Abstract
Oriental melon (Cucumis melo L. var. makuwa) is one of the most important cultivated cucurbits, and is grown widely in Northeast Asian countries. With increasing interest in its biological properties and economic importance, oriental melon has become an attractive model crop for studying various horticultural traits. A previous genome sequence of the melon was constructed from a homozygous double-haploid line. Thus, individual reference genomes are required to perform functional studies and further breeding applications. Here, we report draft genome sequences of two oriental melons, Chang Bougi and SW3. The assembled 344 Mb genome of Chang Bougi was obtained with scaffold N50 1.0 Mb, and 36,235 genes were annotated. The 354 Mb genome of SW3 was assembled with scaffold N50 1.6 Mb, and has 38,173 genes. These newly constructed genomes will enable studies of fruit development, disease resistance, and breeding applications in the oriental melon.
Measurement(s) | DNA |
Technology Type(s) | DNA sequencing |
Factor Type(s) | reference genome |
Sample Characteristic - Organism | Cucumis melo |
Machine-accessible metadata file describing the reported data: https://doi.org/10.6084/m9.figshare.9939629
Similar content being viewed by others
Background & Summary
The oriental melon (Cucumis melo L. var. makuwa), one of the most important annual diploid crops within the Cucurbitaceae family, is grown largely in Northeast Asian countries, including Korea, China, and Japan. It is cultivated primarily for its fruit, which generally has a sweet aromatic flavor and contains soluble sugars, organic acids, minerals, and vitamins1,2,3. Traits of the fruit, such as shape, skin color, flesh color, and sugar content, are highly variable. Because its economic importance and interest in its biological properties have increased, oriental melon has become an attractive model crop for the study of various traits.
Reference genomes from genetically diverse individuals provide insights into genome structures, genome evolution, and diversification within the genus and species. For instance, precise comparison of genome structures and analyses about lineage-specific evolution of gene families in the genus Capsicum became possible through the completion of multiple reference genomes4. In the case of melon, a previous reference genome was constructed from the homozygous DHL92 double-haploid line5, and subsequent improvements to the genome assembly and annotations were reported6. To carry out functional studies, evolutionary studies of gene families, link genetic markers to desirable traits, and further breeding applications in the oriental melon, multiple reference genomes will be required.
Here, we report the construction of draft genomes of two oriental melon types, Chang Bougi and SW3. Chang Bougi, a Korean landrace, is a new source for the breeding of resistance to Cucumber Green Mottle Mosaic Virus (CGMMV), which causes mosaicism in leaves and deterioration of fruits, leading to severe yield and quality losses of cucurbit crops worldwide7. The high-quality breeding line SW3, from NongWoo Bio Company, contains deep-yellow and oval-type fruits with high sugar content.
Figure 1 presents an overview of the study. A combination of paired-end (PE) and mate-pair (MP) libraries were sequenced to generate 231× and 345× of genomic sequencing data8, respectively, for Chang Bougi and SW3 (Table 1). Genome assembly and annotation were then performed (Fig. 1). The assembled genome of Chang Bougi9 comprised 11,309 scaffolds totaling 344 Mb in length, with scaffold N50 of 1.0 Mb. For SW3, 7,202 scaffolds totaling 354 Mb in length were assembled10, with scaffold N50 of 1.6 Mb (Table 2). Repeat annotation was then carried out (Table 3). K-mer frequencies were calculated to provide information related to low frequencies, sequencing depth, level of heterozygosity, and genome size (Fig. 2)11. The estimated genome sizes of Chang Bougi and SW3 were 355 Mb and 373 Mb, respectively, which were similar to previously reported genome sizes5. A total of 36,235 and 38,173 genes were determined as final genes in Chang Bougi and SW3, respectively (Table 2 and Fig. 3). Then functional annotation of final gene models were performed (Table 4 and Fig. 4). Finally, we provide new reference genome of oriental melons for further analysis and breeding program.
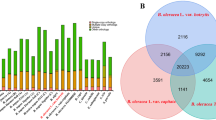
Comparisons of gene models for two oriental melon genomes and other genomes. (a) Gene length distribution (b) CDS length distribution (c) Exon number distribution (d) Intron length distribution (e) Intron number distribution. x-axis stands for length (bp) of gene (a), CDS (b) and intron (d) or numbers of exon (c) and intron (e), respectively. y-axis stands for ratio of genes.
Venn diagram of the number of genes having functional annotation in Chang Bougi and SW3 genomes using multiple public databases. Functional annotation of Chang Bougi (a) and SW3 (b) were primarily performed using Blast2Go. For genes that remained unassigned by Blast2Go, we used NR, GO, KEGG, and InterPro to assign gene function.
Methods
DNA extraction and sequencing
Leaves of two oriental melons were harvested and frozen immediately in liquid nitrogen. Genomic DNA was extracted, and paired-end and mate-pair libraries for next-generation sequencing were constructed according to the manufacturer’s instructions (Illumina, San Diego, CA, USA). The quality of each library was validated using the KAPA SYBR FAST Universal 2× qPCR Master Mix (Kapa Biosystems, Boston, MA, USA). Each library was sequenced with the Illumina HiSeq 2500 platform.
Genome assembly
Pre-processing analyses of raw sequences, using in-house pipeline and genome assembly, were performed as described in previous studies4,12. After pre-processing to remove erroneous sequences in raw data, remaining sequences in paired-end libraries were assembled using Platanus13, with parameters for Chang Bougi (-k 63 -c 5 -d 0.3 -t 40 -m 220) and for SW3 (-k 91 -c 5 -d 0.3 -t 44 -m 200). The scaffolding process was performed with Platanus, using paired-end and mate-pair sequences, with parameters for Chang Bougi (-l 3 -s 61 -u 0.2 -t 40), and for SW3 (-l 3 -u 0.2 -t 15). Remaining gaps were filled with Platanus and GapCloser version 1.10 (http://soap.genomics.org.cn/down/GapCloser_release_2011.tar.gz), using reads from the paired-end and mate-pair libraries. Finally, 344 Mb of Chang Bougi genomic sequence (96.9% of 355 Mb) and 354 Mb of SW3 genomic sequence (94.9% of 373 Mb) were assembled (Table 2).
Repeat annotation
After construction of repeat libraries using the assembled Chang Bougi and SW3 genomes, repeat annotation was implemented using RepeatModeler and RepeatMasker (http://www.repeatmasker.org). A total of 179 Mb (52% of 355 Mb) and 194 Mb (54% of 373 Mb) of repeat sequences were detected in Chang Bougi and SW3, respectively (Table 3).
Genome annotation
Annotation of the two genomes were performed using the KOBIC annotation pipeline (a modified PGA pipeline14), consisting of repeat masking, mapping of different protein sequence sets, and ab initio prediction performed by AUGUSTUS v3.2.215. Transcript assembly was performed with the assembled genome by a reference-based algorithm using HISAT216 and StringTie17. To generate protein-based gene models for consensus modeling, the protein sequences of Arabidopsis thaliana (TAIR10, http://www.arabidopsis.org), Citrullus lanatus18, Cucumis melo5, and Cucumis sativus19 were mapped using GeneWise v2.120. AUGUSTUS was used for gene prediction in the two oriental melon genomes. To validate the predicted gene models, protein sequences from the genomes of C. lanatus, C. melo, C. sativus, and A. thaliana were used as queries in BLASTp, and erratic gene models were filtered with a BLASTp cut-off of query coverage ≥0.3. Also, the assembled transcripts were validated against the same four sets of protein sequences using tLBASTn, and filtered with cut-off values of query coverage ≥0.5 and subject coverage ≥0.3. The GeneWise gene models that remained were reformatted from GeneWise format to GFF3 data, and used to determine the consensus gene model via EVM21, which combines ab initio gene predictions with protein alignments into weighted-consensus gene structures (ab initio predictions = 1, protein alignment = 5, transcript alignment assemblies = 7). Ultimately, the final gene models included a total of 36,235 consensus genes for Chang Bougi and 38,173 consensus genes for SW3 (Table 2 and Fig. 3).
Further functional annotations were performed using the program Blast2Go22, including InterPro23, NR from NCBI, Kyoto Encyclopedia of Genes and Genomes (KEGG)24. Functional annotation of the final gene models (Table 4 and Fig. 4) predicted 2,093, 3,703, and 493 genes as hypothetical protein, uncharacterized protein, and unknown function, respectively, in the Chang Bougi genome. In the SW3 genome, respectively 2,245, 3,827, and 570 genes were predicted as hypothetical protein, uncharacterized protein, and unknown function.
Data Records
All of the raw sequence reads produced by Illumina HiSeq 2500 have been deposited at NCBI Sequence Read Archive (SRA) under BioProject number PRJNA531526 (accession SRP191487)8 and BioSample from SAMN11368505 to SAMN11368524 (SAMN11368505 ~ SAMN11368515 for Chang Bougi; SAMN11368516 ~ SAMN11368524 for SW3). The Whole Genome Shotgun project of Chang Bougi have been deposited at DDBJ/ENA/GenBank under the accession number SSTD000000009 under PRJNA531576 and SAMN11370205. The Whole Genome Shotgun project of SW3 have been deposited at DDBJ/ENA/GenBank under the accession number SSTE0000000010 under BioProject number PRJNA531478 and BioSample SAMN11381272.
Technical Validation
Detection and filtration of misannotated genes
EvidenceModeler predicted 39,977 and 42,535 consensus genes for Chang Bougi and SW3, respectively. We investigated these to detect misannotated genes, as recommended by NCBI GenBank, including genes containing internal stop codons, genes lacking a stop codon, frame-shifted genes, or erroneous start codons. A total of 3,742 and 4,362 misannotated genes were detected and masked in Chang Bougi and in SW3, respectively. Thus, 36,235 genes remained in the Chang Bougi genome, and 38,173 genes remained in SW3.
Evaluation of genome annotation using BUSCO
BUSCO v3.0.225 provides an assessment of assembled genome completeness based on the orthologous group, with single-copy genes from OrthoDB (http://www.orthodb.org), and using a hidden Markov model to profile amino acid alignments. For genome annotation assessments, we used 1,440 gene sets of orthologs conserved in embryophyta (Table 5). The results showed that nearly all of these core genes/orthologs were present in the genomes of Chang Bougi (85.28%) and SW3 (86.81%).
Comparison of gene sets in the genomes of oriental melons Chang Bougi and SW3 with those in the genomes of melon (DHL92 v3.6.1) and cucumber
To compare gene sets between oriental melons and previously reported cucurbit genomes, orthologous and paralogous genes were detected in melon genome (DHL 92 v3.6.1), Chang Bougi, SW3, and cucumber (C. sativus) using the program OrthoFinder26. A total of 113,006 sequences were clustered into 30,738 groups, with 3,475 and 4,469 singleton genes detected in Chang Bougi and in SW3, respectively (Fig. 5). Fewer singleton genes might be expected in the two oriental melons than in the melon genome, which was constructed from a homozygous DHL92 double-haploid line, derived from a cross between Korean landraces of oriental melon (Songwhan Chamoe, PI 161375) and melon (Piel de Sapo). In addition, 2,213 genes were determined as common among melon and the two oriental melons, and 12,983 genes were detected in all four genomes. Functional investigation of singleton genes of Chang Bougi and SW3 indicated that 869 and 1,112 of genes were functionally unknown genes, respectively.
Code availability
The sequence data were generated using software provided by the sequencing platform manufacturer, and were processed with publicly available software and recommended settings, as cited in this report. No custom computer codes were generated in this work.
References
Nunez-Palenius, H. G. et al. Melon fruits: genetic diversity, physiology, and biotechnology features. Crit. Rev. Biotechnol. 28, 13–55, https://doi.org/10.1080/07388550801891111 (2008).
Burger, Y. et al. Genetic variability for valuable fruit quality traits in Cucumis melo. Israel Journal of Plant Sciences 54, 233–242, https://doi.org/10.1560/IJPS_54_3_233 (2006).
Fernández-Trujillo, J. P., Picó, B., Garcia-Mas, J., Álvarez, J. M. & Monforte, A. J. In Breeding for Fruit Quality. 261–278 (John Wiley & Sons, Inc., 2011).
Kim, S. et al. New reference genome sequences of hot pepper reveal the massive evolution of plant disease-resistance genes by retroduplication. Genome Biol. 18, 210, https://doi.org/10.1186/s13059-017-1341-9 (2017).
Garcia-Mas, J. et al. The genome of melon (Cucumis melo L.). Proc. Natl. Acad. Sci. USA 109, 11872–11877, https://doi.org/10.1073/pnas.1205415109 (2012).
Ruggieri, V. et al. An improved assembly and annotation of the melon (Cucumis melo L.) reference genome. Scientific reports 8, 8088, https://doi.org/10.1038/s41598-018-26416-2 (2018).
Sugiyama, M., Ohara, T. & Sakata, Y. Inheritance of Resistance to Cucumber Green Mottle Mosaic Virus in Cucumis melo L. Chang Bougi. Journal of the Japanese Society for Horticultural Science 76, 316–318, https://doi.org/10.2503/jjshs.76.316 (2007).
NCBI Sequence Read Archive, https://identifiers.org/ncbi/insdc.sra:SRP191487 (2019).
Kwon, S.-Y. Cucumis melo var. makuwa cultivar Chang Bougi, whole genome shotgun sequencing project. GenBank, https://identifiers.org/ncbi/insdc:SSTD00000000 (2019).
Kwon, S.-Y. Cucumis melo var. makuwa cultivar SW3, whole genome shotgun sequencing project. GenBank, https://identifiers.org/ncbi/insdc:SSTE00000000 (2019).
Marcais, G. & Kingsford, C. A fast, lock-free approach for efficient parallel counting of occurrences of k-mers. Bioinformatics 27, 764–770, https://doi.org/10.1093/bioinformatics/btr011 (2011).
Kim, Y. M. et al. Genome analysis of Hibiscus syriacus provides insights of polyploidization and indeterminate flowering in woody plants. DNA Res. 24, 71–80, https://doi.org/10.1093/dnares/dsw049 (2017).
Kajitani, R. et al. Efficient de novo assembly of highly heterozygous genomes from whole-genome shotgun short reads. Genome research 24, 1384–1395, https://doi.org/10.1101/gr.170720.113 (2014).
Kim, S. et al. Genome sequence of the hot pepper provides insights into the evolution of pungency in Capsicum species. Nat. Genet. 46, 270–278, https://doi.org/10.1038/ng.2877 (2014).
Stanke, M., Diekhans, M., Baertsch, R. & Haussler, D. Using native and syntenically mapped cDNA alignments to improve de novo gene finding. Bioinformatics 24, 637–644, https://doi.org/10.1093/bioinformatics/btn013 (2008).
Kim, D., Langmead, B. & Salzberg, S. L. HISAT: a fast spliced aligner with low memory requirements. Nature methods 12, 357–360, https://doi.org/10.1038/nmeth.3317 (2015).
Pertea, M. et al. StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat. Biotechnol. 33, 290–295, https://doi.org/10.1038/nbt.3122 (2015).
Tomato Genome, C. The tomato genome sequence provides insights into fleshy fruit evolution. Nature 485, 635–641, https://doi.org/10.1038/nature11119 (2012).
Huang, S. et al. The genome of the cucumber, Cucumis sativus L. Nat. Genet. 41, 1275–1281, https://doi.org/10.1038/ng.475 (2009).
Birney, E., Clamp, M. & Durbin, R. GeneWise and Genomewise. Genome research 14, 988–995, https://doi.org/10.1101/gr.1865504 (2004).
Haas, B. J. et al. Automated eukaryotic gene structure annotation using EVidenceModeler and the Program to Assemble Spliced Alignments. Genome Biol. 9, R7, https://doi.org/10.1186/gb-2008-9-1-r7 (2008).
Conesa, A. & Gotz, S. Blast2GO: A comprehensive suite for functional analysis in plant genomics. Int. J. Plant Genomics 2008, 619832, https://doi.org/10.1155/2008/619832 (2008).
Mitchell, A. L. et al. InterPro in 2019: improving coverage, classification and access to protein sequence annotations. Nucleic Acids Res. 47, D351–D360, https://doi.org/10.1093/nar/gky1100 (2019).
Kanehisa, M. et al. Data, information, knowledge and principle: back to metabolism in KEGG. Nucleic Acids Res. 42, D199–205, https://doi.org/10.1093/nar/gkt1076 (2014).
Kriventseva, E. V., Zdobnov, E. M., Simão, F. A., Ioannidis, P. & Waterhouse, R. M. BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 31, 3210–3212, https://doi.org/10.1093/bioinformatics/btv351 (2015).
Emms, D. M. & Kelly, S. OrthoFinder: solving fundamental biases in whole genome comparisons dramatically improves orthogroup inference accuracy. Genome Biol. 16, 157, https://doi.org/10.1186/s13059-015-0721-2 (2015).
Acknowledgements
This work was supported by grants from the Agricultural Genome Center of the Next Generation Biogreen 21 Program (Project No. PJ011088 and PJ013297) of the Rural Development Administration, Korea, and the Korean Research Institute of Bioscience and Biotechnology Initiative Program.
Author information
Authors and Affiliations
Contributions
Y.-M.K. and S.-Y.K. conceived and designed the project and organized the manuscript. A.-Y.S., N.K. and S.K. generated data, performed analyses, and managed subprojects. A.-Y.S. prepared DNA and RNA samples. S.K. and D.C. performed de novo genome assembly. N.K., Y.M.S. and Y.-M.K. performed genome annotations. A.-Y.S., N.K., S.K., D.C., Y.-M.K. and S.-Y.K. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
The Creative Commons Public Domain Dedication waiver http://creativecommons.org/publicdomain/zero/1.0/ applies to the metadata files associated with this article.
About this article
Cite this article
Shin, AY., Koo, N., Kim, S. et al. Draft genome sequences of two oriental melons, Cucumis melo L. var. makuwa. Sci Data 6, 220 (2019). https://doi.org/10.1038/s41597-019-0244-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41597-019-0244-x