Abstract
Lymphatic filariasis (LF) and onchocerciasis are priority neglected tropical diseases targeted for elimination. The only safe drug treatment with substantial curative activity against the filarial nematodes responsible for LF (Brugia malayi, Wuchereria bancrofti) or onchocerciasis (Onchocerca volvulus) is doxycycline. The target of doxycycline is the essential endosymbiont, Wolbachia. Four to six weeks doxycycline therapy achieves >90% depletion of Wolbachia in worm tissues leading to blockade of embryogenesis, adult sterility and premature death 18–24 months post-treatment. Long treatment length and contraindications in children and pregnancy are obstacles to implementing doxycycline as a public health strategy. Here we determine, via preclinical infection models of Brugia malayi or Onchocerca ochengi that elevated exposures of orally-administered rifampicin can lead to Wolbachia depletions from filariae more rapidly than those achieved by doxycycline. Dose escalation of rifampicin achieves >90% Wolbachia depletion in time periods of 7 days in B. malayi and 14 days in O. ochengi. Using pharmacokinetic-pharmacodynamic modelling and mouse-human bridging analysis, we conclude that clinically relevant dose elevations of rifampicin, which have recently been determined as safe in humans, could be administered as short courses to filariasis target populations with potential to reduce anti-Wolbachia curative therapy times to between one and two weeks.
Similar content being viewed by others
Introduction
Lymphatic Filariasis (LF) and onchocerciasis are vector-borne diseases that endure as public health problems despite the efforts of sustained elimination programmes1,2,3. LF remains wide-spread throughout the tropics, affecting 120 million individuals with 1.1 billion at risk of infection4. It is symptomatically characterised by lymphoedema, hydrocele and elephantiasis and is the second leading cause of global disability5. Onchocerciasis is endemic in much of Sub-Saharan Africa, as well as more limited foci in Brazil, Venezuela and The Yemen with 37 million infected6,7. Onchocerciasis is the cause of skin disease and, in its most severe presentation, a sclerosing ocular keratitis (river blindness) which affects 0.8 million individuals and is the second most prevalent cause of infection-related preventable blindness7,8,9. LF and onchocerciasis are both targeted for elimination as public health problems10,11. The current strategy of elimination is mass drug administration (MDA) with anti-filarial drugs, which target the transmissive first-stage larvae (microfilariae; mf) produced by mating adult filariae2,3,12 For LF, the standard treatment is once single annual treatment with diethylcarbamazine (DEC) and albendazole (ABZ) in Asia, South America and Polynesia13. Due to contraindications of DEC in areas of potential overlapping onchocerciasis distribution in Africa, DEC is contraindicated (due to risk of severe ocular adverse reactions)13,14 and ivermectin (IVM) is substituted in combination with ABZ15,16,17,18. IVM is also used as a monotherapy in once- or twice-annual MDA for the elimination of onchocerciasis19,20. Because standard anti-filarial drugs given in these dosages and/or combinations have little macrofilaricidal activity, they have to be administered with high population coverage and repetitively over many years in order to break the transmission cycle of the long-lived, reproductively active adult filarial infections. This is predicted as at least five annual rounds for LF and fifteen annual rounds for onchocerciasis2,3,16,17,20, with the former a minimum timeframe to initiate transmission assessment surveys.
MDA has undoubtedly both reduced the burden of onchocerciasis disease and achieved nationwide elimination outcomes for LF21,22,23,24,25,26,27,28 in certain country settings. In other countries, however, failures of the approach are manifest. ‘Hot-spot’ residual foci of infections persist despite sustained coverage due to emerging resistance or other factors29,30,31,32, in certain regions, poor adherence to treatment is recorded, in part due to incidence of severe neurological adverse reactions to Loa loa co-infections33,34 in hard-to-reach areas, inadequate coverage is apparent and certain country elimination programmes are yet to commence4,7 In these scenarios, if LF and onchocerciasis elimination targets within ambitious 2030 United Nations Sustainable Development Goal (SDG) timeframes are to be achieved, there is an urgent need to implement alternative strategies. Affordable registered drugs, which have evidence of curative activity against filariae, and could be re-purposed to cure target populations within a test-and-treat delivery, are considered the most expeditious solution toward achieving elimination targets where deployed to mop up residual foci during the elimination ‘end-game’.
The antibiotic, doxycycline, has been shown to indirectly exert significant macrofilaricidal activity by targeting the filarial endosymbiotic bacteria Wolbachia34,35. Through a series of clinical trials in LF36,37,38,39,40,41 and onchocerciasis patients42,43,44,45, sustained, >90% depletion of Wolbachia from filarial tissues consequently mediates inhibition of embryogenesis, infertility, clearance of mf from the blood or skin and ultimately death of the adult filariae in 18–24 months35,46. Effects of depleting Wolbachia within migratory mf have also been demonstrated to hinder development to the infectious larval stage in intermediate vectors47. Sustained amicrofilaraemia/amicrofilaridermia following a single course of doxycycline48,49 leads to symptomatic relief and halts disease progression in onchocerciasis and interrupts transmission in both LF and onchocerciasis. Dose regimen reductions during clinical studies and subsequent modelling of curative trial outcomes for onchocerciasis50 have defined that doxycycline achieves significant macrofilaricidal activity via >90% Wolbachia depletion in both LF48,49,51 and onchocerciasis42,44,52 after 4 to 6 weeks of daily administration, dependent on dose (100 or 200 mg) and the target organism (Wuchereria, Brugia or Onchocerca).
However, two major impediments remain for the implementation of an anti-Wolbachia therapeutic approach based on doxycycline. The long treatment duration of 4 weeks minimum imposes a logistical challenge and risk poor adherence, whereby shorter durations of doxycycline treatment, although achieving significant Wolbachia reductions and impacting on embryogenesis, fail to achieve significant curative outcomes53,54. Secondly, doxycycline is contraindicated in significant proportions of the population such as in pregnancy and children of 8 years of age or younger55.
In this work, a strategy for achieving the equivalent efficacy of long-course doxycycline treatment by using a high dose of rifampicin for 1–2 weeks is presented. For this, preclinical in vivo mouse models of Brugia malayi or Onchocerca ochengi adult worm infections56, are used to compare the activity of different orally administered dosages to establish a PK-PD relationship of rifampicin that is translatable to humans. Using these empirical rifampicin experiments and PK-PD analysis we provide preclinical evidence to justify phase II trials re-purposing high dose rifampicin to deliver curative outcomes in periods of between 1 to 2 weeks.
Results
Intrinsic anti-Wolbachia potency of rifampicin is superior to doxycycline
Table 1 shows in its first row, the in-vitro IC50 levels obtained for doxycycline and rifampicin in the Wolbachia infected C6/36 cell (C6/36 wAlbB) assay57. Rifampicin exhibited an EC50 of 1.3 nM, which was approximately 16.2 fold more potent than that of doxycycline (EC50 = 22 nM), against Wolbachia. Both drugs resulted in killing ~90% of Wolbachia in comparison to vehicle control cell cultures at the end of the 7 day experiment.
Pharmacokinetics of rifampicin are superior to doxycycline
Figure 1 shows the pharmacokinetic profiles measured in SCID mice at days 1 and 7 for doxycycline or rifampicin at a dose of 25 mg/kg bid. Table 2 summarises the pharmacokinetic parameters of each drug. Rifampicin (CL/F = 0.11 L/hr/kg) exhibited ~10 fold higher exposure at the same dose than doxycycline (CL/F = 1.2 L/h/kg). Clearance values at day 1 and day 7 were not statistically different (Mann-Whitney test, n = 5 mice, P > 0.05) and as shown in Table 2 all pharmacokinetics parameters of both drugs did not significantly change after chronic exposure in comparison to single dose administration.
Pharmacokinetic profiles. Systemic exposure to doxycycline (DOX) and rifampicin (RIF) in BALB/c SCID mice are shown as follows. (a) PK profile of DOX after single 25 mg/kg dose. (b) PK profile of DOX at day 7 after 7 bi-daily 25 mg/kg doses. (c) PK profile of RIF after single 25 mg/kg dose. (d) PK profile of RIF at day 7 after 7 bi-daily 25 mg/kg doses.
Anti-Wolbachia activity of rifampicin is superior to doxycycline in adult filarial infection models
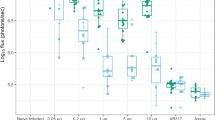
We compared the anti-Wolbachia activity of rifampicin (5 mg/kg qd, 15 mg/kg qd, 35 mg/kg qd or 25 mg/kg bid) with doxycycline (25 mg/kg bid) in B. malayi and O. ochengi SCID mouse models (Fig. 2). The murine doxycycline dose had been previously defined as bioequivalent to clinical dosing with 100 mg/day58. Wolbachia rate-of-kill was examined by four day dosing of mice infected with adult-stage B. malayi followed by termination +24 h after last dose and retrieval of female adults for assessment of Wolbachia loads (Fig. 2a). Changes in Wolbachia post-dosing were calculated as a % reduction compared with the median level in the vehicle control group. Bioequivalent doxycycline mediated a 41.3% reduction in Wolbachia after +4 day exposures. In comparison, low dose rifampicin (5 mg/kg qd) mediated a significantly higher 66.3% reduction over the same time period (1 way ANOVA F = 7.561, P = 0.002, Dunnett’s multiple comparisons test P < 0.05 vs DOX; Fig. 2b) whilst high dose rifampicin (15 mg/kg qd) mediated a 76.0% median reduction (P < 0.01 vs DOX; Fig. 2b).
Pharmacological Outputs: Anti-filarial, anti-Wolbachia pharmacological effects of doxycycline (DOX) and rifampicin (RIF) in vivo. immediate pharmacological effects of DOX and RIF on Brugia malayi female adult Wolbachia loads (a,b) or post-washout (c,d). Effects on B. malayi microfilariae (Bmmf) production (e). Effects on Onchocerca ochengi male adult Wolbachia loads post-washout (f,g). Box and whiskers represent min-max (n = 10–29; B. malayi Wolbachia, n = 4–25; Bmmf, n = 5–17 O. ochengi Wolbachia). Solid line = control median Wolbachia level, long dashed line = 90%, short dashed line = 99% depletion level. Significance indicated *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Elevations in oral rifampicin dose were undertaken to evaluate whether >90% threshold level Wolbachia depletions in female filarial tissues could be achieved in a time frame of seven days dosing. Six to seven week-old B. malayi timed infections were targeted with an additional washout period of five weeks to allow for continued depletion or recrudescence of the Wolbachia population to be evident, as previously reported59,60) (Fig. 2c). Doxycycline, at human bioequivalent (100 mg/day) dose58, was administered for four or six weeks (the latter a clinical dose time frame proven to deliver >90% Wolbachia depletion in B. malayi39 to compare with the efficacy of increasing dosages of short-course rifampicin regimens. As expected for ‘slow-killing’ anti-Wolbachia macrofilaricides, there were no significant changes in B. malayi worm burdens in either doxycycline or rifampicin dosed mouse groups compared with vehicle controls (Table 3).
Doxycycline mediated an 88.1% median reduction in Wolbachia after four week dosing and 99.76% median reduction after six week dosing. In comparison, low dose rifampicin (5 mg/kg qd x 7d) was sub-optimal and statistically inferior to doxycycline, with a 58.3% median reduction (Kruskal-Wallis test P < 0.0001, Dunn’s multiple comparisons test, P < 0.0001 vs +42d DOX; Fig. 2d). Elevation of rifampicin dose to 15 mg/kg qd, 35 mg/kg qd or 25 mg/kg bid delivered 97.7%, 98.2% or 99.5% median Wolbachia reductions, respectively. These dose effects were statistically non-inferior to six-week doxycycline dosing. Rifampicin 25 mg/kg bid dosing for seven days displayed significantly superior anti-Wolbachia activity to four-week doxycycline (P < 0.0001) and 15 mg/kg qd or 25 mg/kg bid rifampicin were superior to 5 mg/kg rifampicin after seven day dosing (P < 0.05 and P < 0.0001, respectively; Fig. 2d). Examining the impact of anti-Wolbachia efficacy on mf release, four or six-week doxycycline mediated a complete absence of viable mf within the peritoneal infection site (5/5 or 4/4 mice mf negative vs 2/25 mf negative vehicle; Kruskal-Wallis test P < 0.0001, Dunn’s multiple comparisons test, P < 0.01 or P < 0.05 vs vehicle; Fig. 2f). Low-dose rifampicin (5 mg/kg qd) did not significantly affect the peritoneal B. malayi mf burden compared with vehicle control levels (0/4 mice mf negative; Fig. 2e). In comparison, seven day dosing with 15 mg/kg qd rifampicin mediated a near complete absence of viable mf from the peritoneum of infected mice (11/12 mice mf negative; P < 0.0001 vs vehicle; Fig. 2e). Seven-day doses of rifampicin exceeding 15 mg/kg qd mediated total absence of viable mf (35 mg/kg qd: 4/4 mice mf negative P < 0.05 vs vehicle, 25 mg/kg bid: 9/9 mice mf negative P < 0.001 vs vehicle; Fig. 2e).
Based on threshold effects of elevated dose rifampicin achieving >90% Wolbachia depletions from Brugia adult female tissues following seven day dosing, we tested efficacy against Onchocerca male worms in a novel SCID mouse model of onchocerciasis56 Rifampicin dosed at 35 mg/kg qd for seven or 14 days was compared with human bioequivalent doxycycline dosed for 28 days (a minimum clinical dose/duration determined to deplete >90% Wolbachia from adult O. volvulus tissues leading to macrofilaricidal activity50, (Fig. 2f). Male Onchocerca worm burdens were not affected by treatment compared with vehicle controls (Table 3). Bioequivalent doxycycline mediated 99.6% median reduction in Wolbachia from male Onchocerca tissues following 28 days dosing (Fig. 2g). Seven-day dosing of rifampicin did not mediate any depletion in Wolbachia following washout period (0% median reduction) and was thus statistically inferior to doxycycline control (Kruskal-Wallis test P = 0.0013, Dunn’s multiple comparisons test, P < 0.001 vs DOX; Fig. 2g). In comparison, 14 day dosing of rifampicin mediated 96.9% median reductions in Onchocerca Wolbachia, a level of efficacy that was statistically non-inferior to doxycycline dosed for 28 days (Fig. 2g).
PK-PD modelling
The empirical PK and pharmacological anti-Wolbachia efficacy measurements of doxycycline and rifampicin were used to construct a PK-PD model (described in the methods section). An in vivo IC50 value was calculated based on the model for rifampicin (65.2 µg/ml), which was ~16-fold more potent than that calculated for doxycycline (1059 µg/ml; Table 1). IC50 values derived from the PK-PD model were 100,000 to 200,000 fold higher than those observed in vitro; however the ratio of calculated in vivo IC50 between rifampicin and doxycycline was aligned to the ratio empirically measured in vitro between the two drugs (~17-fold higher RIF vs DOX, Table 1).
Mouse – Human Bridging analysis
To compare rifampicin exposures observed in the SCID mouse model in comparison to those achieved in standard human clinical dose (600 mg), the overall exposure of rifampicin has been estimated in humans based on literature data (Fig. 3a). From this we determined the exposure in an average population of 70 kg humans after receiving 600 mg dose of rifampicin is significantly lower than the optimal exposure in mice that results in >90% Wolbachia elimination in 7 days or less (this exposure is defined as the 24 hour steady state AUC of the 15 mg/kg rifampicin dose in mice which is 141.5 mg/hr/L). The human dose that would achieve this optimal mouse exposure was then determined via multiple Monte-Carlo simulations (3000 subjects per dose) at different doses from 5 mg/kg up to 45 mg/kg where the percentage of humans predicted to achieve the desired AUC at each dose has been recorded (Fig. 3b). The PK predictions of higher doses of Rifampicin agree with published results where the predicted Cmax and AUC were equivalent to observed PK in humans at elevated doses in clinical studies61. This would indicate that our dosing suggestion will achieve the desired exposure based on clinical PK data.
Human-Mouse Bridging. PK profile of rifampicin in mouse at 15MKD (Black solid line) in comparison to exposures expected in man based on 600 mg dosing in 70 kg individuals (a) (green solid line representing median prediction and dashed red lines representing 5% and 95% percentiles of the prediction). % Probability of achieving optimal rifampicin exposure in humans at a dosing range of 4 mg/kg–45 mg/kg (b). (optimal exposure is defined as the median total 24 hour steady state AUC in mice receiving daily 15 mg/kg dose).
From those simulations, it is predicted that a minimum dose of 30–40 mg/kg is needed to achieve ‘curative’ exposure levels in human filariasis patients which also agrees with high dose RIF clinical studies61 that have shown a dose of 30 mg/kg or higher to achieve a steady-state AUC of 141.5 mg/hr/L in the vast majority of patients
Discussion
Targeting Wolbachia with antibiotics to deliver curative outcomes for filariasis is a promising drug development strategy. The pharmacopeia of registered antibiotics present ‘low hanging fruit’ with the potential for rapid re-purposing62,63. Further, the ‘slow kill’ effect post-Wolbachia depletion, manifest first on circulating or skin mf and then adult worms, avoids the problems of inflammatory adverse reactions which are associated with fast-killing direct filaricidal agents3,33,64,65,66. Indeed, this promotes anti-Wolbachia therapies as a safe drug development strategy for to administer to L. loa co-endemic target populations67 who are at risk of severe neurological adverse reactions to rapid acting filaricides and thus provides a potential solution to problem areas of Central Africa toward an end game of onchocerciasis elimination68. Clinical trials with doxycycline have shown that depletion of Wolbachia from filarial nematodes can deliver significant macrofilaricidal activity when administered for 4 or more weeks38,40,41,42,51. However, the long period of administration and contraindications in children and pregnant women present logistical challenges to widespread scale-up in resource poor settings of Sub-Saharan Africa. Whilst we have recently identified minocycline as a superior anti-Wolbachia agent to doxycycline in the B. malayi SCID mouse model58 as part of the same tetracycline class, the same contraindications would apply. The ‘holy grail’ for an anti-Wolbachia macrofilaricide is thus demonstrable activity in administration times of a minimum of 2 weeks (ideally 1 week) and a safety profile for administration in loiasis co-infections, in children and in pregnant women34,63.
We use here a validated screening system of in vitro cell assays and in vivo preclinical mouse models of filariasis56,57 to test the efficacy of dose elevations of rifampicin and have identified a dose-dependent activity which has the potential to achieve Wolbachia depletions predictive of cure when administered for 1 week against the human lymphatic filaria B. malayi or 2 weeks against O. ochengi, the closest phylogenetic relative of O. volvulus.
We determine that rifampicin is >15 fold more potent than doxycycline in vivo. Rifampicin exhibited significantly higher exposure per mg dose when compared to doxycycline. Also PK-PD modelling showed that rifampicin is more potent in reducing Wolbachia in vivo when exposure levels are matched to doxycycline. This superiority likely reflects the inherently higher (>16 fold) intrinsic potency of rifampicin against Wolbachia that is observed in vitro compared with doxycycline.
Corroborating rifampicin superiority versus doxycycline as an anti-Wolbachia agent, it has previously been reported, in mice experimentally infected with the rodent filaria, Litomosoides sigmodontis, that 25 mg/kg bid dosages of rifampicin result in a more profound decrease in Wolbachia levels in adult worms when compared to a similar dose of doxycycline48. Also, in a pilot study using the related, registered rifamycin, rifapentine, >90% depletion of Wolbachia was measured in O. ochengi males implanted into SCID mice, following two weeks 25 mg/kg bid dosing56. However, clinical studies in humans have shown that rifampicin administered for two or four weeks has inferior activity to six-week doxycycline in onchocerciasis69,70 and neither is combining rifampicin and doxycycline for two weeks sufficient to achieve significant macrofilaricidal activity54.
Our PK-PD model offers a rational explanation for the discrepancy in the activity of rifampicin in mice and humans, where doxycycline activity is similar across mouse model and human clinical studies, yet rifampicin seems to be dramatically more active in the mouse than in humans. We identify effective dosages in the mouse exceed the exposure of rifampicin achieved in humans when the drug is administered at ‘standard dose’ (~10 mg/kg qd or 600 mg). Indeed, exploring dose ranges more in line with standard human dose exposures, (5 mg/kg qd) rifampicin in the mouse is insufficient to achieve >90% Wolbachia depletion in 1 week against B. malayi.
The currently established 600 mg dose of rifampicin has not been predicated on PK-PD analysis but rather has been chosen based on drug cost, due to rifampicin semi-synthetic production in the 1970 s when the dosage was established71. However, recently the cost of rifampicin production has dramatically decreased and dose escalation studies have been initiated for TB patients. Higher doses of rifampicin (up to 35 mg/kg) have recently been proven to be safe in a clinical study61. The same study undertook human PK analysis and has shown that rifampicin exposure increases as the dose is increased. From this data we predict that the target exposure level required to deplete >90% Wolbachia from target filarial species in 1–2 weeks to ultimately deliver short-course curative efficacy against filariasis is achievable in humans when a dose of 30–35 mg/kg is administered. Importantly, clinical data supports that for doses of up to 35 mg/kg, given for periods over 2 weeks, no serious side effects are observed and that grade 1 and grade 2 side effects are randomly distributed across low and higher doses indicating no increased risk of side effects with elevated doses of rifampicin. A recent additional study where a dose of 35 mg/kg of RIF was administered to TB patients has also reported similar conclusions in relation to safety72.
Because elevated rifampicin dosages for filariasis cure are predicted in a 1–2 week dose exposure time frame, this is unlikely to result in resistant strains of M tuberculosis (TB), as there is no evidence for rifampicin resistance occurrence when administered for short periods of time73,74.
Rifampicin has a number of drug-drug interactions of clinical significance that have been previously reported75. Those interactions can result in the reduction of exposure of other drugs that are taken concomitantly with Rifampicin, and doses might need to be altered, especially for patients who are on HIV medication.
Conclusions
Using PK-PD modelling of drug activity in preclinical filarial mouse models, we show that a clinically safe dose of rifampicin can be administered for 1–2 week short courses to elicit >90% reductions in Wolbachia, predictive of the macrofilaricidal activity observed when Wolbachia is targeted by doxycycline, administered for 4–6 weeks. This treatment regimen would be compatible for use in children and during pregnancy and, because of the shortened duration of administration required, would be more readily deliverable by health care systems in resource-poor community settings. A 35 mg/kg dose has been proven as safe over a period of 2 weeks, and it has previously been shown that such short durations of treatment will not produce resistant strains of M. tuberculosis. We recommend that both a 35 mg/kg 1 and 2 week dose of rifampicin be urgently examined in lymphatic filariasis and onchocerciasis randomised controlled clinical trials to test whether such short-course treatment regimens are indeed sufficient to elicit equivalent curative activities to long course doxycycline.
Methods
Mice
Six to ten week old male BALB/c SCID (Harlan Laboratories, UK) or male CB.17 BALB/c SCID mice (Charles River, UK) were used for experiments. Mice were kept at the Biomedical Services Unit (BSU) at the University of Liverpool, UK, in specific pathogen-free (SPF) conditions or at The Research Foundation for Tropical Medicine and The Environment, Buea, Cameroon, in individually-ventilated caging. All experiments on animals were approved by The Animal Welfare and Ethical Review Board, Liverpool School of Tropical Medicine, UK, or the University of Buea Animal Ethics Review Board, Cameroon. Studies were conducted in accordance with Home Office legislation (UK) and matching welfare standards were applied in Cameroon.
In vitro potency studies
The anti-Wolbachia potency of doxycycline and rifampicin for use in the PK-PD model was determined in vitro, utilising the routine A·WOL screening assay as described previously57. In brief the mosquito (Aedes albopictus) derived cell line C6/36 (ATCC number CRL-1660), stably infected with Wolbachia pipientis (wAlbB) (C6/36 wAlbB) was incubated with the relevant drugs in a concentration range in order to determine a dose response. The drugs were incubated for 7 days with 2,000 cells per well on a 384 well plate (CellCarrier-384 Ultra, PerkinElmer) in Leibovitz media (Life Technologies™) supplemented with 20% foetal bovine serum (FBS, Fisher Scientific), 2% tryptose phosphate broth (Sigma-Aldrich) and 1% non-essential amino acids (Sigma-Aldrich). The end-point read out utilised DNA staining of both the host cell nuclei and intracellular Wolbachia (SYTO® 11) combined with a high content imaging system (Operetta®, PerkinElmer) and analysed using the associated Harmony® software through a cytoplasm texture analysis.
Brugia malayi experimental infections
The Brugia malayi lifecycle was maintained through mosquitoes and susceptible Meriones gerbils at LSTM. To generate infective B. malayi larvae for infections, female adult Aedes aegypti mosquitoes were fed with microfilariae collected from infected gerbils by catheterisation, as described previously76, that were mixed with human blood. Mosquitoes were fed through an artificial membrane feeder (Hemotek®). Blood-fed mosquitoes were reared for 14 days to allow for development to the L3 stage. L3 were then collected from infected mosquitoes by crushing and concentrated using a Baermann’s apparatus and RPMI medium. 100 motile L3 were collected and then injected into mice via the intra-peritoneal route. Efficiency of inoculations was confirmed by needle washout.
Onchocerca ochengi surgical implantations
Viable male Onchocerca ochengi were aseptically isolated from naturally parasitized cattle as described previously56. Between 10–11 male Onchocerca were surgically implanted into the peritoneal cavity of CB.17(BALB/c) SCID mice under anaesthesia as described previously56.
Drug treatments
Six weeks after B. malayi experimental infection or 3 days post O. ochengi surgical implantation, SCID mice received 100 µL compound via oral gavage at variable doses and for variable treatment lengths as described and indicated in further detail in the main text. The drug doses and routes used were: Doxycycline (25 mg/kg qd or bid po and rifampicin (1.25 mg/kg–25 mg/kg qd or bid po). Doxycycline was dissolved in water, while rifampicin was dissolved in 55% polyethylene glycol 300; 25% propylene glycol; 20% water. All drugs and vehicle reagents were purchased from Sigma Aldrich.
Parasitological readouts
Seven or twelve weeks after B. malayi experimental infection or 38 days after O. ochengi surgical implantation, mice were necropsied. Adult filariae and released B. malayi mf were recovered by a combination of peritoneal washings using wash media (RPMI containing P/S) and subsequent dissection of abdominal cavities. To quantify B. malayi mf, samples were centrifuged at 1200 rpm for 5 min. Excess supernatants were removed and the remaining volume quantified. Three 20 µL volumes were then enumerated for mf by microscopy, scoring as motile or stretched immotile. Adult filariae were observed for motility, washed in cold PBS and parasite stages enumerated. A minimum of ten B. malayi females and five O. ochengi males per treatment group of 3–5 mice were collected into individual Eppendorf tubes to be processed for qPCR.
DNA extraction and PCR quantification of Wolbachia
DNA was extracted from worm samples using a DNeasy Blood and Tissue Kit (Qiagen) according to manufacturer’s instructions. Levels of Bm Wolbachia surface protein (wsp) gene copy numbers were quantified using qPCR as previously described77. Levels of Onchocerca Wolbachia wsp and filarial gst gene copy numbers were similarly quantified as previously described56.
PK Studies
To establish the pharmacokinetic profiles of rifampicin and doxycycline, rich pharmacokinetic studies were performed in uninfected male SCID mice (weight 24–28 g). Doxycycline (25 mg/kg bid) or rifampicin (5 mg/kg qd or 25 mg/kg bid) were administered orally for up to seven days. For all PK studies, a total of 11 samples were collected at discrete time points between 0 and 24 hour post dose at days 1 and 7). Serial blood samples were collected at days 1 and 7 of dosing via the tail vein where a microincision was performed and 20 µL of blood collected using a pipette with a pre-heparinised tip. Blood samples were directly lysed with 40 µL of ice cold ddH2O and then frozen at −80 °C until time of bioanalysis.
Bioanalysis
Rifampicin concentrations were determined by LCMS (liquid chromatography mass spectrometry) using an appropriate internal standard and were validated to internationally recognised acceptance criteria [FDA-guidelines]. Chromatographic separation was achieved using a gradient programme. The UHPLC (ultrahigh pressure liquid chromatography) system was interfaced with a triple-quadruple TSQ Quantum Access mass spectrometer (Thermo Scientific, Hemel Hempstead, UK) with a heated-electrospray ionization (H-ESI) source. An E2M28 rotary vacuum pump (Edwards High Vacuum International, West Sussex, UK), an NM30LA nitrogen generator (Peak Scientific, Renfrewshire, UK) and 99% pure argon gas (10 L, BIP10, Air Products, Liverpool, UK) were used. Blood samples (20 μl) containing rifampicin were extracted with a mixture of acetonitrile and methanol (80:20 v/v). The subsequent mix was then filtered through 96 well filter plate (Millipore, UK). The resultant filtrate was then transferred to a 96 deep well plate containing 40 µl of (10 mg/ml) ascorbic acid, 10 µl of this resultant mixture was then injected into the LC-MS/MS. The assay was linear in the range 10–10,000 ng/ml for rifampicin.
PK-PD Modelling
A PK-PD model as described in equations 1 and 2 has been developed and used to fit parasitological read-outs from experiments in infected mice. The pharmacological output for all PK/PD analyses was Wolbachia counts as estimated in qPCR experiments in the quantitation of wsp DNA levels. Pmetrics® 78 was used in modelling PK/PD data in all experiments. The following differential equations were used:
For the PK component:
Where C p is the drug concentration in blood at any given time, F is the fraction available for oral absorption, ka is the rate of absorption from gut to blood, V is the volume of distribution and kel is the rate of elimination from the systemic circulation.
For the PD component, a relationship was built to describe the effect of the drug upon Wolbachia levels using a dynamic E max model:
X 1 is the observed Wolbachia levels as estimated by qPCR, kgrowth is the rate of bacterial growth which was set at 0 due to the absence of any evidence of bacterial growth beyond the time of treatment and no evidence of recrudescence within the time frame of the experiment. Kkill is the maximum possible rate of kill of bacteria and has been fixed to the in vitro kill rate that could be achieved at the maximum concentration of drug. IC50 is the blood concentration required to achieve 50% of the maximal kill rate of Wolbachia in the adult worm in the mouse.
Statistics
PK parameters between doxycycline and rifampicin were compared by Mann-Whitney test. Percentage Wolbachia reductions in filariae following in vivo treatments were calculated from median vehicle control levels derived from the same experimental infection and screen. Where repeat experiment data (Wolbachia loads normalised to control, worm enumerations or viable mf enumerations per mouse) were available, data was pooled. Grouped continuous variables were tested for normal distribution by D’Agostino & Pearson omnibus normality tests. Continuous variables failing normal distribution tests were Log10 transformed and re-tested. Continuous variables satisfying the assumptions of normal distribution were examined by 1 way ANOVA with Dunnett’s multiple tests post-hoc. Variables not satisfying the assumption of normality were compared by Kruskal-Wallis Test with Dunn’s Multiple Tests, post-hoc. Significance levels are indicated P < 0.05*, P < 0.01** P < 0.001*** P < 0.0001****. All statistics were undertaken using GraphPad Prism v6 software.
Change history
18 January 2018
A correction to this article has been published and is linked from the HTML version of this paper. The error has been fixed in the paper.
References
WHO. Meeting of the International Task Force for Disease Eradication, January 2014. Releve epidemiologique hebdomadaire/Section d’hygiene du Secretariat de la Societe des Nations=Weekly epidemiological record/Health Section of the Secretariat of the League of Nations 89, 153–160 (2014).
Bockarie, M. J., Kelly-Hope, L. A., Rebollo, M. & Molyneux, D. H. Preventive chemotherapy as a strategy for elimination of neglected tropical parasitic diseases: endgame challenges. Philosophical Transactions of the Royal Society of London B: Biological Sciences 368, 20120144 (2013).
Taylor, M. J., Hoerauf, A. & Bockarie, M. Lymphatic filariasis and onchocerciasis. Lancet 376, 1175–1185, https://doi.org/10.1016/S0140-6736(10)60586-7 (2010).
WHO. Lymphatic Filariasis Factsheet, <http://www.who.int/mediacentre/factsheets/fs102/en/.> (2016).
Molyneux, D. H. & Zagaria, N. Lymphatic filariasis elimination: progress in global programme development. Annals of tropical medicine and parasitology 96(Suppl 2), S15–40 (2002).
[APOC], A. P. f. O. C. In Joint Action Forum (JAF) of APOC (Paris, France, 2005).
WHO. Onchocerciasis Factsheet No 374 (2016).
WHO. Onchocerciasis and its control: Report of a WHO Expert Committee on Onchocerciasis Control (1995).
WHO. African Programme for Onchocerciasis Control - report of the sixth meeting of National Task Forces, October 2009. Releve epidemiologique hebdomadaire/Section d’hygiene du Secretariat de la Societe des Nations = Weekly epidemiological record/Health Section of the Secretariat of the League of Nations 85, 23–28 (2010).
WHO. Global Plan to Combat Neglected Tropical Diseases 2008–2017, <http://apps.who.int/iris/bitstream/10665/69708/1/WHO_CDS_NTD_2007.3_eng.pdf> (2007).
WHO. Accelerating Work to Overcome the Global Impact of Neglected Tropical Diseases: A Roadmap for Implementation, <http://www.who.int/neglected_diseases/NTD_RoadMap_2012_Fullversion.pdf> (2012).
Molyneux, D. H., Bradley, M., Hoerauf, A., Kyelem, D. & Taylor, M. J. Mass drug treatment for lymphatic filariasis and onchocerciasis. Trends in parasitology 19, 516–522 (2003).
Ottesen, E., Duke, B., Karam, M. & Behbehani, K. Strategies and tools for the control/elimination of lymphatic filariasis. Bulletin of the world Health Organization 75, 491 (1997).
Kale, O. O. Onchocerciasis: the burden of disease. Annals of tropical medicine and parasitology 92(Suppl 1), S101–115 (1998).
Ichimori, K. et al. Global programme to eliminate lymphatic filariasis: the processes underlying programme success. PLoS Negl Trop Dis 8, e3328 (2014).
Bockarie, M. J. & Deb, R. M. Elimination of lymphatic filariasis: do we have the drugs to complete the job? Current opinion in infectious diseases 23, 617–620, https://doi.org/10.1097/QCO.0b013e32833fdee5 (2010).
Rebollo, M. P. & Bockarie, M. J. Toward the elimination of lymphatic filariasis by 2020: treatment update and impact assessment for the endgame. Expert review of anti-infective therapy 11, 723–731 (2013).
Gyapong, J. O., Kumaraswami, V., Biswas, G. & Ottesen, E. A. Treatment strategies underpinning the global programme to eliminate lymphatic filariasis. Expert opinion on pharmacotherapy 6, 179–200 (2005).
Coffeng, L. E. et al. African Programme For Onchocerciasis Control 1995–2015: model-estimated health impact and cost. PLoS Negl Trop Dis 7, e2032, https://doi.org/10.1371/journal.pntd.0002032 (2013).
Basáñez, M.-G. et al. River blindness: a success story under threat? PLoS Med 3, e371 (2006).
Sodahlon, Y. K. et al. A success story: Togo is moving toward becoming the first sub-Saharan African nation to eliminate lymphatic filariasis through mass drug administration and countrywide morbidity alleviation. PLoS Negl Trop Dis 7, e2080 (2013).
De-jian, S., Xu-li, D. & Ji-hui, D. The history of the elimination of lymphatic filariasis in China. Infectious diseases of poverty 2, 1 (2013).
WHO. Expert Mission to Sri Lanka for verification of elimination of lymphatic filariasis. (World Health Organisation, New Delhi, India, 2012).
Cheun, H. I. et al. Successful control of lymphatic filariasis in the Republic of Korea. The Korean journal of parasitology 47, 323–335, https://doi.org/10.3347/kjp.2009.47.4.323 (2009).
WHO. Progress toward eliminating onchocerciasis in the WHO Region of the Americas: verification of elimination of transmission in Mexico. Releve epidemiologique hebdomadaire/Section d’hygiene du Secretariat de la Societe des Nations = Weekly epidemiological record/Health Section of the Secretariat of the League of Nations 90, 577–581 (2015).
WHO. Elimination of onchocerciasis in the WHO Region of the Americas: Ecuador’s progress towards verification of elimination. Releve epidemiologique hebdomadaire/Section d’hygiene du Secretariat de la Societe des Nations = Weekly epidemiological record/Health Section of the Secretariat of the League of Nations 89, 401–405 (2014).
WHO. Progress towards eliminating onchocerciasis in the WHO Region of the Americas: verification by WHO of elimination of transmission in Colombia. Releve epidemiologique hebdomadaire/Section d’hygiene du Secretariat de la Societe des Nations = Weekly epidemiological record/Health Section of the Secretariat of the League of Nations 88, 381–385 (2013).
Diawara, L. et al. Feasibility of onchocerciasis elimination with ivermectin treatment in endemic foci in Africa: first evidence from studies in Mali and Senegal. PLoS Negl Trop Dis 3, e497, https://doi.org/10.1371/journal.pntd.0000497 (2009).
Bourguinat, C. et al. Genetic selection of low fertile Onchocerca volvulus by ivermectin treatment. PLoS Negl Trop Dis 1, e72, https://doi.org/10.1371/journal.pntd.0000072 (2007).
Osei-Atweneboana, M. Y. et al. Phenotypic evidence of emerging ivermectin resistance in Onchocerca volvulus. PLoS Negl Trop Dis 5, e998 (2011).
Osei-Atweneboana, M. Y., Boakye, D. A., Awadzi, K., Gyapong, J. O. & Prichard, R. K. Genotypic analysis of β-tubulin in Onchocerca volvulus from communities and individuals showing poor parasitological response to ivermectin treatment. International Journal for Parasitology: Drugs and Drug Resistance 2, 20–28 (2012).
Taylor, M. J. et al. Onchocerciasis Control: Vision for the Future from a Ghanian perspective. Parasites & vectors 2, 1 (2009).
Gardon, J., Gardon-Wendel, N., Kamgno, J., Chippaux, J.-P. & Boussinesq, M. Serious reactions after mass treatment of onchocerciasis with ivermectin in an area endemic for Loa loa infection. The Lancet 350, 18–22 (1997).
Taylor, M. J., Hoerauf, A., Townson, S., Slatko, B. E. & Ward, S. A. Anti-Wolbachia drug discovery and development: safe macrofilaricides for onchocerciasis and lymphatic filariasis. Parasitology 141, 119–127 (2014).
Taylor, M. J. & Hoerauf, A. A new approach to the treatment of filariasis. Current opinion in infectious diseases 14, 727–731 (2001).
Taylor, M. J. et al. Macrofilaricidal activity after doxycycline treatment of Wuchereria bancrofti: a double-blind, randomised placebo-controlled trial. The Lancet 365, 2116–2121 (2005).
Debrah, A. Y. et al. Doxycycline reduces plasma VEGF-C/sVEGFR-3 and improves pathology in lymphatic filariasis. PLoS Pathog 2, e92 (2006).
Debrah, A. Y. et al. Macrofilaricidal effect of 4 weeks of treatment with doxycycline on Wuchereria bancrofti. Tropical Medicine & International Health 12, 1433–1441 (2007).
Supali, T. et al. Doxycycline treatment of Brugia malayi-infected persons reduces microfilaremia and adverse reactions after diethylcarbamazine and albendazole treatment. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America 46, 1385–1393, https://doi.org/10.1086/586753 (2008).
Mand, S. et al. Macrofilaricidal activity and amelioration of lymphatic pathology in bancroftian filariasis after 3 weeks of doxycycline followed by single-dose diethylcarbamazine. The American journal of tropical medicine and hygiene 81, 702–711 (2009).
Gayen, P. et al. A double-blind controlled field trial of doxycycline and albendazole in combination for the treatment of bancroftian filariasis in India. Acta tropica 125, 150–156 (2013).
Hoerauf, A. et al. Wolbachia endobacteria depletion by doxycycline as antifilarial therapy has macrofilaricidal activity in onchocerciasis: a randomized placebo-controlled study. Medical microbiology and immunology 197, 295–311, https://doi.org/10.1007/s00430-007-0062-1 (2008).
Hoerauf, A. et al. Efficacy of 5-week doxycycline treatment on adult Onchocerca volvulus. Parasitology research 104, 437–447, https://doi.org/10.1007/s00436-008-1217-8 (2009).
Turner, J. D. et al. Macrofilaricidal activity after doxycycline only treatment of Onchocerca volvulus in an area of Loa loa co-endemicity: a randomized controlled trial. PLoS Negl Trop Dis 4, e660, https://doi.org/10.1371/journal.pntd.0000660 (2010).
Debrah, A. Y. et al. Doxycycline leads to sterility and enhanced killing of female Onchocerca volvulus worms in an area with persistent microfilaridermia after repeated ivermectin treatment–a randomized placebo controlled double-blind trial. Clinical Infectious Diseases civ363 (2015).
Hoerauf, A., Adjei, O. & Buttner, D. W. Antibiotics for the treatment of onchocerciasis and other filarial infections. Current opinion in investigational drugs 3, 533–537 (2002).
Albers, A. et al. Retarded Onchocerca volvulus L1 to L3 larval development in the Simulium damnosum vector after anti-wolbachial treatment of the human host. Parasites & vectors 5, 1 (2012).
Volkmann, L., Fischer, K., Taylor, M. & Hoerauf, A. Antibiotic therapy in murine filariasis (Litomosoides sigmodontis): comparative effects of doxycycline and rifampicin on Wolbachia and filarial viability. Tropical medicine & international health: TM & IH 8, 392–401 (2003).
Hoerauf, A. et al. Doxycycline as a novel strategy against bancroftian filariasis-depletion of Wolbachia endosymbionts from Wuchereria bancrofti and stop of microfilaria production. Medical microbiology and immunology 192, 211–216, https://doi.org/10.1007/s00430-002-0174-6 (2003).
Walker, M. et al. Therapeutic efficacy and macrofilaricidal activity of doxycycline for the treatment of river blindness. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America 60, 1199–1207, https://doi.org/10.1093/cid/ciu1152 (2015).
Taylor, M. J. et al. Macrofilaricidal activity after doxycycline treatment of Wuchereria bancrofti: a double-blind, randomised placebo-controlled trial. Lancet 365, 2116–2121, https://doi.org/10.1016/S0140-6736(05)66591-9 (2005).
Hoerauf, A., Mand, S., Adjei, O., Fleischer, B. & Buttner, D. W. Depletion of wolbachia endobacteria in Onchocerca volvulus by doxycycline and microfilaridermia after ivermectin treatment. Lancet 357, 1415–1416, https://doi.org/10.1016/S0140-6736(00)04581-5 (2001).
Turner, J. D. et al. A randomized, double-blind clinical trial of a 3-week course of doxycycline plus albendazole and ivermectin for the treatment of Wuchereria bancrofti infection. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America 42, 1081–1089, https://doi.org/10.1086/501351 (2006).
Debrah, A. Y. et al. Macrofilaricidal Activity in Wuchereria bancrofti after 2 Weeks Treatment with a Combination of Rifampicin plus Doxycycline. Journal of parasitology research 2011, 201617, https://doi.org/10.1155/2011/201617 (2011).
Czeizel, A. E. & Rockenbauer, M. Teratogenic study of doxycycline. Obstetrics and gynecology 89, 524–528, https://doi.org/10.1016/S0029-7844(97)00005-7 (1997).
Halliday, A. et al. A murine macrofilaricide pre-clinical screening model for onchocerciasis and lymphatic filariasis. Parasit Vectors 7, 472, https://doi.org/10.1186/s13071-014-0472-z (2014).
Clare, R. H. et al. Development and validation of a high-throughput anti-Wolbachia whole-cell screen: a route to macrofilaricidal drugs against onchocerciasis and lymphatic filariasis. Journal of biomolecular screening 20, 64–69, https://doi.org/10.1177/1087057114551518 (2015).
Sharma, R. et al. Minocycline as a re-purposed anti-Wolbachia macrofilaricide: superiority compared with doxycycline regimens in a murine infection model of human lymphatic filariasis. Scientific reports 6 (2016).
Gilbert, J. et al. Antibiotic chemotherapy of onchocerciasis: in a bovine model, killing of adult parasites requires a sustained depletion of endosymbiotic bacteria (Wolbachia species). Journal of Infectious Diseases 192, 1483–1493 (2005).
Makepeace, B. L., Rodgers, L. & Trees, A. J. Rate of elimination of Wolbachia pipientis by doxycycline in vitro increases following drug withdrawal. Antimicrobial agents and chemotherapy 50, 922–927 (2006).
Boeree, M. J. et al. A dose-ranging trial to optimize the dose of rifampin in the treatment of tuberculosis. American journal of respiratory and critical care medicine 191, 1058–1065, https://doi.org/10.1164/rccm.201407-1264OC (2015).
Johnston, K. L., Ford, L. & Taylor, M. J. Overcoming the challenges of drug discovery for neglected tropical diseases: the A.WOL experience. Journal of biomolecular screening 19, 335–343, https://doi.org/10.1177/1087057113511270 (2014).
Johnston, K. L. et al. Repurposing of approved drugs from the human pharmacopoeia to target Wolbachia endosymbionts of onchocerciasis and lymphatic filariasis. International journal for parasitology. Drugs and drug resistance 4, 278–286, https://doi.org/10.1016/j.ijpddr.2014.09.001 (2014).
Boatin, B. A. & Richards, F. O. Control of onchocerciasis. Advances in parasitology 61, 349–394 (2006).
Kamgno, J., Gardon, J., Gardon-Wendel, N., Duke, B. O. & Boussinesq, M. Adverse systemic reactions to treatment of onchocerciasis with ivermectin at normal and high doses given annually or three-monthly. Transactions of the Royal Society of Tropical Medicine and Hygiene 98, 496–504 (2004).
Hoerauf, A., Pfarr, K., Mand, S., Debrah, A. & Specht, S. Filariasis in Africa—treatment challenges and prospects. Clinical Microbiology and Infection 17, 977–985 (2011).
McGarry, H. F. et al. Evidence against Wolbachia symbiosis in Loa loa. Filaria journal 2, 9, https://doi.org/10.1186/1475-2883-2-9 (2003).
Wanji, S. et al. Relationship between oral declaration on adherence to ivermectin treatment and parasitological indicators of onchocerciasis in an area of persistent transmission despite a decade of mass drug administration in Cameroon. Parasit Vectors 8, 667, https://doi.org/10.1186/s13071-015-1283-6 (2015).
Specht, S. et al. Efficacy of 2- and 4-week rifampicin treatment on the Wolbachia of Onchocerca volvulus. Parasitology research 103, 1303–1309, https://doi.org/10.1007/s00436-008-1133-y (2008).
Richards, F. O. Jr. et al. No depletion of Wolbachia from Onchocerca volvulus after a short course of rifampin and/or azithromycin. Am J Trop Med Hyg 77, 878–882 (2007).
van Ingen, J. et al. Why Do We Use 600 mg of Rifampicin in Tuberculosis Treatment? Clinical infectious diseases: an official publication of the Infectious Diseases Society of America 52, e194–199, https://doi.org/10.1093/cid/cir184 (2011).
Boeree, M. J. et al. High-dose rifampicin, moxifloxacin, and SQ109 for treating tuberculosis: a multi-arm, multi-stage randomised controlled trial. The Lancet Infectious Diseases. https://doi.org/10.1016/S1473-3099(16)30274-2 (2016).
Acocella, G., Brumfitt, W. & Hamilton-Miller, J. M. Evidence that rifampicin can be used safely for non-tuberculous diseases. Thorax 35, 788–791 (1980).
Gruneberg, R. N., Emmerson, A. M. & Cremer, A. W. Rifampicin for non-tuberculous infections? Chemotherapy 31, 324–328 (1985).
Baciewicz, A. M., Chrisman, C. R., Finch, C. K. & Self, T. H. Update on rifampin, rifabutin, and rifapentine drug interactions. Current medical research and opinion 29, 1–12, https://doi.org/10.1185/03007995.2012.747952 (2013).
McGarry, H. F., Plant, L. D. & Taylor, M. J. Diethylcarbamazine activity against Brugia malayi microfilariae is dependent on inducible nitric-oxide synthase and the cyclooxygenase pathway. Filaria journal 4, 4, https://doi.org/10.1186/1475-2883-4-4 (2005).
McGarry, H. F., Egerton, G. L. & Taylor, M. J. Population dynamics of Wolbachia bacterial endosymbionts in Brugia malayi. Molecular and biochemical parasitology 135, 57–67 (2004).
Neely, M. N., van Guilder, M. G., Yamada, W. M., Schumitzky, A. & Jelliffe, R. W. Accurate detection of outliers and subpopulations with Pmetrics, a nonparametric and parametric pharmacometric modeling and simulation package for R. Therapeutic drug monitoring 34, 467–476, https://doi.org/10.1097/FTD.0b013e31825c4ba6 (2012).
Acknowledgements
The A·WOL consortium is supported by a grant from the Bill and Melinda Gates Foundation awarded to the Liverpool School of Tropical Medicine (BMGF OPP1054324). This work was also supported by a BMGF Grand Challenges Explorations grant (OPP1119043) to JDT, SAW and MJT and a Medical Research Council New Investigator Research Grant (MR/L018756/1) to JDT.
Author information
Authors and Affiliations
Contributions
G.A., L.F., J.D.T., S.A.W. and M.J.T. wrote the paper. G.A., L.F., J.D.T., S.A.W. and M.J.T. conceived the work. G.A. and R.S. performed the PK/PD modelling analysis. H.E.T., G.A., J.G., A.G., J.D., D.W., L.F. H.S. N.P. H.M. S.W. and J.D.T. performed or directed the preclinical in vivo work (PK or PD). H.E.T., G.A., J.G., L.H., A.G., J.D., D.W., D.A.N.C. H.S. and J.D.T. performed analysis to obtain quantitative biological readouts. D.W. performed the PK bioanalysis. A.C., R.H.C., L.M., L.H., L.F. and K.L.J. performed or directed the in vitro work. A.S. and D.A.N.C. maintained the parasite lifecycle.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Change History: A correction to this article has been published and is linked from the HTML version of this paper. The error has been fixed in the paper.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A correction to this article is available online at https://doi.org/10.1038/s41598-018-19723-1.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Aljayyoussi, G., Tyrer, H.E., Ford, L. et al. Short-Course, High-Dose Rifampicin Achieves Wolbachia Depletion Predictive of Curative Outcomes in Preclinical Models of Lymphatic Filariasis and Onchocerciasis. Sci Rep 7, 210 (2017). https://doi.org/10.1038/s41598-017-00322-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-00322-5
This article is cited by
-
The pipeline for drugs for control and elimination of neglected tropical diseases: 2. Oral anti-infective drugs and drug combinations for off-label use
Parasites & Vectors (2023)
-
Establishment of Wolbachia infection in Aedes aegypti from Pakistan via embryonic microinjection and semi-field evaluation of general fitness of resultant mosquito population
Parasites & Vectors (2022)
-
The Eagle effect in the Wolbachia-worm symbiosis
Parasites & Vectors (2021)
-
In silico drug repurposing for filarial infection predicts nilotinib and paritaprevir as potential inhibitors of the Wolbachia 5′-aminolevulinic acid synthase
Scientific Reports (2021)
-
Onchocerciasis drug development: from preclinical models to humans
Parasitology Research (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.