Abstract
About 20% of angiosperms employ self-fertilization as their main mating strategy. In this study, we aimed to examine how the selfing timing correlated with floral traits in three Gentianopsis species in which autonomous selfing is achieved through filament elongation. Although the three Gentianopsis species exhibit no significant variation in their capacity for autonomous selfing, flowers of G. grandis last longer, are larger and have a higher corolla biomass, P/O ratios and male biomass allocation than those of G. paludosa, and especially those of G. contorta. Autonomous selfing occurs in the early floral life of G. paludosa and G. contorta and in the later floral life of G. grandis. Seed production mainly results from autonomous selfing in G. paludosa and G. contorta; however, G. grandis could be more described as having a mixed mating system. We suggest that autonomous selfing in later floral life increases the chance of cross-pollination prior to this, while autonomous selfing in early floral life offers a selective advantage to plants by reducing the resource investment in traits that may increase pollinator attraction and visitation.
Similar content being viewed by others
Introduction
Understanding how plants ensure reproductive success is important in plant evolutionary biology1. Plants have evolved various strategies to ensure successful reproduction in diverse environments2. Around 20% of flowering species employ self-fertilization as their main mating strategy3. Selfing plants have a 50% gene transmission advantage compared to outcrossing plants4,5,6, but their offspring may have low fitness because of inbreeding depression and the costs of gamete and seed discounting4,7,8,9. Selection for reproductive assurance, where selfing guarantees seed production when a lack of pollinators or inefficient pollen transfer limit reproductive success, is the most widely accepted hypothesis explaining the evolution of selfing10,11.
In autonomous selfing, the selfing time is a key trait which is generally under the nature selection12,13. There are three categories of autonomous selfing: prior, competing and delayed self-pollination. These occur, respectively, before, during and after opportunities for outcrossing13,14. If pollinators are sometimes unreliable, delayed selfing tends to be the favored type15,16,17. Delayed selfing does not interfere with outcrossing and functions as a “better than nothing” alternative even when it results in high inbreeding depression18. Under such conditions, cross-pollination can occur in early floral life, whilst the delayed selfing guarantees successful pollination at the end of the flower’s life if outcrossing fails13,19,20. If there are no pollinators, competing even prior selfing may offer a selective advantage over delayed selfing, by reducing the resources invested in traits associated with attracting pollinators and optimizing visits, for example floral longevity, floral size and nectar production21,22,23. There is variation in the timing of autonomous selfing even in closely related species12,24 or different populations of a single species25,26,27. This provides opportunities to evaluate the evolutionary significance of different types and timings of autonomous selfing.
Gentianopsis is a genus of flowering plants in the gentian family, commonly known as the fringed gentians; they have blue to purple flowers. Delayed selfing has been reported in two species of Gentianopsis (G. barbata and G. paludosa) on the Qinghai-Tibetan Plateau; in both cases this is achieved by elongation of the filaments as the flower ages28,29. Gentianopsis ciliata, which is found in central Europe, is self-compatible but hardly ever sets seed under natural conditions due to pollen limitation, indicating that this species has not evolved an autonomous selfing mechanism30,31. More studies are needed to understand the evolution of mating systems in this genus. Here, we compared variations in the timing of autonomous selfing in order to examine how various floral traits are adapted in three species of Gentianopsis (G. contorta, G. paludosa and G. grandis,) in NW Yunnan, China. We address the following questions: (1) Are all the three Gentianopsis species capable of autonomous selfing? If so, then (2) how are floral traits correlated with the timing of autonomous selfing?
Materials and Methods
Study species and populations
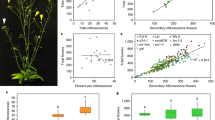
The genus Gentianopsis contains 24 species and is widely distributed across the North Temperate Zone. Five species are native to China and occur at altitudes from 700 m to 4900 m32. Three species of Gentianopsis found in Northwest Yunnan, China – G. contorta, G. grandis, and G. paludosa – were selected for our study. The timing of flowering differs between the three, with G. contorta and G. grandis flowering from the beginning to the end of September, and G. paludosa flowering from the end of August to the middle of September. All the three species are characterized by a long blue to purple corolla tube, four horizontally spreading lobes and four projecting nectaries, which are positioned at the base of the corolla lobe (Fig. 1).
This study was conducted between early August and early October in 2012 and 2013. The population of G. contorta was at the Shangri-La Alpine Botanical Garden, Yunnan, China (27.9050°N, 99.6364°E; altitude, 3300 m a.s.l.). Pollination experiments involving G. paludosa were carried out at Potatso National Park, Shangri-La Yunnan, China (27.8588°N, 99.9952°E; altitude, 3600 m a.s.l.). The Gentianopsis grandis studied was in Xiaozhongdian, Shangri-La, China (27.5672°N, 99.8060°E; altitude, 3250 m a.s.l.). All three sites are species-rich meadows. Floral traits and the autonomous self-pollination mechanism of the three Gentianopsis species were studied during their peak flowering seasons.
Floral traits
Thirty-six to forty-one plants of each species were randomly selected for a survey of the number of flowers per plant. We recorded eight floral traits: flower longevity, corolla tube length, corolla diameter, pollen production per flower, number of ovules per flower, pollen/ovule ratio, male investment/female investment and corolla biomass. In general, we selected apical flowers or floral buds from different individuals of each species for the measurements. To record flower longevity, one unopened bud of 34 to 41 individuals of each species was labeled. We observed these flowers twice a day, in the morning (ca. 08:00) and the evening (at 20:00). The length of the corolla tube and corolla diameter were measured on about twenty flowers per species using a hand-held digital caliper. We also quantified pollen grain and ovule numbers for the three species by selecting thirty flower buds, dissecting the anthers and ovaries and preserving them in 70% alcohol. Pollen production per flower was estimated following the method of Dafni33. The number of ovules in each ovary was counted under a dissecting microscope. For each flower, the pollen/ovule (P/O) ratio was calculated as the number of pollen grains divided by the number of ovules. We also dissected about 30 flower buds of each species, separating the pistil, stamens and corolla before determining their dry weights. All the floral parts were oven-dried for a minimum of 48 h at 80 °C and were weighed to the nearest 0.0001 g. We calculated the male investment/female investment (androecium biomass/gynoecium biomass), to represent the male sex allocation34,35. Floral traits were compared between species using one-way ANOVA with species as the fixed factor.
Flower development
To investigate any change in herkogamy during flower opening, 20 flower buds from different plants of each species were selected and labeled. At the flower bud stage, we removed one lobe of the corolla and each day we measured the heights of the stamens and pistil using a hand-held digital caliper; these measurements continued each day until the flowers withered. Duan et al.29 demonstrated that the removal of a flower lobe in this way does not affect the process of flower development. The heights of the stamens and the pistil were measured from the bottom of the ovary, and the distance from the stigma to the anthers was calculated by subtracting the height of the stamens from the height of the pistil.
The capacity for autonomous self-fertilization
To investigate each species’ capacity for autonomous selfing, we selected 31 to 33 individuals of each and enclosed their inflorescences within fine nylon nets. We performed three pollination treatments for each individual at peak flowering: (1) a flower that was not emasculated, which produced seed as a result of autonomous selfing; (2) a flower that was emasculated prior to flower-opening, but which was pollinated using pollen from other flowers on the same parent plant; and (3) a flower that was emasculated prior to flower-opening. The final treatment (3) was employed to test whether the three Gentianopsis species are able to produce any seeds through apomixis. Fruits were collected once seeds were ripe and seed set was evaluated as the number of seeds divided by the total number of ovules for each flower. Following Lloyd and Schoen14, the index of autonomous selfing [i.e. autofertility (AF)] was calculated as follows: [(mean autonomous seed set of unmanipulated flowers)/(mean seed set of supplemental pollinated flowers)]. Two-way ANOVA was performed to examine the effects of treatment and species on seed set.
The timing of autonomous self-fertilization
The timing of autonomous selfing was investigated by emasculating flowers at different points in the flowering period. According to the floral longevity of each species, the following treatments were applied to about 30 flowers per treatment: anther removal (1) at the day before the first day that the flower was open; (2) at the first day that the flower was open; (3) at the second day that the flower was open, until the end of the flowering day, and (4) a control treatment in which the anthers were left intact. All the fruits were collected before dehiscence and taken back to the laboratory for seed set to be evaluated.
Reproductive assurance
To assess the degree of reproductive assurance, we selected 27 to 41 individuals of each species. A flower from each individual was emasculated before anther dehiscence, and another flower was left intact and unmanipulated as a control. Both treatments were left for open pollination. For each fruit, seed set was determined in the laboratory as the number of seeds/the total number of ovules. Reproductive assurance was estimated as: 1 − (seed set of emasculated flower/seed set of intact flower)4,11. One-way ANOVA with species as a fixed factor was used to compare the difference in the magnitude of reproductive assurance.
Results
Floral traits
There was no statistically significant difference in the total number of flowers that the three Gentianopsis species produced per plant (F2,114 = 1.29, P = 0.28), although G. paludosa had the largest number per plant, whilst G. contorta had the fewest (see Table 1). The Gentianopsis flowers opened from 0900 h and closed at about 1700h every day due to the drop in temperature during the night; on rainy days the flowers closed temporarily and flower life was increased. Flowers lasted an average of 4.05 ± 0.11, 4.58 ± 0.33, 6.25 ± 0.28 days, respectively, for G. contorta, G. paludosa and G. grandis, and the floral longevity in the three species was significantly different (F2,107 = 4.42, P < 0.05). The Gentianopsis species differed significantly in flower size (corolla tube length: F2,59 = 394.30, P < 0.001; corolla diameter: F2,59 = 128.90, P < 0.001; corolla biomass: F2,92 = 18.73, P < 0.001), with G. grandis producing the largest and heaviest flowers, G. paludosa being intermediate, and G. contorta producing the smallest and lightest flowers. The three species also differed significantly in the number of ovules (F2,46 = 65.22, p < 0.001) and pollen grains (F2,46 = 37.88, P < 0.001) produced per flower. Gentianopsis contorta produced the largest number of ovules and an intermediate number of pollen grains, while G. grandis produced the largest number of pollen grains and an intermediate number of ovules. The P/O ratio varied significantly between the three species (F2,46 = 40.59, P < 0.001), with G. grandis having the largest ratio, G. paludosa being intermediate and G. contorta having the smallest. The male sex allocation also differed significantly between the three species (stamens biomass/pistil biomass: F2,92 = 31.74, P < 0.001), with G. grandis allocating more resources to male function (stamens), and G. contorta allocating more resources to female function (pistil).
Flower development
We found the degree of herkogamy (the distance between the stamens and pistil) changed during the flower’s development in the three species, mainly resulting from the elongation of filaments (Figs 1 and 2). At the floral bud stage, the stamens were shorter than the pistil, and then the stamens elongated as the flower developed. The height of the anthers gradually equaled or exceeded the height of the stigmas, resulting in stigma-anther contact. We observed flowers with stigma-anther contact on the first day of flower opening for G. contorta and G. paludosa, but it was about three days after anthesis commenced that the height of the anthers equaled the height of the stigmas in G. grandis.
The capacity for autonomous self-fertilization
None of the bagged flowers that had been emasculated before anther dehiscence produced any seeds, indicating that there was no apomixis in the three Gentianopsis species. There was no difference in seed set between autonomous selfed flowers and hand self-pollinated flowers (F2,190 = 0.06, p = 0.80), nor were the values affected by the treatment and species interaction (F2,190 = 0.37, p = 0.70); however, there was a significant difference between species (F2,190 = 23.99, p < 0.001). The three Gentianopsis showed no significant variation in their capacity for autonomous selfing (F2,95 = 1.17, P = 0.31).
Timing of autonomous selfing
In each of the three study species, the amount of seed set resulting from autonomous selfing increased significantly with flower age, but the rate of increase differed significantly between the species (Table 2). Gentianopsis paludosa and G. contorta achieved half of their total autonomous seed set by the second day of flowering, whereas the rate of increase in autonomous selfing peaked much later in G. grandis and on the fourth day all flowers suddenly set seed as control (Fig. 3).
Reproductive assurance
In all three species, the seed set of the control was significantly higher than when flowers were emasculated (G. contorta: t = 10.47, d. f. = 26, P < 0.001; G. paludosa: t = 10.47, d. f. = 40, P < 0.001; G. grandis: t = 16.08, d. f. = 37, P < 0.001), indicating that autonomous self-pollination delivers reproductive assurance in all three species (Fig. 4). The three species showed significant variation (F2,103 = 84.75, P < 0.001) in their capacity for reproductive assurance (RA = 0.91 ± 0.03, 0.73 ± 0.04 and 0.28 ± 0.04 for G. contorta, G. paludosa, and G. grandis respectively).
Discussion
The changes over time in the relative positions of stigmas and anthers within flowers of self-compatible plants are important in controlling the timing of autonomous selfing11,36. As with previous studies28,29, our research suggests that the three Gentianopsis species (G. contorta, G. paludosa and G. grandis) have the capacity to self autonomously mainly through filament elongation, but the timing of this differs between the species. At the floral bud stage, the stamens are shorter than the pistil in all three species. Stigma and anthers within flowers came into contact with each other on the first day of flower opening in G. contorta and G. paludosa mainly because of filament elongation, while the height of the anthers equaled the height of stigmas about three days after anthesis commenced in G. grandis (Fig. 2). The data from our emasculation experiments indicated that flowers are autonomously fertilized in the early floral lifespan in G. contorta and G. paludosa, suggesting that the timing of autonomous selfing in the two species can certainly be considered to indicate competing selfing, whilst in G. grandis the autonomous selfing occurs in the late floral stage and tends to represent delayed selfing (Fig. 3).
In our study, flowers of G. grandis lasted longer and were significantly larger with higher corolla biomass, P/O ratios and male allocation (stamens biomass/pistil biomass) than those of G. paludosa, and especially those of G. contorta (see Table 1). These results support the idea that the later selfing species retain the floral traits and pay the costs associated with attracting polliantors19,37. Indeed, we frequently observed bumblebee pollinators effectively visiting flowers of G. grandis, whilst only two and one bumblebee visits, respectively, were observed in G. paludosa and G. contorta during the experimental period. The emasculation experiments showed 71%, 27% and 9% of the ovules were fertilized by outcrossing pollen grains for G. grandis, G. paludosa, and G. contorta, respectively (Fig. 4). Although the three Gentianopsis species exhibited no significant variation in their capacity for autonomous selfing, seed production mainly resulted from autonomous selfing in the two competing selfing species, especially G. contorta. Gentianopsis grandis could be characterized as having more of a mixed mating system, producing offspring by autonomous selfing when pollinator-mediated pollination fails.
Duan et al.29 found that anthers made contact with the central stigma and self-pollination occurred at the end of the floral lifespan in G. paludosa on the Qinghai-Tibetan Plateau, which is typical of delayed selfing. However, we found that flowers of G. paludosa were autonomously fertilized in the early floral lifespan and that stigma-anther contact occurred once the flower had opened in plants from Northwest Yunnan. These findings suggest that even within the same species the timing of autonomous selfing may exhibit substantial variation, and such differences may in turn contribute variable amounts of autonomously selfed seeds to total seed set. Further studies are needed to explore how evolution history and ecological factors shape the autonomous selfing traits within Gentianopsis species.
In all the three Gentianopsis species, anther-stigma contact did not indicate that autonomous selfing would result in a large amount of seed production immediately. The anthers were already in contact with the stigma on the first flowering day for G. contorta and G. paludosa, while the amount of seed set resulting from autonomous selfing increased linearly with flower age. We observed stigma-anther contact on the third day of flower opening in G. grandis, but autonomous selfing mainly occurred on the fourth day (Figs 2 and 3). The fact that the timing of autonomous selfing was later than the timing of anther-stigma contact in the three species may result from the gradual exposure of pollen grains on anthers and low P/O ratios in the three Gentianopsis species. In general, pollen grains on anthers gradually become susceptible to removal, thereby preserving pollen grains on the anthers when there are few pollen vectors and/or restricting pollen removal by individual pollinators, so that many pollinators can be involved in pollen dispersal38,39. Furthermore, the P/O ratios of the three species are quite low (6.48 ± 0.61, 25.28 ± 2.03, 26.00 ± 2.27 respectively for G. contorta, G. paludosa and G. grandis). Although anthers dehisce once flowers open in the three species, only some of the pollen grains could touch and germinate on stigmas when anther-stigma contact occurs; thus, only a small proportion of ovules would be fertilized. All three Gentianopsis species still have some chance of outcrossing since the flowers set at least some seeds after emasculation (Fig. 4). It would be worth examining the mating system of the three Gentianopsis species using molecular makers to determine whether the interval between the anther-stigma contact and the actual occurrences of autonomous selfing allows the opportunity for stigmas to receive outcrossing pollen grains.
Conclusion
Our results clearly indicate that autonomous selfing in later floral life preserves the chance of cross-pollination in early floral life and that autonomous selfing in early floral life offers a selective advantage to plants by reducing their investment in traits that may increase pollinator attraction and visitation. Flowers are autonomously fertilized in the early floral lifespan in G. contorta and G. paludosa, but in G. grandis autonomous selfing occurs later. The three Gentianopsis species show no significant variation in their capacity for autonomous selfing, although flowers of G. grandis last longer and are larger with higher corolla biomass, P/O ratios and male biomass allocation than those of G. paludosa, and especially those of G. contorta. We also found that the timing of autonomous selfing was later than the timing of anther-stigma contact, probably resulting from the low P/O ratios and the gradual exposure of pollen grains on anthers in the three Gentianopsis species, although plants with obligate autogamous mating systems usually have low P/O ratios40,41.
References
Barrett, S. C. H. Mating strategies in flowering plants: the outcrossing–selfing paradigm and beyond. Philos. Trans. R. Soc. London, Ser. B358, 991–1004 (2003).
Barrett, S. C. H. Major Evolutionary Transitions in Flowering Plant Reproduction: An Overview. Int. J. Plant Sci 169, 1–5 (2008).
Barrett, S. C. H. The evolution of plant sexual diversity. Nat. Rev. Genet. 3, 274–284 (2002).
Lloyd, D. G. Self-and cross-fertilization in plants. II. The selection of self-fertilization. Int. J. Plant Sci. 153, 370–380 (1992).
Holsinger, K. Reproductive systems and evolution in vascular plants. Proc. Natl. Acad. Sci. USA 97, 7037–7042 (2000).
Eckert, C. G. & Herlihy, C. R. Using a cost–benefit approach to understand the evolution of self-fertilization in plants: the perplexing case of Aquilegia canadensis (Ranunculaceae). Plant Spec. Biol. 19, 159–173 (2004).
Goodwillie, C., Kalisz, S. & Eckert, C. G. The Evolutionary Enigma of Mixed Mating Systems in Plants: Occurrence, Theoretical Explanations, and Empirical Evidence. Annu. Rev. Ecol. Evol. Syst. 36, 47–79 (2005).
Eckert, C. G., Samis, K. E. & Dart, S. Reproductive Assurance and the Evolution of Uniparental Reproduction in Flowering Plants Ecology and evolution of flowers [Harder, L. D., & Barrett, S. C. H. (eds)] [183–203] (Oxford University Press, Oxford, 2006).
Fisher, R. A. Average excess and average effect of a gene substitution. Ann. Eugen. 11, 53–63 (1941).
Busch, J. W. & Delph, L. F. The relative importance of reproductive assurance and automatic selection as hypotheses for the evolution of self-fertilization. Ann. Bot. 109, 553 (2012).
Zhang, Z. Q. & Li, Q. J. Autonomous selfing provides reproductive assurance in an alpine ginger Roscoea schneideriana (Zingiberaceae). Ann. Bot. 102, 531–538 (2008).
Brys, R. & Jacquemyn, H. Variation in the functioning of autonomous self-pollination, pollinator services and floral traits in three Centaurium species. Ann. Bot. 107, 917–925 (2011).
Lloyd, D. G. Some Reproductive Factors Affecting the Selection of Self-Fertilization in Plants. Am. Nat. 113, 67–79 (1979).
Lloyd, D. G. & Schoen, D. J. Self- and Cross-Fertilization in Plants. I. Functional Dimensions. Int. J. Plant Sci. 153, 358–369 (1992).
Fenster, C. B. & Martén‐Rodríguez, S. Reproductive Assurance and the Evolution of Pollination Specialization. Int. J. Plant Sci. 168, 215–228 (2007).
Kalisz, S. et al. The mechanism of delayed selfing in Collinsia verna (Scrophulariaceae). Am. J. Bot. 86, 1239–1247 (1999).
Tsitrone, A., Duperron, S. & David, P. Delayed selfing as an optimal mating strategy in preferentially outcrossing species: theoretical analysis of the optimal age at first reproduction in relation to mate availability. Ame. Nat. 162, 318–331 (2003).
Zhang, C., Zhou, G. Y., Yang, Y. P. & Duan, Y. W. Better than nothing: Evolution of autonomous selfing under strong inbreeding depression in an alpine annual from the Qinghai-Tibet Plateau. J. Syst. Evol. 52, 363–367 (2014).
Kalisz, S. & Vogler, D. W. Benefits of autonomous selfing under unpredictable pollinator environments. Ecology 84, 2928–2942 (2003).
Jacquemyn, H. & Honnay, O. Mating system evolution under strong clonality: towards self-compatibility or self-incompatibility? Evol. Ecol. 22, 483–486 (2008).
Ornduff, R. Reproductive Biology in Relation to Systematics. Taxon 18, 121–133 (1969).
Elle, E. & Carney, R. Reproductive assurance varies with flower size in Collinsia parviflora (Scrophulariaceae). Am. J. Bot. 90, 888–896 (2003).
Carleial, S., van Kleunen, M. & Stift, M. Small reductions in corolla size and pollen: ovule ratio, but no changes in flower shape in selfing populations of the North American Arabidopsis lyrata. Oecologia 183, 401–413 (2017).
Kalisz, S. et al. Dichogamy correlates with outcrossing rate and defines the selfing syndrome in the mixed-mating genus Collinsia. Ann. Bot. 109, 571 (2012).
Elle, E., Gillespie, S., Guindre-Parker, S. & Parachnowitsch, A. L. Variation in the timing of autonomous selfing among populations that differ in flower size, time to reproductive maturity, and climate. Am. J. Bot. 97, 1894–1902 (2010).
Schouppe, D., Brys, R., Vallejo-Marin, M. & Jacquemyn, H. Geographic variation in floral traits and the capacity of autonomous selfing across allopatric and sympatric populations of two closely related Centaurium species. Sci. Rep. 7, 46410 (2017).
Brys, R., Geens, B., Beeckman, T. & Jacquemyn, H. Differences in dichogamy and herkogamy contribute to higher selfing in contrasting environments in the annual Blackstonia perfoliata (Gentianaceae). Ann. Bot. 111, 651–661 (2013).
Duan, Y. W., He, Y. P., Zhang, T. F. & Liu, J. Q. Delayed selfing in an alpine species Gentianopsis barbata. J. Plant. Ecol. 31, 110–117 (2007).
Duan, Y. W., Dafni, A., Hou, Q. Z., He, Y. P. & Liu, J. Q. Delayed selfing in an alpine biennial Gentianopsis paludosa (Gentianaceae) in the Qinghai‐Tibetan Plateau. J Integr. Plant Biol. 52, 593–599 (2010).
Kéry, M. & Matthies, D. Reduced fecundity in small populations of the rare plant Gentianopsis ciliate (Gentianaceae). Plant Biol. 6, 683–688 (2004).
Oostermeijer, J. G. B., Luijten, S. H., Ellis-Adam, A. C. & Nijs, J. C. M. D. Future prospects for the rare, late-flowering Gentianella germanica and Gentianopsis ciliata in Dutch nutrient-poor calcareous grasslands. Biol. Conserv. 104, 339–350 (2002).
Ho, T. N., & Pringle, J. S. Gentianaceae Flora of China, Vol. 16 [Wu, Z. -Y., & Raven P. H. (eds)]. (Sciences Press, Beijing and Missouri Botanical Garden, St. Louis, Missouri. 1995).
Dafni, A. Pollination ecology: a practical approach. (IRL Press Ltd, Oxford, 1992).
Cruden, R. & Lyon, D. Patterns of biomass allocation to male and female functions in plants with different mating systems. Oecologia 66, 299–306 (1985).
Guo, H., Mazer, S. J. & Du, G. Geographic variation in primary sex allocation per flower within and among 12 species of Pedicularis (Orobanchaceae): Proportional male investment increases with elevation. Am. J. Bot. 97, 1334 (2010).
Ren, Y.-Q., Xu, Y.-P., Zhang, T., Ma, Y.-P. & Marczewski, T. Growth discrepancy between filament and style facilitates self-fertilization in Brandisia hancei (Paulowniaceae). Plant Spec. Biol. 31, 153–158 (2016).
Lloyd, D. G. Allocations to Pollen, Seeds and Pollination Mechanisms in Self-Fertilizing Plants. Funct. Ecol. 1, 83–89 (1987).
Harder, L. D. & Wilson, W. G. Floral evolution and male reproductive success: optimal dispensing schedules for pollen dispersal by animal-pollinated plants. Evol. Ecol. 8, 542–559 (1994).
Harder, L. D. & Thomson, J. D. Evolutionary options for maximizing pollen dispersal of animal-pollinated plants. Ame. Nat. 133, 323–344 (1989).
Cruden, R. W. Pollen-Ovule Ratios: A Conservative indicator of breeding Systems in flowering plants. Evolution 31, 32–46 (1977).
Michalski, S. G. & Durka, W. Pollination mode and life form strongly affect the relation between mating system and pollen to ovule ratios. New Phytol. 183, 470–479 (2009).
Acknowledgements
This research was supported by the National Natural Science Foundation of China (no. 31500194). We would especially like to thank Potatso National Park and the Shangri-La Alpine Botany Garden for logistical support.
Author information
Authors and Affiliations
Contributions
X.F.Z., J.Q.Y., and Q.J.L. designed the research; J.Q.Y., X.F.Z., F.Y.L. and X.F.J. performed the experiments; J.Q.Y., F.Y.L. and X.F.Z. analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, JQ., Fan, YL., Jiang, XF. et al. Correlation between the timing of autonomous selfing and floral traits: a comparative study from three selfing Gentianopsis species (Gentianaceae). Sci Rep 8, 3634 (2018). https://doi.org/10.1038/s41598-018-21930-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-21930-9
This article is cited by
-
Autonomous selfing is altered by pollination in delayed-selfing species Gentianopsis paludosa (Gentianaceae)
Brazilian Journal of Botany (2022)
-
Dynamics of secondary pollen presentation in Campanula medium (Campanulaceae)
Journal of Plant Research (2019)
-
Spontaneous caudicle reconfiguration in Dactylorhiza fuchsii: A new self-pollination mechanism for Orchideae
Plant Systematics and Evolution (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.