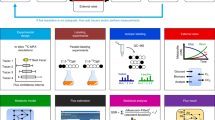
Abstract
Genome-scale stoichiometric modeling of metabolism has become a standard systems biology tool for modeling cellular physiology and growth. Extensions of this approach are emerging as a valuable avenue for predicting, understanding and designing microbial communities. Computation of microbial ecosystems in time and space (COMETS) extends dynamic flux balance analysis to generate simulations of multiple microbial species in molecularly complex and spatially structured environments. Here we describe how to best use and apply the most recent version of COMETS, which incorporates a more accurate biophysical model of microbial biomass expansion upon growth, evolutionary dynamics and extracellular enzyme activity modules. In addition to a command-line option, COMETS includes user-friendly Python and MATLAB interfaces compatible with the well-established COBRA models and methods, as well as comprehensive documentation and tutorials. This protocol provides a detailed guideline for installing, testing and applying COMETS to different scenarios, generating simulations that take from a few minutes to several days to run, with broad applicability to microbial communities across biomes and scales.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Data availability
The COMETS Protocols GitHub repository (https://github.com/segrelab/COMETS_Protocols) contains all input files and jupyter notebooks from which one can reproduce the results presented in this protocol. The data are distributed under the Creative Commons CC0 1.0 Universal license.
Code availability
COMETS (https://www.runcomets.org) is an open-source code, and it is available at https://github.com/segrelab/comets. The code is distributed under the GNU General Public License Version 3. The documentation is available at https://segrelab.github.io/comets-manual/, which is structured as a tutorial and contains additional examples not shown in this protocol. The MATLAB toolbox is available at https://github.com/segrelab/comets-toolbox, distributed under the GNU General Public License Version 3. The COMETS Python toolbox is available at https://github.com/segrelab/cometspy, distributed under the GNU General Public License Version 3. The code in this protocol has been peer-reviewed.
References
Shou, W., Ram, S. & Vilar, J. M. G. Synthetic cooperation in engineered yeast populations. Proc. Natl Acad. Sci. USA 104, 1877–1882 (2007).
Vorholt, J. A., Vogel, C., Carlström, C. I. & Müller, D. B. Establishing causality: opportunities of synthetic communities for plant microbiome research. Cell Host Microbe 22, 142–155 (2017).
Kehe, J. et al. Massively parallel screening of synthetic microbial communities. Proc. Natl Acad. Sci. USA 116, 12804–12809 (2019).
Venturelli, O. S. et al. Deciphering microbial interactions in synthetic human gut microbiome communities. Mol. Syst. Biol. 14, e8157 (2018).
Johns, N. I., Blazejewski, T., Gomes, A. L. & Wang, H. H. Principles for designing synthetic microbial communities. Curr. Opin. Microbiol. 31, 146–153 (2016).
Grosskopf, T. & Soyer, O. S. Synthetic microbial communities. Curr. Opin. Microbiol. 18, 72–77 (2014).
Thompson, L. R. et al. A communal catalogue reveals Earth’s multiscale microbial diversity. Nature 551, 457–463 (2017).
Andreote, F. D. & de Cássia Pereira e Silva, M. Microbial communities associated with plants: learning from nature to apply it in agriculture. Curr. Opin. Microbiol. 37, 29–34 (2017).
Moran, M. A. The global ocean microbiome. Science 350, aac8455 (2015).
Integrative HMP (iHMP) Research Network Consortium. The Integrative Human Microbiome Project. Nature 569, 641–648 (2019).
Friedman, J. & Gore, J. Ecological systems biology: the dynamics of interacting populations. Curr. Opin. Syst. Biol. 1, 114–121 (2017).
Foster, K. R., Schluter, J., Coyte, K. Z. & Rakoff-Nahoum, S. The evolution of the host microbiome as an ecosystem on a leash. Nature 548, 43–51 (2017).
Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature 486, 207–214 (2012).
Noronha, A. et al. The Virtual Metabolic Human database: integrating human and gut microbiome metabolism with nutrition and disease. Nucleic Acids Res. 47, D614–D624 (2019).
Lozupone, C. A., Stombaugh, J. I., Gordon, J. I., Jansson, J. K. & Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 489, 220–230 (2012).
Saifuddin, M., Bhatnagar, J. M., Segrè, D. & Finzi, A. C. Microbial carbon use efficiency predicted from genome-scale metabolic models. Nat. Commun. 10, 3568 (2019).
Gilbert, J. A., Jansson, J. K. & Knight, R. Earth Microbiome Project and Global Systems Biology. mSystems 3, e00217–17 (2018).
Ibarbalz, F. M. et al. Global trends in marine plankton diversity across kingdoms of life. Cell 179, 1084–1097.e21 (2019).
Ko, Y.-S. et al. Tools and strategies of systems metabolic engineering for the development of microbial cell factories for chemical production. Chem. Soc. Rev. 49, 4615–4636 (2020).
Keasling, J. D. Manufacturing molecules through metabolic engineering. Science 330, 1355–1358 (2010).
Pacheco, A. R., Moel, M. & Segrè, D. Costless metabolic secretions as drivers of interspecies interactions in microbial ecosystems. Nat. Commun. 10, 103 (2019).
Germerodt, S. et al. Pervasive selection for cooperative cross-feeding in bacterial communities. PLoS Comput. Biol. 12, e1004986 (2016).
Rakoff-Nahoum, S., Foster, K. R. & Comstock, L. E. The evolution of cooperation within the gut microbiota. Nature 533, 255–259 (2016).
Zelezniak, A. et al. Metabolic dependencies drive species co-occurrence in diverse microbial communities. Proc. Natl Acad. Sci. USA 112, 6449–6454 (2015).
Widder, S. et al. Challenges in microbial ecology: building predictive understanding of community function and dynamics. ISME J. 10, 2557–2568 (2016).
Goldford, J. E. et al. Emergent simplicity in microbial community assembly. Science 361, 469–474 (2018).
Magnúsdóttir, S. & Thiele, I. Modeling metabolism of the human gut microbiome. Curr. Opin. Biotechnol. 51, 90–96 (2018).
Harcombe, W. R. et al. Metabolic resource allocation in individual microbes determines ecosystem interactions and spatial dynamics. Cell Rep. 7, 1104–1115 (2014).
Gu, C., Kim, G. B., Kim, W. J., Kim, H. U. & Lee, S. Y. Current status and applications of genome-scale metabolic models. Genome Biol. 20, 121 (2019).
Bordbar, A., Monk, J. M., King, Z. A. & Palsson, B. O. Constraint-based models predict metabolic and associated cellular functions. Nat. Rev. Genet. 15, 107–120 (2014).
O’Brien, E. J., Monk, J. M. & Palsson, B. O. Using genome-scale models to predict biological capabilities. Cell 161, 971–987 (2015).
Orth, J. D., Thiele, I. & Palsson, B. Ø. What is flux balance analysis? Nat. Biotechnol. 28, 245–248 (2010).
Arkin, A. P. et al. KBase: The United States Department of Energy Systems Biology Knowledgebase. Nat. Biotechnol. 36, 566–569 (2018).
Karp, P. D. et al. The BioCyc collection of microbial genomes and metabolic pathways. Brief. Bioinform. 20, 1085–1093 (2019).
King, Z. A. et al. BiGG Models: a platform for integrating, standardizing and sharing genome-scale models. Nucleic Acids Res. 44, D515–D522 (2016).
Lieven, C. et al. MEMOTE for standardized genome-scale metabolic model testing. Nat. Biotechnol. 38, 272–276 (2020).
Machado, D., Andrejev, S., Tramontano, M. & Patil, K. R. Fast automated reconstruction of genome-scale metabolic models for microbial species and communities. Nucleic Acids Res. 46, 7542–7553 (2018).
Willi, G., Olivier Brett, G., Bruggeman Frank, J. & Bas, T. Constraint-based stoichiometric modelling from single organisms to microbial communities. J. R. Soc. Interface 13, 20160627 (2016).
Zomorrodi, A. R. & Maranas, C. D. OptCom: a multi-level optimization framework for the metabolic modeling and analysis of microbial communities. PLoS Comput. Biol. 8, e1002363 (2012).
Khandelwal, R. A., Olivier, B. G., Röling, W. F. M., Teusink, B. & Bruggeman, F. J. Community flux balance analysis for microbial consortia at balanced growth. PLoS One 8, e64567 (2013).
Stolyar, S. et al. Metabolic modeling of a mutualistic microbial community. Mol. Syst. Biol. 3, 92 (2007).
Chen, J. et al. Spatiotemporal modeling of microbial metabolism. BMC Syst. Biol. 10, 21 (2016).
Mahadevan, R., Edwards, J. S. & Doyle, F. J. 3rd Dynamic flux balance analysis of diauxic growth in Escherichia coli. Biophys. J. 83, 1331–1340 (2002).
Höffner, K., Harwood, S. M. & Barton, P. I. A reliable simulator for dynamic flux balance analysis. Biotechnol. Bioeng. 110, 792–802 (2013).
Henson, M. A. & Hanly, T. J. Dynamic flux balance analysis for synthetic microbial communities. IET Syst. Biol. 8, 214–229 (2014).
Zhuang, K. et al. Genome-scale dynamic modeling of the competition between Rhodoferax and Geobacter in anoxic subsurface environments. ISME J. 5, 305–316 (2011).
Tzamali, E., Poirazi, P., Tollis, I. G. & Reczko, M. A computational exploration of bacterial metabolic diversity identifying metabolic interactions and growth-efficient strain communities. BMC Syst. Biol. 5, 167 (2011).
Hanly, T. J. & Henson, M. A. Dynamic flux balance modeling of microbial co-cultures for efficient batch fermentation of glucose and xylose mixtures. Biotechnol. Bioeng. 108, 376–385 (2011).
Thiele, I. & Palsson, B. Ø. A protocol for generating a high-quality genome-scale metabolic reconstruction. Nat. Protoc. 5, 93–121 (2010).
Rocha, I., Förster, J. & Nielsen, J. Design and application of genome-scale reconstructed metabolic models. Methods Mol. Biol. 416, 409–431 (2008).
Nadell, C. D., Drescher, K. & Foster, K. R. Spatial structure, cooperation and competition in biofilms. Nat. Rev. Microbiol. 14, 589–600 (2016).
Chacón, J. M., Möbius, W. & Harcombe, W. R. The spatial and metabolic basis of colony size variation. ISME J. 12, 669–680 (2018).
Hynes, W. F. et al. Bioprinting microbial communities to examine interspecies interactions in time and space. Biomed. Phys. Eng. Express 4, 055010 (2018).
DiMucci, D., Kon, M. & Segrè, D. Machine learning reveals missing edges and putative interaction mechanisms in microbial ecosystem networks. mSystems 3, e00181–18 (2018).
Harcombe, W. R., Chacón, J. M., Adamowicz, E. M., Chubiz, L. M. & Marx, C. J. Evolution of bidirectional costly mutualism from byproduct consumption. Proc. Natl Acad. Sci. USA 115, 12000–12004 (2018).
Hammarlund, S. P., Chacón, J. M. & Harcombe, W. R. A shared limiting resource leads to competitive exclusion in a cross-feeding system. Environ. Microbiol. 21, 759–771 (2019).
Bajić, D., Vila, J. C. C., Blount, Z. D. & Sánchez, A. On the deformability of an empirical fitness landscape by microbial evolution. Proc. Natl Acad. Sci. USA 115, 11286–11291 (2018).
García-Jiménez, B., García, J. L. & Nogales, J. FLYCOP: metabolic modeling-based analysis and engineering microbial communities. Bioinformatics 34, i954–i963 (2018).
Yu, B. et al. Experiments and simulations on short chain fatty acid production in a colonic bacterial community. Preprint at bioRxiv https://doi.org/10.1101/444760 (2018)
Heirendt, L. et al. Creation and analysis of biochemical constraint-based models using the COBRA Toolbox v.3.0. Nat. Protoc. 14, 639–702 (2019).
Feist, A. M. & Palsson, B. O. The biomass objective function. Curr. Opin. Microbiol. 13, 344–349 (2010).
Ibarra, R. U., Edwards, J. S. & Palsson, B. O. Escherichia coli K-12 undergoes adaptive evolution to achieve in silico predicted optimal growth. Nature 420, 186–189 (2002).
Fong, S. S. & Palsson, B. Ø. Metabolic gene–deletion strains of Escherichia coli evolve to computationally predicted growth phenotypes. Nat. Genet. 36, 1056–1058 (2004).
Segrè, D., Vitkup, D. & Church, G. M. Analysis of optimality in natural and perturbed metabolic networks. Proc. Natl Acad. Sci. USA 99, 15112–15117 (2002).
Wintermute, E. H., Lieberman, T. D. & Silver, P. A. An objective function exploiting suboptimal solutions in metabolic networks. BMC Syst. Biol. 7, 98 (2013).
Lewis, N. E. et al. Omic data from evolved E. coli are consistent with computed optimal growth from genome-scale models. Mol. Syst. Biol. 6, 390 (2010).
Matsushita, M. et al. Interface growth and pattern formation in bacterial colonies. Phys. A Stat. Mech. Appl. 249, 517–524 (1998).
Farrell, F. D. C., Hallatschek, O., Marenduzzo, D. & Waclaw, B. Mechanically driven growth of quasi-two-dimensional microbial colonies. Phys. Rev. Lett. 111, 168101 (2013).
Tronnolone, H. et al. Diffusion-limited growth of microbial colonies. Sci. Rep. 8, 5992 (2018).
Lacasta, A. M., Cantalapiedra, I. R., Auguet, C. E., Peñaranda, A. & Ramírez-Piscina, L. Modeling of spatiotemporal patterns in bacterial colonies. Phys. Rev. E 59, 7036–7041 (1999).
Kozlovsky, Y., Cohen, I., Golding, I. & Ben-Jacob, E. Lubricating bacteria model for branching growth of bacterial colonies. Phys. Rev. E Stat. Phys. Plasmas Fluids Relat. Interdiscip. Topics 59, 7025–7035 (1999).
Giverso, C., Verani, M. & Ciarletta, P. Branching instability in expanding bacterial colonies. J. R. Soc. Interface 12, 20141290 (2015).
Henrichsen, J. Bacterial surface translocation: a survey and a classification. Bacteriol. Rev. 36, 478–503 (1972).
Vassallo, L., Hansmann, D. & Braunstein, L. A. On the growth of non-motile bacteria colonies: an agent-based model for pattern formation. Eur. Phys. J. B 92, 216 (2019).
Ben-Jacob, E. et al. Generic modelling of cooperative growth patterns in bacterial colonies. Nature 368, 46–49 (1994).
Dornic, I., Chaté, H. & Muñoz, M. A. Integration of Langevin equations with multiplicative noise and the viability of field theories for absorbing phase transitions. Phys. Rev. Lett. 94, 100601 (2005).
Press, W. H. Numerical Recipes in C: The Art of Scientific Computing 2nd edn (Cambridge University Press, 1992).
LeVeque, R. J. Finite Difference Methods for Ordinary and Partial Differential Equations: Steady-State and Time-Dependent Problems (SIAM, 2007).
Varma, A. & Palsson, B. O. Stoichiometric flux balance models quantitatively predict growth and metabolic by-product secretion in wild-type Escherichia coli W3110. Appl. Environ. Microbiol. 60, 3724–3731 (1994).
Orth, J. D., Palsson, B. Ø. & Fleming, R. M. T. Reconstruction and use of microbial metabolic networks: the core Escherichia coli metabolic model as an educational guide. EcoSal Plus 4 (2010).
Zeng, H. & Yang, A. Bridging substrate intake kinetics and bacterial growth phenotypes with flux balance analysis incorporating proteome allocation. Sci. Rep. 10, 4283 (2020).
Sauro, H. M. Enzyme Kinetics for Systems Biology (Ambrosius Publishing, 2012).
Ebrahim, A., Lerman, J. A., Palsson, B. O. & Hyduke, D. R. COBRApy: COnstraints-Based Reconstruction and Analysis for Python. BMC Syst. Biol. 7, 74 (2013).
Ofaim, S., Sulheim, S., Almaas, E., Sher, D. & Segrè, D. Dynamic allocation of carbon storage and nutrient-dependent exudation in a revised genome-scale model of Prochlorococcus. Front. Genet. 12, 586293 (2021).
Cezairliyan, B. & Ausubel, F. M. Investment in secreted enzymes during nutrient-limited growth is utility dependent. Proc. Natl Acad. Sci. USA 114, E7796–E7802 (2017).
Rakoff-Nahoum, S., Coyne, M. J. & Comstock, L. E. An ecological network of polysaccharide utilization among human intestinal symbionts. Curr. Biol. 24, 40–49 (2014).
van Zyl, W. H., Lynd, L. R., den Haan, R. & McBride, J. E. Consolidated bioprocessing for bioethanol production using Saccharomyces cerevisiae. Adv. Biochem. Eng. Biotechnol. 108, 205–235 (2007).
Traving, S. J., Thygesen, U. H., Riemann, L. & Stedmon, C. A. A model of extracellular enzymes in free-living microbes: which strategy pays off? Appl. Environ. Microbiol. 81, 7385–7393 (2015).
Huang, X.-F. et al. Rhizosphere interactions: root exudates, microbes, and microbial communities. Botany 92, 267–275 (2014).
Nunan, N. The microbial habitat in soil: scale, heterogeneity and functional consequences. J. Plant Nutr. Soil Sci. 180, 425–429 (2017).
Müller, J. & Van Saarloos, W. Morphological instability and dynamics of fronts in bacterial growth models with nonlinear diffusion. Phys. Rev. E Stat. Nonlin. Soft Matter Phys. 65, 061111 (2002).
Korolev, K. S., Avlund, M., Hallatschek, O. & Nelson, D. R. Genetic demixing and evolution in linear stepping stone models. Rev. Mod. Phys. 82, 1691–1718 (2010).
Covert, M. W., Schilling, C. H. & Palsson, B. Regulation of gene expression in flux balance models of metabolism. J. Theor. Biol. 213, 73–88 (2001).
Covert, M. W. & Palsson, B. O. Constraints-based models: regulation of gene expression reduces the steady-state solution space. J. Theoret. Biol. 221, 309–325 (2003).
Asenjo, J. A., Diaz, H., Cintolesi, A., Rapaport, I. & Andrews, B. A. Metabolomics of recombinant yeast: Gene expression, flux analysis and a mathematical model for gene regulation of metabolism. J. Biotechnol. 136, S19 (2008).
Thanamit, K., Hoerhold, F., Oswald, M. & Koenig, R. Gene expression profiles based flux balance model to predict the carbon source for Bacillus subtilis. Preprint at bioRxiv https://doi.org/10.1101/842518 (2020).
Goelzer, A., Fromion, V. & Scorletti, G. Cell design in bacteria as a convex optimization problem. Automatica 47, 1210–1218 (2011).
Mori, M., Hwa, T., Martin, O. C., De Martino, A. & Marinari, E. Constrained allocation flux balance analysis. PLoS Comput. Biol. 12, e1004913 (2016).
Becker, S. A. & Palsson, B. O. Context-specific metabolic networks are consistent with experiments. PLoS Comput. Biol. 4, e1000082 (2008).
Gutiérrez, M. et al. A new improved and extended version of the multicell bacterial simulator gro. ACS Synth. Biol. 6, 1496–1508 (2017).
Bauer, E., Zimmermann, J., Baldini, F., Thiele, I. & Kaleta, C. BacArena: individual-based metabolic modeling of heterogeneous microbes in complex communities. PLoS Comput. Biol. 13, e1005544 (2017).
Shade, A. et al. Fundamentals of microbial community resistance and resilience. Front. Microbiol. 3, 417 (2012).
Allison, S. D. & Martiny, J. B. H. Resistance, resilience, and redundancy in microbial communities. Proc. Natl Acad. Sci. USA 105, 11512–11519 (2008).
Pacheco, A. R., Osborne, M. L. & Segrè, D. Non-additive microbial community responses to environmental complexity. Nat. Commun. 12, 2365 (2021).
Pacheco, A. R. & Segrè, D. Pacheco A. R. An evolutionary algorithm for designing microbial communities via environmental modification. J. R. Soc. Interface 18, 20210348 (2021).
Bricaud, A., Claustre, H., Ras, J. & Oubelkheir, K. Natural variability of phytoplanktonic absorption in oceanic waters: influence of the size structure of algal populations. J. Geophys. Res. 109, C11010 (2004).
Partensky, F., Hoepffner, N., Li, W., Ulloa, O. & Vaulot, D. Photoacclimation of Prochlorococcus sp. (Prochlorophyta) strains isolated from the North Atlantic and the Mediterranean Sea. Plant Physiol. 101, 285–296 (1993).
Casey, J. R., Mardinoglu, A., Nielsen, J. & Karl, D. M. Adaptive evolution of phosphorus metabolism in Prochlorococcus. mSystems 1, e00065–16 (2016).
Stramski, D., Bricaud, A. & Morel, A. Modeling the inherent optical properties of the ocean based on the detailed composition of the planktonic community. Appl. Opt. 40, 2929–2945 (2001).
Morel, A., Ahn, Y.-H., Partensky, F., Vaulot, D. & Claustre, H. Prochlorococcus and Synechococcus: a comparative study of their optical properties in relation to their size and pigmentation. J. Marine Res. 51, 617–649 (1993).
Pope, R. M. & Fry, E. S. Absorption spectrum (380–700 nm) of pure water II Integrating cavity measurements. Appl. Optics 36, 8710 (1997).
Morel, A. & Bricaud, A. Theoretical results concerning light absorption in a discrete medium, and application to specific absorption of phytoplankton. Deep Sea Res. A 28, 1375–1393 (1981).
Zomorrodi, A. R. & Maranas, C. D. Improving the iMM904 S. cerevisiae metabolic model using essentiality and synthetic lethality data. BMC Syst. Biol. 4, 178 (2010).
Biggs, M. B. & Papin, J. A. Novel multiscale modeling tool applied to Pseudomonas aeruginosa biofilm formation. PLoS One 8, e78011 (2013).
Cole, J. A. et al. Spatially-resolved metabolic cooperativity within dense bacterial colonies. BMC Syst. Biol. 9, 15 (2015).
Borer, B., Ataman, M., Hatzimanikatis, V. & Or, D. Modeling metabolic networks of individual bacterial agents in heterogeneous and dynamic soil habitats (IndiMeSH). PLoS Comput. Biol. 15, e1007127 (2019).
Acknowledgements
We are grateful to members of the Segrè, Sanchez and Harcombe labs for helpful inputs and discussions at multiple stages of the development of COMETS. We also thank M. Hasson for his contribution to the development of the code. The development of COMETS was initially supported by the U.S. Department of Energy, Office of Science, Office of Biological & Environmental Research, grant DE-SC0004962 to D.S. D.S. also acknowledges funding from the U.S. Department of Energy, Office of Science, Office of Biological & Environmental Research through the Microbial Community Analysis and Functional Evaluation in Soils SFA Program (m-CAFEs) under contract number DE-AC02-05CH11231 to Lawrence Berkeley National Laboratory; the NIH (T32GM100842, 5R01DE024468, R01GM121950), the National Science Foundation (1457695 and NSFOCE-BSF 1635070), the Human Frontiers Science Program (RGP0020/2016) and the Boston University Interdisciplinary Biomedical Research Office. A.R.P. was supported by a Howard Hughes Medical Institute Gilliam Fellowship and a National Academies of Sciences, Engineering, and Medicine Ford Foundation Predoctoral Fellowship. S.S. was funded by SINTEF, the Norwegian graduate research school in bioinformatics, biostatistics and systems biology (NORBIS) and by the INBioPharm project of the Centre for Digital Life Norway (Research Council of Norway grant no. 248885). W.R.H. acknowledges funding from RO1GM121498. Work by A.S., D.B. and J.C.C.V. was supported by a young investigator award from the Human Frontier Science Program (RGY0077/2016), by a Packard Fellowship from the David and Lucile Packard foundation, and by the National Institutes of Health through grant 1R35 GM133467-01 to A.S. K.S.K. was supported by Simons Foundation Grants #409704 and by the Research Corporation for Science Advancement through Cottrell Scholar Award #24010.
Author information
Authors and Affiliations
Contributions
Overall management of COMETS platform: D.S., I.D. Conceptualization of COMETS capabilities: D.S., I.D., A.S., W.H., K.K. Writing and maintenance of initial COMETS code: I.D., W.J.R. Development of current COMETS software and capabilities: I.D., D.B., J.C., M.Q. Writing of specific modules: W.J.R., I.D., J.C., D.B., M.Q., S.S. Preparing and implementing protocols: I.D., J.C., D.B., M.Q., A.R.P., S.S., J.V. Conceptualization and preparation of the manuscript: I.D., D.B., J.C., M.Q., J.V., S.S., A.R.P., D.B.B., K.K., A.S., W.H., D.S.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Protocols thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Harcombe, W. R. et al. Cell Rep. 7, 1104–1115 (2104): https://doi.org/10.1016/j.celrep.2014.03.070
Chacón, J. M., Möbius, W. & Harcombe, W. R. ISME J. 12, 669–680 (2018): https://doi.org/10.1038/s41396-017-0038-0
Bajić, D. et al. Proc. Natl Acad. Sci. USA 115, 11286–11291 (2018): https://doi.org/10.1073/pnas.1808485115
Extended data
Extended Data Fig. 1 Sensitivity of the simulation results depending on the value of the finite time step.
Starting with a simulation identical to the one in Procedure 7, we repeated it with four different values of the time step: a, images of the final colony morphologies; b, plot of the total biomass change with time, illustrating the magnitude of the error due to the finite time step size. The simulation time step size should be chosen such that final simulation result is within the tolerated error.
Extended Data Fig. 2 Sensitivity of the simulation results depending on the value of the finite spatial grid size.
Starting with a simulation identical to the one in Procedure 7, we repeated it with four different values of the grid size: a, images of the final colony morphologies; b, plot of the total biomass change with time, illustrating the magnitude of the error due to the finite grid size. The simulation finite spatial grid size should be chosen such that the final simulation result is within the tolerated error.
Extended Data Fig. 3 Sensitivity of the simulation results depending on the value of the amplitude of the demographic noise.
Starting with a simulation identical to the one in Procedure 7, we repeated it with two different magnitudes of the noise amplitude σ: a,b, images of three replicas of a colony simulation (a), and plot of the total biomass change with time of the three replicas simulations with σ = 0.01 (b); c,d, Images of three replicas of a colony simulation (c), and plot of the total biomass change with time of the three replicas simulations with σ = 0.001 (d). A finalized result of a simulation study in presence of noise should be averaged over several replicas of the stochastic simulation. The change of the noise amplitude, however, may have a substantial effect of the growth rate and the final morphology. The value of the noise amplitude should be chosen to best represent an experimental result.
Extended Data Fig. 4 COMETS simulations time benchmarking.
In order to benchmark the performance of COMETS with increasing complexity of the simulated system, we performed a 24 h batch culture run similar to that in Procedure 1, with either 1, 10 or 100 models (the E. coli model iJO1366 was used in all instances). The settings were identical to Procedure 1 in the main text. We tested three timesteps, 0.01 h (circles), 0.1 h (triangles) and 0.5 h. (squares). The x-axis shows simulated time (i.e., number of simulation steps × timeStep, in h); the y-axis shows elapsed simulation time (the time taken by the computer to run the program), in min. Simulations were performed in Python using cometspy in a personal laptop running linux (Intel Core i7-10610U CPU at 1.80 GHz × 4 cores, 15.3 GiB memory).
Extended Data Fig. 5 The GUI of COMETS.
COMETS simulations can be started from the GUI by loading a previously prepared layout, models and parameters files. It is meant mostly as a training tool with limited functionality. Future development of COMETS will focus on the development of a comprehensive GUI.
Supplementary information
Supplementary Information
Supplementary Discussions 1–5, Supplementary Table 1 and References.
Extended Data Video 1
Branching colony of two identical strains of E. coli. Procedure 7, option A: growth regime without genetic demixing.
Extended Data Video 2
Branching colony of two identical strains of E. coli. Procedure 7, option B: growth regime with genetic demixing.
Rights and permissions
About this article
Cite this article
Dukovski, I., Bajić, D., Chacón, J.M. et al. A metabolic modeling platform for the computation of microbial ecosystems in time and space (COMETS). Nat Protoc 16, 5030–5082 (2021). https://doi.org/10.1038/s41596-021-00593-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-021-00593-3
This article is cited by
-
Technologies for design-build-test-learn automation and computational modelling across the synthetic biology workflow: a review
Network Modeling Analysis in Health Informatics and Bioinformatics (2024)
-
Discretised Flux Balance Analysis for Reaction–Diffusion Simulation of Single-Cell Metabolism
Bulletin of Mathematical Biology (2024)
-
PhysiBoSS 2.0: a sustainable integration of stochastic Boolean and agent-based modelling frameworks
npj Systems Biology and Applications (2023)
-
Vitamin interdependencies predicted by metagenomics-informed network analyses and validated in microbial community microcosms
Nature Communications (2023)
-
Microbiome engineering for bioremediation of emerging pollutants
Bioprocess and Biosystems Engineering (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.