Abstract
Early-life stress in adolescence has a long-lasting influence on brain function in adulthood, and it is mostly recognized as a predisposing factor for mental illnesses, such as anxiety and posttraumatic stress disorder. Previous studies also indicated that adolescent predictable chronic mild stress (PCMS) in early life promotes resilience to depression- and anxiety-like behaviors in adulthood. However, the role of PCMS in associated memory process is still unclear. In the present study, we found that adolescent PCMS facilitated extinction and inhibited fear response in reinstatement and spontaneous recovery tests in adult rats, and this effect was still present 1 week later. PCMS in adolescence increased the activity of brain-derived neurotrophic factor (BDNF)-extracellular signal-regulated kinase 1/2 (ERK1/2) signaling in infralimbic cortex (IL) but not prelimbic cortex in adulthood. Intra-IL infusion of BDNF antibody and the ERK1/2 inhibitor U0126 reversed PCMS-induced enhancement of fear extinction. Moreover, we found that PCMS decreased DNA methylation of the Bdnf gene at exons IV and VI and elevated the mRNA levels of Bdnf in the IL. Our findings indicate that adolescent PCMS exposure promotes fear memory extinction in adulthood, which reevaluates the traditional notion of adolescent stress.
Similar content being viewed by others
Introduction
Excessive fear and anxiety are hallmarks of anxiety disorders, such as posttraumatic stress disorder (PTSD). Understanding the mechanisms of fear inhibition is important for prevention and treatment of fear- and anxiety-related disorders1. Extinction of Pavlovian fear conditioning is an extensively used model for studying fear inhibition in rodents and humans2, 3. Extinction training does not erase the original fear memories but rather generates a new memory, known as extinction memory, to suppress fear responses4. Therefore, the extinguished fear can be spontaneously recovered, renewed, or reinstated5. Promoting extinction memory and preventing return of fear response is a prominent strategy to relieve fear-induced anxieties6.
Experience during early-life period can produce a long-lasting impact on adult behaviors and neuroendocrine effects7, 8. Decades of human and animal studies suggest that stress during adolescence may be a major risk factor for the development and persistence of mental disorders, including PTSD9,10,11,12. Early life stress and chronic exposure to corticosterone impair fear conditioning and extinction13, 14. Chronic unpredictable stress (CUS) during adolescence impairs stress-coping ability and cognitive flexibility15. Chronic restrain stress (CRS) slows fear extinction and enhances anxiety-like behavior16. On the contrary, predictable chronic mild stress (PCMS), stress exposure within a fixed time window, is reported to improve mood, learning, and memory in adult rats by increasing neurogenesis in the hippocampus17. Our previous study also suggested that adolescent PCMS had positive effects on brain functions and enhanced resistance to stress-induced depressive-like behavior in adulthood18. However, unknown is the effect of adolescent PCMS experience on fear conditioning and extinction in adulthood.
Medial prefrontal cortex (mPFC) has important roles in modulating the formation, expression and extinction of fear memory19, 20, and its different subregions may have distinct functions. Infralimbic cortex (IL) regulates inhibitory memory that underlies extinction and prelimbic cortex (PrL) modulates expression of learned fear21, 22. Brain-derived neurotrophic factor (BDNF) participates in many neuronal activity-dependent processes23, 24. BDNF knockout in the PrL impaired short- and long-term fear memory but not extinction memory25, whereas BDNF in the IL played a critical role in generation and maintenance of extinction memory26, 27. Otherwise, BDNF signaling and epigenetic modifications of Bdnf are correlated with the early life trauma. BDNF ameliorates CRS-induced impairments in spatial memory28 and its expression is inhibited by adolescent CUS exposure15. Early maltreatment produces persisting changes in DNA methylation of Bdnf 29. However, the roles of BDNF and its epigenetic regulation in the adaptive effect of PCMS are still unknown.
In the present study, we investigated whether PCMS exposure during adolescence promotes fear conditioning and extinction in adulthood. The underlying mechanisms concerning BDNF signaling and DNA methylation of Bdnf were also explored.
Results
PCMS but not CRS nor CUS during adolescence enhanced extinction of fear memory in adulthood
We first investigated whether PCMS, CRS and CUS during adolescence influenced locomotor activities. After 28 days of PCMS, CRS, CUS or handling, locomotor activity was tested. The total distances traveled were analyzed using one-way ANOVA and there was no significant main effect of stress. The Tukey’s post hoc test revealed that PCMS, CRS, and CUS had no effects on locomotor activities (all p > 0.05; Supplementary Fig. S1).
Then we investigated whether PCMS, CRS and CUS during adolescence impacted the acquisition, short-term memory (STM) and long-term memory (LTM) of contextual fear conditioning. One day after 28 days of PCMS, CRS, CUS or handling, the rats (n = 8–10 per group) underwent contextual fear conditioning (PND56) and were tested 1 h after training, and another four groups of rats (n = 8–10 per group) were tested 24 h after training (Fig. 1a). The freezing scores, expressed as the percentage of the freezing time, during conditioning were compared between groups, and repeated-measures ANOVA revealed that PCMS, CRS, and CUS had no effect on the acquisition of fear memory (p > 0.05). Tukey’s post hoc test showed there was no difference between groups (all p > 0.05; Fig. 1b). The freezing scores of STM and LTM tests were analyzed using one-way ANOVA and there was no significant main effect of stress. The Tukey’s post hoc test revealed that PCMS, CRS, or CUS had no effect on the STM and LTM of contextual fear conditioning, respectively (all p > 0.05; Fig. 1c).
PCMS but not CRS nor CUS during adolescence facilitated the extinction and prevented fear memory return in adulthood. (a,d) Experimental timeline. (b) PCMS, CRS or CUS during adolescence had no significant effect on the conditioning of contextual fear memory (n = 8–10 per group). (c) PCMS, CRS nor CUS during adolescence had no significant effect on the short term and long term fear memory (n = 8–10 per group). (e) PCMS exposure during adolescence reduced fear responses in the extinction test and reinstatement test in adulthood (n = 8–10 per group). (f) PCMS exposure during adolescence reduced fear responses in the extinction test and spontaneous recovery test in adulthood (n = 8–10 per group). The data are expressed as the mean ± SEM. *p < 0.05, compared with the control group. PCMS, predictable chronic mild stress; CRS, chronic restraint stress; CUS, chronic unpredictable stress.
To examine the effects of adolescent PCMS, CUS, and CRS on the extinction of fear memory, the rats (n = 9–10 per group) were subjected to extinction training 24 h after fear conditioning, extinction test 24 h after extinction training and reinstatement test 24 h after footshock (Fig. 1d ). The freezing scores during extinction training were analyzed using repeated-measures ANOVA, with extinction phase as the within-subjects factor and stress (control, PCMS, CRS, and CUS) as the between-subjects factor. The analysis revealed main effects of stress (F 3,32 = 41.884, p < 0.01) and extinction phase (F 5,160 = 35.514, p < 0.01) and an extinction phase × stress interaction (F 15,160 = 3.505, p < 0.01; Fig. 1e, line graph). Tukey’s post hoc test showed that freezing scores decreased in the PCMS group during phases II and IV, increased in the CRS group in phase IV, and increased in the CUS group in phases II-VI (all p < 0.05) compared with the control group. For the reinstatement of fear memory, the freezing scores during the extinction test and reinstatement test were analyzed using repeated-measures ANOVA, with test as within-subjects factor and stress (control, PCMS, CRS, and CUS) as the between-subjects factor. This analysis revealed significant effects of test (F 1,34 = 10.341, p < 0.01) and stress (F 3,34 = 17.181, p < 0.01; Fig. 1e, bar graph) but no test × stress interaction (p > 0.05). Tukey’s post hoc test revealed that freezing scores were decreased in the PCMS group during the extinction test and reinstatement test (both p < 0.05) and increased in the CRS group and CUS group in the extinction test (both p < 0.05) compared with the control group. Paired-samples t-test revealed that freezing scores of control rats were increased in reinstatement test compared with those in extinction test (t9 = −2.298, p < 0.05), whereas there was no significant difference in freezing scores between reinstatement test and extinction test in PCMS, CRS and CUS groups (all p > 0.05). These results indicate that PCMS in adolescence promoted extinction and prevented the reinstatement of fear memory in adulthood.
To examine the effects of PCMS, CUS, and CRS during adolescence on the spontaneous recovery of fear memory, after extinction training, the rats (n = 9–10 per group) were housed in their home cages for 2 weeks before spontaneous recovery test (Fig. 1d). The freezing scores during extinction were analyzed using repeated-measures ANOVA, with test as the within-subjects factor and stress (control, PCMS, CRS, and CUS) as the between-subjects factor. The analysis revealed significant main effects of stress (F 3,32 = 35.204, p < 0.01) and extinction phase (F 5,160 = 26.345, p < 0.01) and an extinction phase × stress interaction (F 15,160 = 2.954, p < 0.01; Fig. 1f, line graph). Tukey’s post hoc test showed that freezing scores decreased in the PCMS group during phases III and IV, increased in the CRS group in phase IV, and increased in the CUS group in phases II-VI (all p < 0.05), compared with control group. In the extinction test and spontaneous recovery test, the repeated-measures ANOVA analysis revealed significant effects of test (F 1,34 = 7.162, p < 0.05) and stress (F 3,34 = 18.041, p < 0.01; Fig. 1f, bar graph) but no test × stress interaction (p > 0.05). Tukey’s post hoc test revealed that freezing scores decreased in the PCMS group during the extinction test and spontaneous recovery test (both p < 0.05) and increased in the CRS group and CUS group in the extinction test (both p < 0.05) compared with the control group. Paired-samples t-test revealed that freezing scores in control group were significantly increased in spontaneous recovery test compared with those in extinction test (t9 = −2.320, p < 0.05), whereas there was no significant difference in freezing scores between spontaneous recovery test and extinction test in PCMS, CRS and CUS groups (all p > 0.05). These results indicate that PCMS in adolescence promoted extinction and blocked the spontaneous recovery of fear memory in adulthood.
Altogether, these results demonstrate that PCMS exposure in adolescence had no effect on the acquisition of fear memory, but enhanced extinction and prevented the return of fear memory in adulthood.
PCMS during adolescence enhanced the activity of BDNF-ERK1/2 signaling in the mPFC
We examined the effects of PCMS, CRS and CUS in adolescence on the activity of BDNF-ERK1/2 signaling in the mPFC in adulthood. One day after 28 days of PCMS, CRS, CUS or handling, the rats (n = 6 per group) were decapitated, and brain tissues were removed and then prepared for subsequent measurements of the levels of BDNF, total TrkB (TrkB), phosphorylated TrkB (pTrkB), total ERK1/2 (ERK1/2), and phosphorylated ERK1/2 (pERK1/2) in the mPFC (Fig. 2a). The western blot data in PrL and IL were analyzed among groups using One-way ANOVA, respectively. In the IL, Tukey’s post hoc test showed that, the levels of BDNF (F 3,20 = 11.673, p < 0.05), pTrkB (F 3,20 = 10.696, p < 0.05), and pERK1/2 (F 3,20 = 15.184, p < 0.05) were elevated in PCMS-exposed rats compared with the control group and the opposite trend of these proteins expression were found in CRS- and CUS-exposed rats, whereas no differences were found in the levels of TrkB and ERK1/2 among all groups (Fig. 2b). In the PrL, Tukey’s post hoc test showed that, the levels of BDNF (F 3,20 = 4.635, p < 0.05), pTrkB (F 3,20 = 4.909, p < 0.05), and pERK1/2 (F 3,20 = 3.778, p < 0.05) were decreased in CRS- and CUS-exposed rats compared with the control group but no significant changes were found in PCMS-exposed rats. Meanwhile, all of three stress had no effect on the expression of TrkB and ERK1/2 compared with the control group (all p > 0.05; Fig. 2c). These results indicate that PCMS during adolescence enhanced the activity of BDNF signaling in the IL.
PCMS during adolescence elevated BDNF-ERK1/2 signaling activity in the IL in adulthood. (a) Experimental timeline. (b,c) Protein levels and representative western blot bands of BDNF, pTrkB, TrkB, pERK1/2, and ERK1/2 in the IL (b) and PrL (c) in different stress manipulated and control rats. Blots shown are cropped from full-length. The data are expressed as the mean ± SEM (n = 6 per group). *p < 0.05, compared with the control group.
PCMS during adolescence caused long-lasting enhancement of fear memory extinction and suppressed reinstatement and spontaneous recovery
We next explored whether PCMS during adolescence has a prolonged effect on fear extinction in adulthood. One week after 28 days of PCMS or handling, the rats (n = 7–9 per group) underwent contextual fear conditioning (PND63) followed by extinction training. One day after the extinction test, the rats were given a footshock and tested for reinstatement 24 h later (Fig. 3a). The freezing scores during extinction training were analyzed using repeated-measures ANOVA, with extinction phase as the within-subjects factor and stress (control and PCMS) as the between-subjects factor. This analysis revealed significant main effects of extinction phase (F 5,70 = 43.356, p < 0.01), stress (F 1,14 = 6.580, p < 0.05) and a stress × extinction phase interaction (F 5,70 = 2.471, p < 0.05; Fig. 3b, line graph). Tukey’s post hoc test showed that freezing scores decreased in the PCMS group during phases III-V (all p < 0.05) compared with control group. In the extinction test and reinstatement test, the repeated-measures ANOVA analysis revealed significant effects of test (F 1,14 = 27.938, p < 0.01) and stress (F 1,14 = 18.033, p < 0.01) but no test × stress interaction (F 1,14 = 2.516, p = 0.135; Fig. 3b, bar graph). Tukey’s post hoc test showed that freezing scores decreased in the PCMS group in the extinction test and reinstatement test (both p < 0.05). Paired-samples t-test revealed that freezing scores in control group were significantly increased in reinstatement test compared with those in extinction test (t7 = −5.523, p < 0.01), whereas in PCMS group, freezing scores in reinstatement test were tendentially but not significantly higher than those in extinction test (t7 = −2.363, p = 0.05).
PCMS during adolescence had a prolonged extinction-enhancing effect. (a,d) Experimental timeline. (b,c) One week after adolescent PCMS exposure, the rats exhibited an acceleration of fear extinction and lower fear responses in the extinction test, reinstatement test, and spontaneous recovery test. (b) Fear responses were reduced in the extinction test and reinstatement test 1 week after PCMS exposure (n = 7–9 per group). (c) Fear responses in the extinction test and spontaneous recovery test were reduced 1 week after PCMS exposure (n = 7–9 per group). (e,f) Protein levels and representative western blot bands of BDNF, pTrkB, TrkB, pERK1/2, and ERK1/2 in the IL (e) and PrL (f) in PCMS-exposed and control rats (n = 8–10 per group). Blots shown are cropped from full-length. The data are expressed as the mean ± SEM. *p < 0.05, compared with the control group.
One day after the extinction test, another two groups of rats were housed in their home cages for another 2 weeks and tested for spontaneous recovery (Fig. 3a). The freezing scores during extinction training were analyzed using repeated-measures ANOVA, with extinction phase as the within-subjects factor and stress (control and PCMS) as the between-subjects factor. The analysis revealed significant main effects of extinction phase (F 5,70 = 37.415, p < 0.01) and stress (F 1,14 = 5.866, p < 0.05) but no stress × extinction phase interaction (F 5,70 = 1.766, p = 0.131; Fig. 3c, line graph). Tukey’s post hoc test showed that, freezing scores decreased in the PCMS group during phases III and IV compared with control group (both p < 0.05). The repeated-measures ANOVA analysis of freezing scores in the extinction test and spontaneous recovery test revealed significant effects of test (F 1,13 = 22.581, p < 0.01) and stress (F 1,13 = 29.223, p < 0.01) but no test × stress interaction (F 1,13 = 2.756, p = 0.121; Fig. 3c, bar graph). Tukey’s post hoc test showed, freezing scores decreased in the PCMS group during the extinction test and spontaneous recovery test compared with the control group (both p < 0.05). Paired-samples t-test revealed that freezing scores in control group were increased in spontaneous recovery test compared with those in extinction test (t7 = −7.143, p < 0.001), whereas there was no significant difference in freezing scores between spontaneous recovery test and extinction test in PCMS group (p > 0.05).
BDNF in the IL may protect individual from perplexity of fear memory26, 27. Above we found that BDNF-pERK1/2 signaling in the IL was activated 1 day after PCMS. We suspected that long-lasting enhancement of fear extinction by PCMS may be mediated by persistent activity of BDNF-ERK1/2 signaling in the IL. Thus, we explored whether PCMS in adolescence has a prolonged effect on the activation of BDNF-ERK1/2 signaling. One week after 28 days of PCMS or handling, the rats (n = 8–10 per group) were decapitated, and brain tissues were collected. The levels of BDNF, TrkB, pTrkB, ERK1/2, and pERK1/2 in the mPFC were measured (Fig. 3d). The western blot data were analyzed using Student’s t-test. In the IL, the levels of BDNF (t14 = −2.435, p < 0.05), pTrkB (t14 = −2.542, p < 0.05), and pERK1/2 (t14 = −3.690, p < 0.05) were elevated in PCMS-exposed rats compared with the control group, whereas no difference was found in the levels of TrkB and ERK1/2 (Fig. 3e). In the PrL. Student’s t-test revealed that the levels of BDNF, TrkB, pTrkB, ERK1/2, and pERK1/2 did not significantly change between groups (all p > 0.05; Fig. 3f).
These results indicate that the enhancement of extinction and suppression of reinstatement and spontaneous recovery of fear memory induced by adolescent PCMS exposure were still present 1 week after PCMS exposure, which may be associated with the activation of BDNF-ERK1/2 signaling in the IL.
Inhibition of BDNF in the IL prevented the PCMS-induced enhancement of fear extinction
BDNF regulated the development of the central nervous system by interacting with TrkB receptors30. The inactivation of BDNF-TrkB signaling effectively reversed behavioral changes that were induced by enhanced BDNF expression in the PFC31. To investigate the causal role of mPFC BDNF signaling in the effects of adolescent PCMS, we blocked BDNF activity by infusing BDNF antibody in the IL. The rats were assigned to control+vehicle, control+BDNF antibody, PCMS+vehicle, and PCMS+BDNF antibody groups (n = 8–10 per group) and underwent stereotaxic surgery after 28 days of PCMS exposure or handling, followed by 7 days of recovery. On PND63, the rats underwent fear conditioning, received an intra-IL infusion of BDNF antibody (0.5 µg/0.5 µl/side) or vehicle (saline) 30 min before fear extinction on the next day, and were subjected to the extinction test 24 h later. The day after the extinction test, the rats were given a footshock and tested for reinstatement 24 h later (Fig. 4a). The freezing scores during extinction training were analyzed using two-way repeated-measures ANOVA, with extinction phase as the within-subjects factor and drug (vehicle and BDNF antibody) and stress (control and PCMS) as the between-subjects factors. This analysis revealed significant effects of extinction phase (F 5,140 = 33.487, p < 0.05) and drug (F 1,28 = 10.991, p < 0.05, Fig. 4b). No significant effects of stress (F 1,28 = 1.582, p > 0.05) and extinction phase×drug interaction (F 5,140 = 1.002, p > 0.05) were found, but Tukey’s post hoc test revealed that freezing scores were lower in the PCMS+vehicle group than in the control+vehicle group during extinction phases V (p < 0.05; Fig. 4b, line graph). Tukey’s post hoc test also revealed that compared with the PCMS+vehicle group, the freezing scores in the PCMS+BDNF antibody group increased in phases V and VI (both p < 0.05). The analysis of the extinction test and reinstatement test using two-way repeated-measures ANOVA, with test (extinction test and reinstatement test) as the within-subject factors and drug (vehicle and BDNF antibody) and stress (control and PCMS) as the between-subjects factors, revealed significant effects of test (F 1,28 = 17.798, p < 0.05), stress (F 1,28 = 8.020, p < 0.05), and drug (F 1,28 = 27.275, p < 0.05; Fig. 4b, bar graph) and a stress × drug interaction (F 1,28 = 7.283, p < 0.05), but no test × stress, test × drug, or test × drug × stress interaction (all p > 0.05). In the extinction test and reinstatement test, Tukey’s post hoc test revealed freezing scores decreased in PCMS+vehicle group compared with the control+vehicle group (both p < 0.05; Fig. 4b, bar graph). However, Tukey’s post hoc test also showed that the attenuating effect of adolescent PCMS on freezing scores during the extinction test and reinstatement test were reversed by the intra-IL infusion of BDNF antibody (both p < 0.05; Fig. 4b, bar graph). These results indicate that the intra-IL infusion of BDNF antibody reversed the enhancing effect of PCMS on fear extinction.
Intra-IL infusion of BDNF antibody reversed the PCMS-induced enhancement of fear extinction. (a,c) Experimental timeline. (b) Intra-IL infusion of BDNF antibody (0.5 μg/0.5 μl per side) blocked the enhancement of extinction and decrease in reinstatement induced by PCMS. The data are expressed as the mean±SEM (n = 8–10 per group). *p < 0.05, compared with control+vehicle group; # p < 0.05, compared with PCMS+vehicle group. (d) Protein levels and representative western blot bands of pTrkB, TrkB, pERK1/2, and ERK1/2 in the IL after BDNF antibody infusion. Blots shown are cropped from full-length. The data are expressed as the mean±SEM (n = 6–8 per group). *p < 0.05, compared with control+no extinction group; # p < 0.05, compared with PCMS+vehicle+extinction group.
We then assessed the effects of BDNF inhibition in the IL on the expression of downstream signaling, including pTrkB, TrkB, pERK1/2, and ERK1/2. The rats were assigned to control+no extinction, control+extinction, PCMS+vehicle+extinction, and PCMS+BDNF antibody+extinction groups (n = 6–8 per group) and underwent fear conditioning on PND63. The next day, the PCMS+vehicle+extinction and PCMS+BDNF antibody+extinction groups received an intra-IL infusion of saline or BDNF antibody (0.5 µg/0.5 µl/side). Thirty minutes later, the control+extinction, PCMS+vehicle+extinction, and PCMS+BDNF antibody+extinction groups were subjected to extinction training. Two hours later, brain tissues were collected from the four groups (Fig. 4c). The western blot data were analyzed among groups using One-way ANOVA. There were significant effects on expression of pTrkB (F 3,27 = 3.349, p < 0.05) and pERK1/2 (F 3.27 = 10.359, p < 0.05). Tukey’s post hoc test revealed that, the extinction of conditioned fear was accompanied by a significant increase in pERK1/2 (p < 0.05; Fig. 4d) but not pTrkB, TrkB, or ERK1/2. Tukey’s post hoc test also showed that compared with the control+no extinction group, the levels of pTrkB and pERK1/2 increased in the PCMS+vehicle+extinction group, which were reversed by an intra-IL infusion of BDNF antibody (all p < 0.05; Fig. 4d). Student’s t-test revealed that infusion of BDNF antibody into IL decreased levels of pTrkB and pERK1/2 compared with PCMS+vehicle+extinction group (pTrkB: t10 = 2.454, p < 0.05; pERK1/2: t10 = 2.466, p < 0.05; Supplementary Fig. S3). These results indicate that BDNF signaling is involved in the enhancing effect of adolescent PCMS on fear extinction in adulthood.
Inhibition of ERK1/2 in the IL reversed the PCMS-induced enhancement of fear extinction
BDNF binds to TrkB receptors, triggering the activation of ERK1/2. The BDNF-ERK1/2 signaling pathway is crucial for the retention of fear memory32. To investigate the function of BDNF-ERK1/2 signaling in the IL in the enhancing effect of PCMS on fear extinction, we assigned rats to control+vehicle, control+U0126, PCMS+vehicle, and PCMS+U0126 groups and infused U0126, an inhibitor of ERK1/2 phosphorylation33, or vehicle (20% DMSO) in the IL 30 min before extinction training (Fig. 5a). The freezing scores during extinction training were analyzed using two-way repeated-measures ANOVA, with extinction phase as the within-subjects factor and stress (control and PCMS) and drug (vehicle and U0126) as the between-subjects factors. This analysis revealed significant effects of extinction phase (F 5,150 = 12.239, p < 0.05) and drug (F 1,30 = 20.437, p < 0.05) and an extinction phase × drug interaction (F 5,150 = 2.616, p < 0.05; Fig. 5b, line graph). No significant main effect of stress was found (F 1,30 = 3.956, p = 0.056). Tukey’s post hoc test revealed that freezing scores were higher in the PCMS+U0126 group than in the PCMS+vehicle group during extinction phases II and V-VI (both p < 0.05). Tukey’s post hoc test also showed that compared with the control+vehicle group, freezing scores significantly decreased in the PCMS+vehicle group in extinction phases II and V (p < 0.05). The analysis of the extinction test and reinstatement test using two way repeated-measures ANOVA, with test (extinction test and reinstatement test) as the within-subjects factor and drug (vehicle and U0126) and stress (control and PCMS) as the between-subjects factors, revealed significant effects of test (F 1,30 = 30.051, p < 0.05), stress (F 1,30 = 9.116, p < 0.05), and drug (F 1,30 = 16.692, p < 0.05) and a stress×drug interaction (F 1,30 = 4.240, p < 0.05; Fig. 5b, bar graph), but no test × stress, test × drug, or test × drug × stress interaction (all p > 0.05). Tukey’s post hoc test revealed that in the extinction test and reinstatement test, freezing scores decreased in the PCMS+vehicle group compared with the control+vehicle group (both p < 0.05). The attenuating effect of adolescent PCMS on freezing scores during the extinction test and reinstatement test were reversed by the intra-IL infusion of U0126 (both p < 0.05). These results indicate that inhibiting the activity of ERK1/2 in the IL reverses the effect of PCMS on fear extinction.
Intra-IL infusion of the ERK1/2 inhibitor U0126 reversed the PCMS-induced enhancement of fear extinction. (a) Experimental timeline. (b) U0126 blocked the effect of PCMS on extinction, extinction retention, and reinstatement. The data are expressed as the mean±SEM (n = 8–10 per group). *p < 0.05, compared with control+vehicle group; # p < 0.05, compared with PCMS+vehicle group.
PCMS during adolescence decreased methylation modification of the Bdnf gene
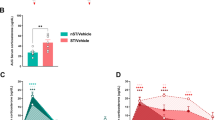
Methylation status is long lasting34 and DNA methylation regulates the transcription of several genes that are responsible for stress response35. Thus, we assessed whether PCMS exposure triggered prolonged changes in the DNA methylation of Bdnf. We first examined total mRNA levels of Bdnf at exon IX in the IL one week after rats were exposed to 28 days of PCMS during adolescence (Fig. 6a). Student’s t-test revealed that the levels of Bdnf mRNA in PCMS-exposed rats were significantly elevated in the IL compared with the control group (t 18 = −3.082, p < 0.05; Fig. 6b). These results indicate that Bdnf gene transcription increased in the IL after PCMS exposure during adolescence.
PCMS during adolescence increased Bdnf gene transcription and decreased the level of Bdnf DNA methylation in the IL. (a) Experimental timeline. (b) Total mRNA levels of Bdnf in the IL. (c–f) Levels of DNA methylation and unmethylation of Bdnf at exons I, II, IV, and VI in the IL. The data are expressed as the mean±SEM (n = 10 per group). *p < 0.05, compared with the control group.
The Bdnf gene has multiple initiation sites that allow isoform-specific mRNA transcription36. We investigated whether PCMS-induced alterations in Bdnf gene transcription were correlated with Bdnf DNA methylation. The analysis of the methylation-specific real-time PCR data using Student’s t-test showed that PCMS exposure during adolescence decreased the levels of methylated DNA at exon IV (t 18 = 2.577, p < 0.05; Fig. 6e) and exon VI (t 18 = 2.245, p < 0.05; Fig. 6f) and increased the levels of unmethylated DNA at exon IV (t 18 = −2.327, p < 0.05; Fig. 6e) and exon VI (t 18 = −2.252, p < 0.05; Fig. 6f). Student’s t-test showed that no significant changes in the levels of methylated and unmethylated DNA at exons I and II were found after PCMS exposure (all p > 0.05; Fig. 6c,d). These findings showed that PCMS during adolescence decreased the level of DNA methylation of Bdnf and increased Bdnf gene transcription in the adult IL.
Discussion
In the present study, we evaluated the effects of early-life PCMS exposure on fear conditioning and extinction. PCMS exposure during adolescence accelerated extinction and inhibited fear return in adulthood, while CUS and CRS suppressed extinction. The extinction-enhancement effect of PCMS was present 1 week after PCMS. PCMS increased the activity of BDNF-ERK1/2 signaling in IL. An intra-IL infusion of BDNF antibody or ERK1/2 inhibitor reversed the enhancing effect of adolescent PCMS on fear extinction in adulthood. Lastly, we found that PCMS exposure in adolescence decreased the levels of DNA methylation of Bdnf at exons IV and VI. Altogether, our results indicate that PCMS during adolescence promoted fear extinction and prevented fear return, which may be mediated by the BDNF-ERK1/2 signaling in IL.
Early experiences and experience-related neural changes play pivotal roles in emotion and cognition in adulthood37. In the present study, we found that PCMS exposure during adolescence enhanced fear extinction, and inhibited the return of fear memory. The results suggested that adolescent PCMS, which is a predictable and much milder stress, can produce a potentially adaptive advantage in adult stressful environments. The results were consistent with our previous study that PCMS during adolescence produced antidepressant- and anxiolytic-like effects and enhanced the abilities to resist depressive- and anxiety-like behaviors by chronic unpredictable stress (CUS) in adulthood18. Parihar et al. found PCMS during adulthood also decreased depressive- and anxiety-like behaviors and enhanced memory function in Morris water maze and novel object recognition, indicating that the protective effects of PCMS might not be specific for adolescence. However, adolescence, transiting from childhood to adulthood7, is a critical period of brain maturation, during which the brain undergo extensive morphological and functional remodeling38. Compared with adulthood, the stage of brain development in adolescence was vulnerable to environmental influence and experience38, 39. Stress in this brain-developing period could produce long-lasting impact on brain function while the effects of stress on adult brain were transient39, 40. It was also supported by our result that the extinction-enhancement effect of PCMS in adolescence can persist for at least one week. Overall, we speculate that PCMS in adolescence may affect brain development and maturation, thus producing long-lasting effect on behaviors in adulthood.
In this study, we found that PCMS exposure during adolescence accelerated extinction, while CUS and CRS suppressed extinction. This effect was stress paradigm-specific. The characteristics of the stressor include controllability, predictability, ambiguity, chronicity, and intermittence41. These specific characteristics of stressors are important factors in determining whether early life experiences ultimately produce protective or deleterious effect42. Prior chronic unpredictable and high strength events, including chronic unpredictable stress (CUS) and chronic restraint stress (CRS), usually lead to negative effects in later life. CUS during adolescence decreased BDNF-ERK1/2 signaling in mPFC and impaired stress-coping ability15. It also resulted in spatial memory impairment43. CRS during adolescence decreased hippocampal neurogenesis, leading to impairments in hippocampus-dependent fear memory44. CRS also increased anxiety-like behaviors and impaired the acquisition and retrieval of fear extinction16, 45,46,47,48. Chronic social-isolated adolescent rats showed significant deficits in the extinction of conditioned fear with the mechanism involving noradrenergic signaling48. CRS impaired acquisition of fear extinction in adolescent rats but not adult rats45. Consistent with these studies, we found that CUS and CRS suppressed the extinction of conditioned fear memory. In these cases, excessive intensity and duration of the stressor exceeded endurance, inducing aberrant adaptation. In contrast, some controllable and mild stressors help adolescents develop adaptive stress responses and become resilient to negative stressor in future17, 18, 49. For example, intermittent mother-offspring separation and PCMS relieved anxiety18, 50. We found that PCMS in adolescence can facilitate extinction and prevent fear return in the present study. However, the protective and adaptive roles of specific stress and the underlying mechanisms need to be further investigated. Collectively, our results compelled a reevaluation of the traditional notion that early stress predicts adverse outcomes in the future.
For the mechanisms underlying the protective effect of PCMS, a previous study showed that PCMS moderately increased corticosterone concentrations for shorter durations, which facilitated beneficial responses in the brain17. Mild stress or lower doses of corticosterone or dexamethasone induced transient increases in the levels of neurotrophic factors, such as BDNF, insulin-like growth factor-1, and fibroblast growth factor-251, 52. Moreover, animals with early stress that exhibited enhanced BDNF levels had improved performance in the Morris water maze42. In the present study, rats that were exposed to PCMS in adolescence exhibited higher levels of BDNF, indicating that adolescent PCMS exposure might closely resemble the effects of low-dose corticosterone exposure. Besides, our previous study demonstrated that adolescent PCMS had positive effects on brain functions and promoted the resistance to stress in adulthood, and mPFC is critical for these adaptive changes18. In this study, PCMS exposure in adolescence increased the levels of BDNF, pTrkB, and pERK1/2 in IL, while CUS and CRS decreased levels of BDNF, pTrkB and pERK1/2 in both IL and PrL. The enhancement of extinction memory induced by adolescent PCMS exposure was offset by the inhibition of BDNF or ERK1/2 in IL. These results were consistent with previous studies that fear extinction requires BDNF in IL but not PrL26, 27 and requires the activation of ERK1/2 signaling53,54,55. CUS exposure during adolescence inhibited BDNF- ERK1/2 signaling in adult mPFC, which was consistent with our present study15. Besides, previous studies reported that dendritic morphology in mPFC was changed by chronic stress, e.g. CRS decreased the spine density of pyramidal neurons in mPFC56, 57. BDNF overexpression could increase spine density in hippocampus and amygdala58, 59. These finding indicate that BDNF-ERK1/2 signaling in IL plays a vital role in the extinction-enhancing effect of adolescent PCMS exposure.
DNA methylation is an epigenetic modification, regulating gene expression by adding a methyl group at the C5 position of cytosine to form 5-methylcytosine, and plays a critical role in gene silencing through chromatin remodeling60. The methylation changes were enduring11. Early neglect or trauma can epigenetically program the stress system, leading to aberrant regulation of the HPA axis and maladaptive, prolonged responses to stressors encountered in later life11, 35, 61, 62. Increases in BDNF were shown to be associated with a decrease in methylation of the Bdnf gene63, 64. Epigenetic modifications of Bdnf were related to memory, stress, and neuropsychiatric disorders64. The predator exposure significantly increased Bdnf DNA methylation in the dorsal hippocampus65. Maternal separation decreased repressive histone methylation of the Bdnf exon IV promoter and enhanced BDNF levels42. In the present study, an increase in the level of Bdnf mRNA and decrease in the levels of Bdnf methylation at exons IV and VI were found in adolescent PCMS-exposed rats, which might influence the persisting BDNF expression and brain functions in adulthood29, 66. Our results were consistent with previous findings that Bdnf exon IV is regulated by fear memory and early life adversity29, 35. Histone methylation at the Bdnf IV promoter was decreased in maternal separated rats with improved performance in the Morris water maze in adulthood42. With regard to previous findings that BDNF in IL is involving in generation and maintainance of extinction memory26, 27, we speculated that epigenetic regulation of BDNF expression in IL is crucial for the enhancement effect of adolescent PCMS exposure on fear extinction in adulthood.
In conclusion, our results indicate that PCMS exposure during adolescence promoted fear extinction and inhibited return of fear memory in adulthood, while CUS and CRS suppressed extinction. BDNF-related signaling pathways and epigenetic modification of the Bdnf gene may be involved in the enhancement of fear extinction by adolescent PCMS exposure. Our results support the hypothesis that specific predictable mild stress exposure during adolescence may ameliorate fear-related disorders.
Materials and Methods
Animals
All of the experiments were performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and were approved by the Biomedical Ethics Committee for Animal Use and Protection of Peking University. Three hundred and sixty-one male Sprague Dawley rats, 21 days old and weighing 45–55 g upon arrival, were obtained from the Laboratory Animal Center, Peking University Health Science Center. The animals were allowed to acclimate for 7 days before stress exposure. All of the rats were housed under standard laboratory conditions with controlled room temperature (23 ± 2 °C) and humidity (50 ± 5%) and free access to food and water under a 12 h/12 h light/dark cycle.
Predictable chronic mild stress
The PCMS protocol was conducted according to previous studies17, 18. Beginning on postnatal day 28 (PND28), the rats were guided into stainless-steel cylinders that had a ventilation grille on the head side and sliding door on the tail side. They remained inside the cylinder for 5 min daily, during which they were unable to move. They were then returned to their home cages. The size of the restraint cylinder was adjusted to the growth of rats. The stress paradigm was conducted for 28 continuous days at the same time each day (between 3:00 PM and 5:00 PM) to ensure that the time of the stress was predictable. Animals in the control group received no stress but were handled every day.
Chronic unpredictable stress
The chronic unpredictable stress (CUS) protocol was based on previous studies18, 67, 68. A total of 12 different stressors were used, including cold for 1 h at 4 °C, restraint for 1 h, food deprivation for 24 h, water deprivation for 24 h, light/dark cycle reversal for 36 h, vibration for 1 h, tilting cages (45°) for 24 h, forced cold swim for 5 min, crowding for 24 h, no bedding for 24 h, soiled bedding for 24 h, and tail clamp for 1 min. Beginning on PND28, each rat was exposed to two of these stressors each day for 28 continuous days. The time at which the stressors were applied was unpredictable.
Chronic restraint stress
The chronic restraint stress (CRS) protocol was conducted as previously described18. Beginning on PND28, each rat was fixed in a stainless-steel cylinder (the same one used for PCMS), where it remained immobile for 2 h daily (3:00 PM to 5:00 PM) for 28 continuous days.
Contextual fear conditioning
Contextual fear conditioning was conducted using a video-based fear conditioning system (Beijing Macro Ambition S&T Development Co., Ltd, Beijing, China) as previously reported69, 70. All of the procedures were conducted in conditioning chambers (30 cm × 30 cm × 50 cm). The walls were constructed with black polyvinyl chloride and the floor was constructed with stainless steel rods (0.5 cm diameter, 1.0 cm apart) that were used to deliver foot shocks. Each conditioning chamber was enclosed in an acoustic isolation box. A dim light illuminated the chamber during the procedures.
Conditioning
On the training day, the rats were placed in the conditioning chamber and allowed to explore it for 2 min, after which they received three electric footshocks (0.8 mA, 1 s) at 2-min intervals. After the last shock, the rats were allowed to explore the conditioning chamber for an additional 1 min before being returned to their home cages. The chambers were cleaned with 75% alcohol to eliminate any residual odor before placing the rats in them.
Extinction
During extinction, the rats were exposed to the conditioning chamber for 30 min without footshock. The 30-min extinction training was divided into six phases (phase I: 0–5 min, phase II: 6–10 min, phase III: 11–15 min, phase IV: 16–20 min, phase V: 21–25 min, and phase VI: 26–30 min).
Test
All memory tests were conducted by exposing the rats to the conditioning chamber without footshock for 5 min. The extinction test was performed 24 h after extinction. In reinstatement experiment, the rats received a footshock (0.8 mA, 1 s) in another context 24 h after the extinction test. The reinstatement test was conducted 24 h after the footshock. The spontaneous recovery test was performed in another group of rats 2 weeks after the extinction test.
Freezing
Freezing behavior was video-recorded and offline analyzed using JLbehv-LAG-4 software (Shanghai Jiliang Software Technology Co. Ltd, Shanghai, China). The freezing score is expressed as the percentage of the time spent freezing during the test. Freezing behavior was defined as the lack of all movements, with the exception of respiration.
Locomotor Activity Test
Locomotor Activity Test was conducted based on previous studies69, 71. Locomotor activity was measured with an automated video tracking system (DigBehv-LM4; Shanghai Jiliang Software Technology, Shanghai, China) that contained clear Plexiglas chambers (40 cm*40 cm*65 cm). Vdeo files were analyzed using DigBehv analysis software. Locomotor activity is expressed as the total distance traveled in centimeters during a predetermined period of time (in 30 min).
Surgery
The rats (weighing 270–290 g) were anesthetized with sodium pentobarbital (50 mg/kg, i.p.). Stainless-steel guide cannulas (22 gauge) were bilaterally implanted in the IL (anterior/posterior, +2.9 mm; medial/lateral, ±2.3 mm; dorsal/ventral, −4.8 mm)72,73,74,. The cannulas were placed at a 16° angle toward the midline to avoid penetration of the lateral ventricle. The cannulas were anchored to the skull with screws and dental cement. After surgery, the rats were single-housed and allowed 7 days to recover, during which they were handled every day until fear conditioning.
Intracranial drug infusions and systemic treatment
BDNF antibody (lyophilized powder; Millipore, Bedford, MA, USA) was dissolved in saline and intracranially administered at a dose of 0.5 µg/0.5 µl/side based on a previous study75. U0126 (Sigma-Aldrich, St. Louis, MO, USA), an inhibitor of extracellular signal-regulated kinase 1/2 (ERK1/2), was dissolved in 20% dimethylsulfoxide (DMSO) and intracranially administered (100 ng/0.5 µl/side) according to our previous studies76, 77. For intracranial drug infusions, the drugs were infused bilaterally over 2 min, and the needle was kept in place for an additional 1 min to allow drug diffusion.
Histology
The cannula placements were confirmed in 30-μm-thick sections using Nissl staining under light microscopy (Supplementary Fig. S2). Five rats with misplaced cannulas were excluded from the statistical analysis. Indian ink (0.5 µl/side) was infused bilaterally into IL to confirm the diffusion route (Supplementary Fig. S2).
Western blot assays
The rats were decapitated 1 or 7 days after 28 days of PCMS exposure or handling (control), or 2 h after fear extinction training, and the protocol of tissue preparing and western blot was based on our previous study6, 72, 78, 79. The brains were rapidly extracted and frozen in dry ice. The brains were stored at −80 °C. Bilateral tissue punches (12 gauge) of the PrL and IL were lysed in RIPA lysis buffer (Applygen Technology, Beijing, China) with protease inhibitor and phosphatase inhibitor mixture for 30 min. They were then homogenized (15 s × 3, 5 s intervals) with an electrical disperser (Wiggenhauser, Sdn Bhd). The tissue homogenates were subjected to 10,000 × g centrifugation at 4 °C for 15 min. The protein concentrations of all of the samples were determined using the bicinchoninic acid (BCA) assay kit (Beyotime Biotechnology, Shanghai, China). All of the samples were further diluted in RIPA lysis buffer to normalize the protein concentrations. Five × loading buffer (16% glycerol, 20% mercaptoethanol, 2% sodium dodecyl sulfate [SDS], 0.05% bromophenol blue) was added to each sample (4:1, sample: loading buffer) before boiling for 5 min. The samples were stored at −80 °C until analysis. The samples were subjected to SDS-polyacrylamide gel electrophoresis (10% acrylamide/0.27% N,N’-methylenebisacrylamide resolving gel) for approximately 40 min at 80 V in stacking gel and approximately 1 h at 120 V in resolving gel. Proteins were electrophoretically transferred to polyvinylidene fluoride membranes (Millipore, Bedford, MA, USA) at 250 mA for 1–3 h. Membranes were washed with Tris-buffered saline plus 0.05% Tween-20 (TBST, pH 7.4) and then incubated in blocking buffer (5% bovine serum albumin in TBST) overnight at 4 °C. The next day, the membranes were incubated for 45 min at room temperature on a shaker with anti-BDNF antibody (1:1000, Abcam, Cambridge, UK), anti-tropomyosin-related kinase B (TrkB) antibody (1:1000, Abcam, Cambridge, UK), anti-pTrkB antibody (1:1000, Abcam, Cambridge, UK), anti-ERK1/2 antibody (1:1000, Cell Signaling Technology, MA, USA), anti-pERK1/2 antibody (1:1000, Cell Signaling Technology, MA, USA), or anti-β-actin antibody (1:2000, Sigma-Aldrich, St. Louis, MO, USA) in TBST plus 5% bovine serum albumin and 0.05% sodium azide. After four 5-min washes in TBST buffer, the blots were incubated for 45–55 min at room temperature on a shaker with horseradish peroxidase (HRP)-conjugated secondary antibody (goat anti-rabbit immunoglobulin G for BDNF, TrkB, pTrkB, ERK1/2, pERK1/2; goat anti-mouse for β-actin; Santa Cruz Biotechnology, Santa Cruz, CA, USA) diluted 1:5000 in blocking buffer. The blots then underwent four 5-min washes with TBST, and immunostaining was visualized using the EZ-ECL chemiluminescence detection kit. The immunoblots were quantified with the Gel Doct EZ system (Bio-Rad, Hercules, CA, USA). Band intensities for BDNF, TrkB, pTrkB, ERK1/2, and pERK1/2 were normalized to β-actin and analyzed using Quantity One 4.4.0 software (Bio-Rad, Hercules, CA, USA).
Measuring mRNA levels by RT-PCR
The protocol was based on our previous study71. The preparation of brain tissues from the IL was the same as western blot assay, and total RNA was extracted using the PureLink RNA Micro Kit (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. The purified RNA samples were stored at −80 °C until the RT-PCR assay. Reverse transcription was performed using the PrimeScript RT reagent Kit (TakaRa, otsu,Japan) and SYBR select master mix (Invitrogen, Carlsbad, CA, USA). Genomic DNA removal was performed using the CFX96 real-time PCR system (Bio-Rad, Hercules, CA, USA) at 42 °C for 2 min. cDNA amplification was performed at 95 °C for 30 s, followed by 40 cycles of 95 °C for 5 s and 60 °C for 30 s and incubation at 70 °C for 10 min. β-Tubulin-4 quantification was used as an internal control to normalize mRNA levels. The primers for Bdnf mRNA at exon IX and β-tubulin-4 mRNA were synthesized according to a previous study29.
Methylation-specific real-time PCR analysis
The protocol was based on our previous study71. DNA was isolated from brain tissues of the IL and purified using the TIANamp Micro DNA Kit (Tiangen, Beijing, China) according to the manufacturer’s instructions. Methylation levels were assessed using methylation-specific real-time PCR. The DNA was processed for bisulfite modification using the BisulFlash DNA modification kit (Eptigentek, Farmingdale, NY, USA). Quantitative RT-PCR was used to examine the DNA methylation of Bdnf. β-Tubulin-4 quantification was used as an internal control to normalize the DNA methylation levels. The methylated and unmethylated primers for Bdnf at exons I, II, IV, and VI and β-Tubulin-4 were based on a previous study29. The comparative cycle threshold method was used to calculate differences in gene expression between samples. DNA methylation status in different manipulation groups is presented as fold changes relative to the naive group.
Statistical analysis
All of the statistical analyses were performed using SPSS 17.0 software (SPSS, Chicago, IL, USA). The data are expressed as mean ± standard error of mean (SEM) and were analyzed using analysis of variance (ANOVA) with appropriate between- and within-subjects factors or using the paired-samples t-test or student’s t-test (see Results for details). Significant main effects and interactions (p < 0.05, two-tailed) in the factorial ANOVAs were further analyzed using Tukey’s post hoc test as appropriate. Values of p < 0.05 were considered statistically significant.
Data Availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Kheirbek, M. A., Klemenhagen, K. C., Sahay, A. & Hen, R. Neurogenesis and generalization: a new approach to stratify and treat anxiety disorders. Nat Neurosci 15, 1613–1620 (2012).
Pavlov, I. P. Conditioned Reflexes Oxford University Press: London (1927).
Myers, K. M. & Davis, M. Mechanisms of fear extinction. Mol Psychiatry 12, 120–150 (2007).
Bouton, M. E. & King, D. A. Contextual control of the extinction of conditioned fear: tests for the associative value of the context. J Exp Psychol Anim Behav Process 9, 248–265 (1983).
Maren, S., Phan, K. L. & Liberzon, I. The contextual brain: implications for fear conditioning, extinction and psychopathology. Nat Rev Neurosci 14, 417–428 (2013).
Liu, J. F. et al. Role of hippocampal beta-adrenergic and glucocorticoid receptors in the novelty-induced enhancement of fear extinction. J Neurosci 35, 8308–8321 (2015).
Spear, L. P. The adolescent brain and age-related behavioral manifestations. Neurosci Biobehav Rev 24, 417–463 (2000).
McCormick, C. M., Mathews, I. Z., Thomas, C. & Waters, P. Investigations of HPA function and the enduring consequences of stressors in adolescence in animal models. Brain Cogn 72, 73–85 (2010).
McCormick, C. M., Smith, C. & Mathews, I. Z. Effects of chronic social stress in adolescence on anxiety and neuroendocrine response to mild stress in male and female rats. Behav Brain Res 187, 228–238 (2008).
Pohl, J., Olmstead, M. C., Wynne-Edwards, K. E., Harkness, K. & Menard, J. L. Repeated exposure to stress across the childhood-adolescent period alters rats’ anxiety- and depression-like behaviors in adulthood: The importance of stressor type and gender. Behav Neurosci 121, 462–474 (2007).
Ross, D. A. et al. An Integrated Neuroscience Perspective on Formulation and Treatment Planning for Posttraumatic Stress Disorder: An Educational Review. JAMA Psychiatry 74, 407–415 (2017).
Brewin, C. R., Andrews, B. & Valentine, J. D. Meta-analysis of risk factors for posttraumatic stress disorder in trauma-exposed adults. J Consult Clin Psychol 68, 748–766 (2000).
Gourley, S. L., Kedves, A. T., Olausson, P. & Taylor, J. R. A history of corticosterone exposure regulates fear extinction and cortical NR2B, GluR2/3, and BDNF. Neuropsychopharmacology 34, 707–716 (2009).
Stevenson, C. W., Spicer, C. H., Mason, R. & Marsden, C. A. Early life programming of fear conditioning and extinction in adult male rats. Behav Brain Res 205, 505–510 (2009).
Zhang, Y., Shao, F., Wang, Q., Xie, X. & Wang, W. Neuroplastic correlates in the mPFC underlying the impairment of stress-coping ability and cognitive flexibility in adult rats exposed to chronic mild stress during adolescence. Neural Plast 2017, 9382797 (2017).
Negron-Oyarzo, I., Perez, M. A., Terreros, G., Munoz, P. & Dagnino-Subiabre, A. Effects of chronic stress in adolescence on learned fear, anxiety, and synaptic transmission in the rat prelimbic cortex. Behav Brain Res 259, 342–353 (2014).
Parihar, V. K., Hattiangady, B., Kuruba, R., Shuai, B. & Shetty, A. K. Predictable chronic mild stress improves mood, hippocampal neurogenesis and memory. Mol Psychiatry 16, 171–183 (2011).
Suo, L. et al. Predictable chronic mild stress in adolescence increases resilience in adulthood. Neuropsychopharmacology 38, 1387–1400 (2013).
Sotres-Bayon, F. & Quirk, G. J. Prefrontal control of fear: more than just extinction. Curr Opin Neurobiol 20, 231–235 (2010).
Quirk, G. J., Garcia, R. & Gonzalez-Lima, F. Prefrontal mechanisms in extinction of conditioned fear. Biol Psychiatry 60, 337–343 (2006).
Milad, M. R. & Quirk, G. J. Neurons in medial prefrontal cortex signal memory for fear extinction. Nature 420, 70–74 (2002).
Herry, C. & Garcia, R. Prefrontal cortex long-term potentiation, but not long-term depression, is associated with the maintenance of extinction of learned fear in mice. J Neurosci 22, 577–583 (2002).
Alonso, M. et al. BDNF-triggered events in the rat hippocampus are required for both short- and long-term memory formation. Hippocampus 12, 551–560 (2002).
Alonso, M., Vianna, M. R., Izquierdo, I. & Medina, J. H. Signaling mechanisms mediating BDNF modulation of memory formation in vivo in the hippocampus. Cell Mol Neurobiol 22, 663–674 (2002).
Choi, D. C. et al. Prelimbic cortical BDNF is required for memory of learned fear but not extinction or innate fear. Proc Natl Acad Sci USA 107, 2675–2680 (2010).
Peters, J., Dieppa-Perea, L. M., Melendez, L. M. & Quirk, G. J. Induction of fear extinction with hippocampal-infralimbic BDNF. Science 328, 1288–1290 (2010).
Rosas-Vidal, L. E., Do-Monte, F. H., Sotres-Bayon, F. & Quirk, G. J. Hippocampal–prefrontal BDNF and memory for fear extinction. Neuropsychopharmacology 39, 2161–2169 (2014).
Radecki, D. T., Brown, L. M., Martinez, J. & Teyler, T. J. BDNF protects against stress-induced impairments in spatial learning and memory and LTP. Hippocampus 15, 246–253 (2005).
Roth, T. L., Lubin, F. D., Funk, A. J. & Sweatt, J. D. Lasting epigenetic influence of early-life adversity on the BDNF gene. Biol Psychiatry 65, 760–769 (2009).
Klein, R. et al. The trkB tyrosine protein kinase is a receptor for brain-derived neurotrophic factor and neurotrophin-3. Cell 66, 395–403 (1991).
Vassoler, F. M., White, S. L., Schmidt, H. D., Sadri-Vakili, G. & Pierce, R. C. Epigenetic inheritance of a cocaine-resistance phenotype. Nat Neurosci 16, 42–47 (2013).
Castillo, D. V. & Escobar, M. L. A role for MAPK and PI-3K signaling pathways in brain-derived neurotrophic factor modification of conditioned taste aversion retention. Behav Brain Res 217, 248–252 (2011).
Davies, S. P., Reddy, H., Caivano, M. & Cohen, P. Specificity and mechanism of action of some commonly used protein kinase inhibitors. Biochem J 351, 95–105 (2000).
Miller, C. A. et al. Cortical DNA methylation maintains remote memory. Nat Neurosci 13, 664–666 (2010).
Kundakovic, M. et al. DNA methylation of BDNF as a biomarker of early-life adversity. Proc Natl Acad Sci U S A 112, 6807–6813 (2015).
Timmusk, T. et al. Multiple promoters direct tissue-specific expression of the rat BDNF gene. Neuron 10, 475–489 (1993).
Lupien, S. J., McEwen, B. S., Gunnar, M. R. & Heim, C. Effects of stress throughout the lifespan on the brain, behaviour and cognition. Nat Rev Neurosci 10, 434–445 (2009).
Casey, B. J., Getz, S. & Galvan, A. The adolescent brain. Dev Rev 28, 62–77 (2008).
Chen, Y. & Baram, T. Z. Toward understanding how early-life stress reprograms cognitive and emotional brain networks. Neuropsychopharmacology 41, 197–206 (2016).
van Os, J., Kenis, G. & Rutten, B. P. The environment and schizophrenia. Nature 468, 203–212 (2010).
Anisman, H. & Matheson, K. Stress, depression, and anhedonia: caveats concerning animal models. Neurosci Biobehav Rev 29, 525–546 (2005).
Suri, D. et al. Early stress evokes age-dependent biphasic changes in hippocampal neurogenesis, BDNF expression, and cognition. Biol Psychiatry 73, 658–666 (2013).
Borcel, E. et al. Chronic stress in adulthood followed by intermittent stress impairs spatial memory and the survival of newborn hippocampal cells in aging animals: prevention by FGL, a peptide mimetic of neural cell adhesion molecule. Behav Pharmacol 19, 41–49 (2008).
Yun, J. et al. Chronic restraint stress impairs neurogenesis and hippocampus-dependent fear memory in mice: possible involvement of a brain-specific transcription factor Npas4. J Neurochem 114, 1840–1851 (2010).
Zhang, W. & Rosenkranz, J. A. Repeated restraint stress enhances cue-elicited conditioned freezing and impairs acquisition of extinction in an age-dependent manner. Behav Brain Res 248, 12–24 (2013).
Schneider, A. M. et al. Stress-dependent opioid and adrenergic modulation of newly retrieved fear memory. Neurobiol Learn Mem 109, 1–6 (2014).
Wilber, A. A. et al. Chronic stress alters neural activity in medial prefrontal cortex during retrieval of extinction. Neuroscience 174, 115–131 (2011).
Skelly, M. J., Chappell, A. E., Carter, E. & Weiner, J. L. Adolescent social isolation increases anxiety-like behavior and ethanol intake and impairs fear extinction in adulthood: Possible role of disrupted noradrenergic signaling. Neuropharmacology 97, 149–159 (2015).
Southwick, S. M. & Charney, D. S. The science of resilience: implications for the prevention and treatment of depression. Science 338, 79–82 (2012).
Parker, K. J., Buckmaster, C. L., Schatzberg, A. F. & Lyons, D. M. Prospective investigation of stress inoculation in young monkeys. Arch Gen Psychiatry 61, 933–941 (2004).
Molteni, R. et al. Acute stress responsiveness of the neurotrophin BDNF in the rat hippocampus is modulated by chronic treatment with the antidepressant duloxetine. Neuropsychopharmacology 34, 1523–1532 (2009).
Molteni, R. et al. Modulation of fibroblast growth factor-2 by stress and corticosteroids: from developmental events to adult brain plasticity. Brain Res Brain Res Rev 37, 249–258 (2001).
Herry, C., Trifilieff, P., Micheau, J., Luthi, A. & Mons, N. Extinction of auditory fear conditioning requires MAPK/ERK activation in the basolateral amygdala. Eur J Neurosci 24, 261–269 (2006).
Fischer, A. et al. Hippocampal Mek/Erk signaling mediates extinction of contextual freezing behavior. Neurobiol Learn Mem 87, 149–158 (2007).
Leon, W. C., Bruno, M. A., Allard, S., Nader, K. & Cuello, A. C. Engagement of the PFC in consolidation and recall of recent spatial memory. Learn Mem 17, 297–305 (2010).
Radley, J. J. et al. Repeated stress induces dendritic spine loss in the rat medial prefrontal cortex. Cereb Cortex 16, 313–320 (2006).
Cook, S. C. & Wellman, C. L. Chronic stress alters dendritic morphology in rat medial prefrontal cortex. J Neurobiol 60, 236–248 (2004).
An, J. J. et al. Distinct role of long 3′ UTR BDNF mRNA in spine morphology and synaptic plasticity in hippocampal neurons. Cell 134, 175–187 (2008).
Govindarajan, A. et al. Transgenic brain-derived neurotrophic factor expression causes both anxiogenic and antidepressant effects. Proc Natl Acad Sci USA 103, 13208–13213 (2006).
Moore, L. D., Le, T. & Fan, G. DNA methylation and its basic function. Neuropsychopharmacology 38, 23–38 (2013).
Gunnar, M. & Quevedo, K. The neurobiology of stress and development. Annu Rev Psychol 58, 145–173 (2007).
McGowan, P. O. et al. Epigenetic regulation of the glucocorticoid receptor in human brain associates with childhood abuse. Nat Neurosci 12, 342–348 (2009).
Martinowich, K. et al. DNA methylation-related chromatin remodeling in activity-dependent BDNF gene regulation. Science 302, 890–893 (2003).
Andero, R. & Ressler, K. J. Fear extinction and BDNF: translating animal models of PTSD to the clinic. Genes Brain Behav 11, 503–512 (2012).
Roth, T. L., Zoladz, P. R., Sweatt, J. D. & Diamond, D. M. Epigenetic modification of hippocampal Bdnf DNA in adult rats in an animal model of post-traumatic stress disorder. J Psychiatr Res 45, 919–926 (2011).
Lubin, F. D., Roth, T. L. & Sweatt, J. D. Epigenetic regulation of BDNF gene transcription in the consolidation of fear memory. J Neurosci 28, 10576–10586 (2008).
Willner, P., Towell, A., Sampson, D., Sophokleous, S. & Muscat, R. Reduction of sucrose preference by chronic unpredictable mild stress, and its restoration by a tricyclic antidepressant. Psychopharmacology (Berl) 93, 358–364 (1987).
Zhu, W. L. et al. Hippocampal CA3 calcineurin activity participates in depressive-like behavior in rats. J Neurochem 117, 1075–1086 (2011).
Chai, N. et al. Delayed noradrenergic activation in the dorsal hippocampus promotes the long-term persistence of extinguished fear. Neuropsychopharmacology 39, 1933–1945 (2014).
Liu, J. et al. An unconditioned stimulus retrieval extinction procedure to prevent the return of fear memory. Biol Psychiatry 76, 895–901 (2014).
Chen, C. et al. Epigenetic modification of PKMzeta rescues aging-related cognitive impairment. Sci Rep 6, 22096 (2016).
Xue, Y. X. et al. Overexpression of protein kinase M zeta in the prelimbic cortex enhances the formation of long-term fear memory. Neuropsychopharmacology 40, 2146–2156 (2015).
He, Y. Y. et al. PKMzeta maintains drug reward and aversion memory in the basolateral amygdala and extinction memory in the infralimbic cortex. Neuropsychopharmacology 36, 1972–1981 (2011).
Paxinos, G. & Watson, C. The Rat Brain in Stereotaxic Coordinates. 5th Edn. Elsevier Academic Press: San Diego, CA. (2005).
Radiske, A. et al. Requirement for BDNF in the reconsolidation of fear extinction. J Neurosci 35, 6570–6574 (2015).
Lu, L. et al. Central amygdala ERK signaling pathway is critical to incubation of cocaine craving. Nat Neurosci 8, 212–219 (2005).
Li, Y. Q. et al. Central amygdala extracellular signal-regulated kinase signaling pathway is critical to incubation of opiate craving. J Neurosci 28, 13248–13257 (2008).
Han, Y. et al. AMPK signaling in the dorsal hippocampus negatively regulates contextual fear memory formation. Neuropsychopharmacology 41, 1849–1864 (2016).
Jian, M. et al. eIF2alpha dephosphorylation in basolateral amygdala mediates reconsolidation of drug memory. J Neurosci 34, 10010–10021 (2014).
Acknowledgements
This work was supported in part by the National Basic Research Program of China (no. 2015CB856400 and 2015CB553503) and National Natural Science Foundation of China (no. 81521063, 31230033, and 91432303).
Author information
Authors and Affiliations
Contributions
J.D., W.Y., Y.H., J.S. and L.L. conceived the project and designed the experiments. J.D., W.Y., Y.H., C.C., S.M., C.S., L.X., Y.X., and X.G. performed the experiments. J.D., W.Y., Y.H., N.C. S.M., F.Z., and Y.M. analyzed the data. W.Y. and J.D. wrote the paper. Y.H., J.S. and L.L revised the paper, with contributions from all of the other authors. We thank Dr. Wei-Li Zhu for comments on the experimental design.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Deng, JH., Yan, W., Han, Y. et al. Predictable Chronic Mild Stress during Adolescence Promotes Fear Memory Extinction in Adulthood. Sci Rep 7, 7857 (2017). https://doi.org/10.1038/s41598-017-08017-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-08017-7
This article is cited by
-
Behavioral Animal Models and Neural-Circuit Framework of Depressive Disorder
Neuroscience Bulletin (2024)
-
Persistent muscle hyperalgesia after adolescent stress is exacerbated by a mild-nociceptive input in adulthood and is associated with microglia activation
Scientific Reports (2022)
-
Pain sensitivity increases with sleep disturbance under predictable chronic mild stress in mice
Scientific Reports (2021)
-
Single neonatal dexamethasone administration has long-lasting outcome on depressive-like behaviour, Bdnf, Nt-3, p75ngfr and sorting receptors (SorCS1-3) stress reactive expression
Scientific Reports (2021)
-
Fasting enhances extinction retention and prevents the return of fear in humans
Translational Psychiatry (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.