrupting chemicals and public health protection: A statement of principles from the endocrine society. Endocrinol. 153, 4097–4110 (2012).
Diamanti-Kandarakis, E. et al. Endocrine-disrupting chemicals: An endocrine society scientific statement. Endocr. Rev. 30, 293–342 (2009).
Casati, L., Sendra, R., Poletti, A., Negri-Cesi, P. & Celotti, F. Androgen receptor activation by polychlorinated biphenyls: Epigenetic effects mediated by the histone demethylase Jarid1b. Epigenetics 8, 1061–1068 (2013).
Fisher, J. S. Environmental anti-androgens and male reproductive health: Focus on phthalates and testicular dysgenesis syndrome. Reproduction 127, 305–315 (2004).
Hess-Wilson, J. K. & Knudsen, K. E. Endocrine disrupting compounds and prostate cancer. Cancer Lett. 241, 1–12 (2006).
Hu, W. Y., Shi, G. B., Hu, D. P., Nelles, J. L. & Prins, G. S. Actions of estrogens and endocrine disrupting chemicals on human prostate stem/progenitor cells and prostate cancer risk. Mol. Cell. Endocrinol. 354, 63–73 (2012).
Green, S. M., Mostaghel, E. A. & Nelson, P. S. Androgen action and metabolism in prostate cancer. Mol. Cell. Endocrinol. 360, 3–13 (2012).
Ralph, J. L., Orgebin-Crist, M. C., Lareyre, J. J. & Nelson, C. C. Disruption of androgen regulation in the prostate by the environmental contaminant hexachlorobenzene. Environ. Health Perspect. 111, 461–466 (2003).
Ellis, R. J. et al. In vivo and in vitro assessment of the androgenic potential of a pulp and paper mill effluent. Environ. Toxicol. Chem. 22, 1448–1456 (2003).
Sone, K. et al. Effects of an androgenic growth promoter 17beta-trenbolone on masculinization of mosquitofish (Gambusia affinis affinis). Gen. Comp. Endocrinol. 143, 151–160 (2005).
Asnake, S., Pradhan, A., Banjop-Kharlyngdoh, J., Modig, C. & Olsson, P.-E. 1,2-dibromo-4-(1,2 dibromoethyl) cyclohexane (TBECH)-mediated steroid hormone receptor activation and gene regulation in chicken LMH cells. Environ. Toxicol. Chem. 33, 891–899 (2014).
Khalaf, H. et al. Diastereomers of the brominated flame retardant 1,2-dibromo-4-(1,2 dibromoethyl)cyclohexane induce androgen receptor activation in the HepG2 hepatocellular carcinoma cell line and the LNCaP prostate cancer cell line. Environ. Health Perpect. 117, 1853–1859 (2009).
Kharlyngdoh, J. B., Asnake, S., Pradhan, A. & Olsson, P.-E. TBECH, 1,2-dibromo-4-(1,2 dibromoethyl) cyclohexane, alters androgen receptor regulation in response to mutations associated with prostate cancer. Toxicol. Appl. Pharmacol. 307, 91–101 (2016).
Park, B. J., Palace, V., Wautier, K., Gemmill, B. & Tomy, G. 2011. Thyroid axis disruption in juvenile brown trout (Salmo trutta) exposed to the flame retardant β- tetrabromoethylcyclohexane (β-TBECH) via the diet. Environ. Sci. Technol. 45, 7923–7927 (2011).
Pradhan, A., Kharlyngdoh, J. B., Asnake, S. & Olsson, P.-E. The brominated flame retardant TBECH activates the zebrafish (Danio rerio) androgen receptor, alters gene transcription and causes developmental disturbances. Aquat. Toxicol. 142–143, 63–72 (2013).
Larsson, A., Eriksson, L. A., Andersson, P. L., Ivarson, P. & Olsson, P.-E. Identification of the brominated flame retardant 1,2-dibromo-4-(1,2-dibromoethyl)cyclohexane as an androgen agonist. J. Med. Chem. 49, 7366–72 (2006).
Kharlyngdoh, J. B. et al. Identification of a group of brominated flame retardants as novel androgen receptor antagonists and potential neuronal and endocrine disrupters. Environ. Int. 74, 60–70 (2015).
Blystone, C. R. et al. Prochloraz inhibits testosterone production at dosages below those that affect androgen-dependent organ weights or the onset of puberty in the male Sprague Dawley rat. Toxicol. Sci. 97, 65–74 (2007).
Kubota, K. et al. Effects of vinclozolin administration on sperm production and testosterone biosynthetic pathway in adult male rat. J. Reprod. Dev. 49, 403–412 (2003).
Simon, L. et al. Exposure of neonatal rats to anti-androgens induces penile mal-developments and infertility comparable to those induced by oestrogens. Int. J. Androl. 35, 364–376 (2012).
Song, R. et al. Effects of fifteen PBDE metabolites, DE71, DE79 and TBPPA on steroidogenesis in the H295R cell line. Chemosphere 71, 1888–1894 (2008).
Fa, S. et al. Acute effects of hexabromocyclododecane on leydig cell cyclic nucleotide signaling and steroidogenesis in vitro. Toxicol. Lett. 218, 81–90 (2013).
Stoker, T. E. et al. Assessment of DE-71, a commercial polybrominated diphenyl ether (PBDE) mixture, in the EDSP male and female pubertal protocols. Toxicol. Sci. 78, 144–155 (2004).
Stoker, T. E. et al. In vivo and in vitro anti-androgenic effects of DE-71, a commercial polybrominated diphenyl ether (PBDE) mixture. Toxicology and applied pharmacology 207, 78–88 (2005).
Porter, E., Crump, D., Egloff, C., Chiu, S. & Kennedy, S. W. Use of an avian hepatocyte assay and the avian toxchip polymerse chain reaction array for testing prioritization of 16 organic flame retardants. Environ. Toxicol. Chem. 33, 573–582 (2014).
Shoeib, M., Harner, T., Webster, G. M., Sverko, E. & Cheng, Y. Legacy and current-use flame retardants in house dust from Vancouver, Canada. Environ. Pollut. 169, 175–182 (2012).
Sauer, J., Antusch, E. & Ripp, C. Monitoring of lipophilic organic pollutants in sewer systems by sewer slime analysis. Vom Wasser 88, 49–69 (1997).
Cequier, E. et al. Occurrence of a broad range of legacy and emerging flame retardants in indoor environments in Norway. Environ. Sci. Technol. 48, 6827–6835 (2014).
Tomy, G. T. et al. Identification of the N cycloaliphatic brominated flame retardant 1,2-dibromo-4-(1,2-dibromoethyl)cyclo-hexane in Canadian arctic beluga (Delphinapterus leucas). Environ. Sci. Technol. 42, 543–549 (2008).
von Recke, R. & Vetter, W. Synthesis and characterisation of 2,3.dibromopropyl-2.4.6-tribromophenyl ether (DPTE) and structurally related compounds evidenced in seal blubber and brain. Environ. Sci. Technol. 41, 1590–1595 (2007).
Newton, S., Sellstrom, U. & de Wit, C. A. Emerging flame retardants, PBDEs, and HBCDDs in indoor and outdoor media in Stockholm, Sweden. Environ. Sci. Technol. 49, 2912–2920 (2015).
Geens, T., Ali, N., Roosens, L., Neels, H. & Covaci, A. Analytical characteristics of several new brominated flame retardants. Talanta 81, 1865–1869 (2010).
Vorkamp, K. et al. Novel brominated flame retardants and dechlorane plus in Greenland air and biota. Environ. Pollut. 196C, 284–291 (2014).
Brown, F. R., Whitehead, T. P., Park, J. S., Metayer, C. & Petreas, M. X. Levels of non-polybrominated diphenyl ether brominated flame retardants in residential house dust samples and fire station dust samples in California. Environ. Res. 135, 9–14 (2014).
Kolic, T. M. et al. The analysis of halogenated flame retardants by GC-HRMS in environmental samples. J. Chromatog. Sci. 47, 83–91 (2009).
Wong, F., Kurt-Karakus, P. & Bidleman, T. F. Fate of brominated flame retardants and organochlorine pesticides in urban soil: Volatility and degradation. Environ. Sci. Technol. 46, 2668–2674 (2012).
Masiello, D., Cheng, S., Bubley, G. J., Lu, M. L. & Balk, S. P. Bicalutamide functions as an androgen receptor antagonist by assembly of a transcriptionally inactive receptor. J. Biol. Chem. 277, 26321–26326 (2002).
Wong, C. I., Zhou, Z. X., Sar, M. & Wilson, E. M. Steroid requirement for androgen receptor dimerization and DNA binding. Modulation by intramolecular interactions between the NH2-terminal and steroid-binding domains. J. Biol. Chem. 268, 19004–19012 (1993).
Guerrero, J., Alfaro, I. E., Gomez, F., Protter, A. A. & Bernales, S. Enzalutamide, an androgen receptor signaling inhibitor, induces tumor regression in a mouse model of castration-resistant prostate cancer. Prostate 73, 1291–1305 (2013).
Goldstein, D., Djeu, J., Latter, G., Burbeck, S. & Leavitt, J. Abundant synthesis of the transformation-induced protein of neoplastic human fibroblasts, plastin, in normal lymphocytes. Cancer Res. 45, 5643–5647 (1985).
Zheng, J. et al. Steroid hormone induction and expression patterns of L-plastin in normal and carcinomatous prostate tissues. Am. J. Pathol. 150, 2009–2018 (1997).
Lin, C. S., Lau, A., Yeh, C. C., Chang, C. H. & Lue, T. F. Upregulation of L-plastin gene by testosterone in breast and prostate cancer cells: Identification of three cooperative androgen receptor-binding sequences. DNA Cell Biol. 19, 1–7 (2000).
Sakai, H. et al. Prognostic significance of beta-microseminoprotein mRNA expression in prostate cancer. Prostate 38, 278–284 (1999).
El-Atta, H. M. et al. DNA fragmentation, caspase 3 and prostate-specific antigen genes expression induced by arsenic, cadmium, and chromium on non-tumorigenic human prostate cells. Biol. Trace Elem. Res. 162, 95–105 (2014).
Hsieh, J. T., Wu, H. C., Gleave, M. E., von Eschenbach, A. C. & Chung, L. W. Autocrine regulation of prostate-specific antigen gene expression in a human prostatic cancer (LNCaP) subline. Cancer Res. 53, 2852–2857 (1993).
Morohashi, K. I. & Omura, T. Ad4BP/SF-1, a transcription factor essential for the transcription of steroidogenic cytochrome P450 genes and for the establishment of the reproductive function. FASEB J. 10, 1569–1577 (1996).
Hampl, R., Kubatova, J. & Starka, L. Steroids and endocrine disruptors-history, recent state of art and open questions. J. Steroid Biochem. Mol. Biol. 155, 217–23 (2014).
Manna, P. R. & Stocco, D. M. Regulation of the steroidogenic acute regulatory protein expression: Functional and physiological consequences. Curr. Drug Targets Immune Endocr. Metabol. Disord. 5, 93–108 (2005).
Sharifi, N. & Auchus, R. J. Steroid biosynthesis and prostate cancer. Steroids 77, 719–726 (2012).
Li, J. et al. Androgen regulation of 5alpha-reductase isoenzymes in prostate cancer: Implications for prostate cancer prevention. PLoS One 6, e28840 (2011).
Morthorst, J. E., Holbech, H. & Bjerregaard, P. Trenbolone causes irreversible masculinization of zebrafish at environmentally relevant concentrations. Aquat. Toxicol. 98, 336–343 (2010).
Hotchkiss, A. K. et al. Prenatal testosterone exposure permanently masculinizes anogenital distance, nipple development, and reproductive tract morphology in female Sprague-Dawley rats. Toxicol. Sci. 96, 335–345 (2007).
Duda, M. et al. The impact of antiandrogen 2-hydroxyflutamide on the expression of steroidogenic enzymes in cultured porcine ovarian follicles. Mol Biol Rep. 41, 4213–4222 (2014).
Schmittgen, T. D. & Livak, K. J. Analyzing real-time PCR data by the comparative Ct method. Nat. Protoc. 3, 1101–1108 (2008).
Acknowledgements
The research was financed by the Knowledge Foundation, Sweden and Örebro University (to PEO).
Author information
Authors and Affiliations
Contributions
J.B.K., A.P. and P.E.O. wrote the main manuscript text. J.B.K. and A.P. performed the experiments. J.B.K. prepared Figs 1 and 5 while J.B.K. and A.P. prepared Figs 2 and 3. PEO prepared Figs 4 and 6. All three authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kharlyngdoh, J.B., Pradhan, A. & Olsson, PE. Androgen receptor modulation following combination exposure to brominated flame-retardants. Sci Rep 8, 4843 (2018). https://doi.org/10.1038/s41598-018-23181-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-23181-0
This article is cited by
-
Research advances in identification procedures of endocrine disrupting chemicals
Environmental Science and Pollution Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.