Abstract
The mechanism for initiating DNA cleavage by DDE-family enzymes, including the RAG endonuclease, which initiates V(D)J recombination, is not well understood. Here we report six cryo-EM structures of zebrafish RAG in complex with one or two intact recombination signal sequences (RSSs), at up to 3.9-Å resolution. Unexpectedly, these structures reveal DNA melting at the heptamer of the RSSs, thus resulting in a corkscrew-like rotation of coding-flank DNA and the positioning of the scissile phosphate in the active site. Substrate binding is associated with dimer opening and a piston-like movement in RAG1, first outward to accommodate unmelted DNA and then inward to wedge melted DNA. These precleavage complexes show limited base-specific contacts of RAG at the conserved terminal CAC/GTG sequence of the heptamer, thus suggesting conservation based on a propensity to unwind. CA and TG overwhelmingly dominate terminal sequences in transposons and retrotransposons, thereby implicating a universal mechanism for DNA melting during the initiation of retroviral integration and DNA transposition.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Aziz, R. K., Breitbart, M. & Edwards, R. A. Transposases are the most abundant, most ubiquitous genes in nature. Nucleic Acids Res. 38, 4207–4217 (2010).
Hickman, A. B., Chandler, M. & Dyda, F. Integrating prokaryotes and eukaryotes: DNA transposases in light of structure. Crit. Rev. Biochem. Mol. Biol. 45, 50–69 (2010).
Montaño, S. P. & Rice, P. A. Moving DNA around: DNA transposition and retroviral integration. Curr. Opin. Struct. Biol. 21, 370–378 (2011).
Schatz, D. G., Oettinger, M. A. & Baltimore, D. The V(D)J recombination activating gene, RAG-1. Cell 59, 1035–1048 (1989).
Oettinger, M. A., Schatz, D. G., Gorka, C. & Baltimore, D. RAG-1 and RAG-2, adjacent genes that synergistically activate V(D)J recombination. Science 248, 1517–1523 (1990).
Tonegawa, S. Somatic generation of antibody diversity. Nature 302, 575–581 (1983).
Fanning, L., Connor, A., Baetz, K., Ramsden, D. & Wu, G. E. Mouse RSS spacer sequences affect the rate of V(D)J recombination. Immunogenetics 44, 146–150 (1996).
Ngo, V. N. et al. Oncogenically active MYD88 mutations in human lymphoma. Nature 470, 115–119 (2011).
Akira, S., Okazaki, K. & Sakano, H. Two pairs of recombination signals are sufficient to cause immunoglobulin V-(D)-J joining. Science 238, 1134–1138 (1987).
Ramsden, D. A., Baetz, K. & Wu, G. E. Conservation of sequence in recombination signal sequence spacers. Nucleic Acids Res. 22, 1785–1796 (1994).
Schatz, D. G. & Ji, Y. Recombination centres and the orchestration of V(D)J recombination. Nat. Rev. Immunol. 11, 251–263 (2011).
Schatz, D. G. & Swanson, P. C. V. V(D)J recombination: mechanisms of initiation. Annu. Rev. Genet. 45, 167–202 (2011).
Ru, H. et al. Molecular mechanism of V(D)J recombination from synaptic RAG1-RAG2 complex structures. Cell 163, 1138–1152 (2015).
Kim, M. S. et al. Cracking the DNA code for V(D)J recombination. Mol. Cell 70, 358–370.e4 (2018).
Lieber, M. R. The mechanism of double-strand DNA break repair by the nonhomologous DNA end-joining pathway. Annu. Rev. Biochem. 79, 181–211 (2010).
Lee, I. & Harshey, R. M. Patterns of sequence conservation at termini of long terminal repeat (LTR) retrotransposons and DNA transposons in the human genome: lessons from phage Mu. Nucleic Acids Res. 31, 4531–4540 (2003).
Yang, W., Lee, J. Y. & Nowotny, M. Making and breaking nucleic acids: two-Mg2+-ion catalysis and substrate specificity. Mol. Cell 22, 5–13 (2006).
Swanson, P. C. The bounty of RAGs: recombination signal complexes and reaction outcomes. Immunol. Rev. 200, 90–114 (2004).
Swanson, P. C. Fine structure and activity of discrete RAG-HMG complexes on V(D)J recombination signals. Mol. Cell. Biol. 22, 1340–1351 (2002).
Kim, M. S., Lapkouski, M., Yang, W. & Gellert, M. Crystal structure of the V(D)J recombinase RAG1–RAG2. Nature 518, 507–511 (2015).
Hesse, J. E., Lieber, M. R., Mizuuchi, K. & Gellert, M. V(D)J recombination: a functional definition of the joining signals. Genes Dev. 3, 1053–1061 (1989).
Grawunder, U. & Lieber, M. R. A complex of RAG-1 and RAG-2 proteins persists on DNA after single-strand cleavage at V(D)J recombination signal sequences. Nucleic Acids Res. 25, 1375–1382 (1997).
Patel, D. J., Shapiro, L. & Hare, D. Nuclear magnetic resonance and distance geometry studies of DNA structures in solution. Annu. Rev. Biophys. Biophys. Chem. 16, 423–454 (1987).
Ramsden, D. A., McBlane, J. F., van Gent, D. C. & Gellert, M. Distinct DNA sequence and structure requirements for the two steps of V(D)J recombination signal cleavage. EMBO J. 15, 3197–3206 (1996).
Hiom, K. & Gellert, M. A stable RAG1-RAG2-DNA complex that is active in V(D)J cleavage. Cell 88, 65–72 (1997).
Cuomo, C. A., Mundy, C. L. & Oettinger, M. A. DNA sequence and structure requirements for cleavage of V(D)J recombination signal sequences. Mol. Cell. Biol. 16, 5683–5690 (1996).
Peak, M. M., Arbuckle, J. L. & Rodgers, K. K. The central domain of core RAG1 preferentially recognizes single-stranded recombination signal sequence heptamer. J. Biol. Chem. 278, 18235–18240 (2003).
Swanson, P. C. & Desiderio, S. V(D)J recombination signal recognition: distinct, overlapping DNA-protein contacts in complexes containing RAG1 with and without RAG2. Immunity 9, 115–125 (1998).
Santagata, S. et al. The RAG1/RAG2 complex constitutes a 3′ flap endonuclease: implications for junctional diversity in V(D)J and transpositional recombination. Mol. Cell 4, 935–947 (1999).
Yanagihara, K. & Mizuuchi, K. Progressive structural transitions within Mu transpositional complexes. Mol. Cell 11, 215–224 (2003).
Hickman, A. B. et al. Structural basis of hAT transposon end recognition by Hermes, an octameric DNA transposase from Musca domestica. Cell 158, 353–367 (2014).
Hickman, A. B. et al. Molecular architecture of a eukaryotic DNA transposase. Nat. Struct. Mol. Biol. 12, 715–721 (2005).
Davies, D. R., Mahnke Braam, L., Reznikoff, W. S. & Rayment, I. The three-dimensional structure of a Tn5 transposase-related protein determined to 2.9-A resolution. J. Biol. Chem. 274, 11904–11913 (1999).
Davies, D. R., Goryshin, I. Y., Reznikoff, W. S. & Rayment, I. Three-dimensional structure of the Tn5 synaptic complex transposition intermediate. Science 289, 77–85 (2000).
Lee, I. & Harshey, R. M. The conserved CA/TG motif at Mu termini: T specifies stable transpososome assembly. J. Mol. Biol. 330, 261–275 (2003).
Bai, X. C., Rajendra, E., Yang, G., Shi, Y. & Scheres, S. H. Sampling the conformational space of the catalytic subunit of human γ-secretase. eLife 4, e11182 (2015).
Chen, S. et al. High-resolution noise substitution to measure overfitting and validate resolution in 3D structure determination by single particle electron cryomicroscopy. Ultramicroscopy 135, 24–35 (2013).
Yuan, Z. et al. Structure of the eukaryotic replicative CMG helicase suggests a pumpjack motion for translocation. Nat. Struct. Mol. Biol. 23, 217–224 (2016).
Yin, F. F. et al. Structure of the RAG1 nonamer binding domain with DNA reveals a dimer that mediates DNA synapsis. Nat. Struct. Mol. Biol. 16, 499–508 (2009).
Pettersen, E. F. et al. UCSF Chimera: a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, (213–221 (2010).
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 66, 12–21 (2010).
Allegretti, M., Mills, D. J., McMullan, G., Kühlbrandt, W. & Vonck, J. Atomic model of the F420-reducing [NiFe] hydrogenase by electron cryo-microscopy using a direct electron detector. eLife 3, e01963 (2014).
Bartesaghi, A., Matthies, D., Banerjee, S., Merk, A. & Subramaniam, S. Structure of β-galactosidase at 3.2-Å resolution obtained by cryo-electron microscopy. Proc. Natl Acad. Sci. USA 111, 11709–11714 (2014).
Delano, W.L. The PyMOL Molecular Graphics System (DeLano Scientific, 2002).
Acknowledgements
Some of this work was performed with the assistance of H. Wei at the Simons Electron Microscopy Center and the National Resource for Automated Molecular Microscopy at the New York Structural Biology Center, supported by grants from the Simons Foundation (349247), NYSTAR, and the NIH National Institute of General Medical Sciences (GM103310). This work was supported by the National Cancer Institute’s National Cryo-EM Facility at the Frederick National Laboratory for Cancer Research. We also acknowledge research funding from the National Institutes of Health (AI125535 to H.W. and M.L., AI0240067-36 to F.W.A., and AI032524 to D.G.S.) and an Irvington Postdoctoral Fellowship from the Cancer Research Institute (H.R.). F.W.A. is supported as an HHMI investigator.
Author information
Authors and Affiliations
Contributions
H.R., H.W., and F.W.A. conceived and initiated the project. H.W. and H.R. participated in project design and analysis. H.R. prepared RAG complexes for biochemical studies and EM experiments. P.Z. assisted with preliminary biochemical assays. W.M. performed cryo-EM data collection on a Polara microscope at Harvard Medical School, and W.M., H.R., and H.W. participated in cryo-EM data collection on a Titan Krios microscope at the Simons Electron Microscopy Center. M.L., H.R., W.M., and H.W. carried out image processing and reconstruction. H.R. performed model building, refinement, and calculation of FSC curves. H.W., H.R., D.G.S., M.L., and F.W.A. performed data analysis, and discussion and interpretation of results. H.W. and H.R. made the figures and wrote the manuscript, and all authors edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
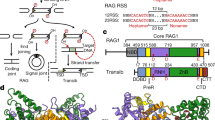
Supplementary Figure 1 V(D)J recombination process and DNA sequences used in this study.
a, Schematic representation of a simplified V(D)J recombination process at the human lgH locus. V, D, J and constant regions are shown as light purple, light green, light blue and gray rectangles, and 12- and 23-RSSs are shown as red and orange triangles. Randomly incorporated nucleotides between V, D and J coding segments are shown as blue rectangles. b, 12-RSS and 23-RSS sequences used in this study. Heptamer sequences (red) are numbered from 1 to 7 for the forward strand and from 1* to 7* for the reverse strand. Coding flank sequences are numbered from −16 to −1 for the forward strand and from −16* to −1* for the reverse strand. The nicking site is between A–1 (coding flank) and C1 (RSS) with a 3’-OH on A–1.
Supplementary Figure 2 Cryo-EM structure determination.
a, A flow chart of cryo-EM structure determination for the data collected using an FEI Titan Krios microscope on an intact DNA complex sample prepared at 21 °C. Final maps are enclosed in black boxes. b, A flow chart of cryo-EM structure determination for the combined data collected using an FEI Polara microscope on an intact DNA complex sample prepared at 37 °C and using an FEI Titan Krios microscope on an intact DNA complex sample prepared at 21 °C. Final maps are enclosed in black boxes. Some major classes were not pursued further because they either did not contain bound DNA or contained a DNA state for which we already had a reconstruction at a good resolution.
Supplementary Figure 3 Cryo-EM structure determination
a, An alternative flow chart of cryo-EM structure determination for the combined data collected using an FEI Polara microscope on an intact DNA complex sample prepared at 37 °C and using an FEI Titan Krios microscope on an intact DNA complex sample prepared at 21 °C. Final maps are enclosed in black boxes. b, A flow chart of cryo-EM structure determination for the RAG in complex with nicked RSS intermediates. Final maps are enclosed in black boxes. Some major classes were not pursued further because they either did not contain bound DNA or contained a DNA state for which we already had a reconstruction at a good resolution.
Supplementary Figure 4 Fourier shell correlation curves for all the maps.
Black lines: FSC curves between one half-map to the other half-map. Red lines: FSC curves between a final refined model and the sum of two half-maps. Green lines: FSCwork curves between a half-map and a model refined against the same half-map. Blue lines: FSCfree curves between a half-map and a model refined against the other half-map. Black and red dashed lines indicate the resolutions at FSC = 0.143 and 0.5 for half maps to half maps and models to maps, respectively.
Supplementary Figure 5 Overview of cryo-EM maps and models.
a,b, Cryo-EM maps of RAG singly bound to unmelted (a) and melted (b) 12-RSS at 4.2 and 4.7 Å resolution. One RAG1 is omitted in order to present the bound 12-RSS DNAs (red) inside the protein. The black circle in (b) indicates the melted region in the RSS. c, Cryo-EM map of C2-symmetrized RAG doubly bound to nicked RSSs at 3.0 Å resolution. One RAG1 is omitted in order to present the bound nicked-RSS DNAs (red and orange) inside the protein. d, Orthogonal view of cryo-EM map of RAG doubly bound to melted 12- and 23-RSS at 5.0 Å resolution. One RAG1 is omitted in order to present the bound RSS DNAs (red and orange) inside the protein. The black circle indicates the melted region in the RSS. e, Ribbon diagram of the RAG model doubly bound to unmelted 12- and 23-RSS at 4.3 Å resolution. RAG1: lemon green and cyan; RAG2: green and slate. 12-RSS and 23-RSS are shown in salmon, except that heptamers are highlighted in red and nonamers in magenta. f, Orthogonal view of C2 symmetrized cryo-EM map of RAG doubly bound to melted RSSs at 4.4 Å resolution. One RAG1 is omitted (left) in order to present the bound RSS DNAs (red and orange) inside the protein. The black circle indicates the melted region in the RSS. g-i, Representative protein regions of the 4.0 Å-resolution averaged map with one RSS melted and the other RSS unmelted (shown in mesh) superimposed with the final models (shown in ribbon). These regions include α22 (I954-E970) and α23 (I975-M996) helices of the first RAG1 molecule for the melted DNA (g), β-sheet region (F611-M622, K638-I651 and F722-K731) of RNH domain of the first RAG1 molecule for the melted DNA (h) and β4-β5 loop region (V625-K638) of the second RAG1 molecule relative to the melted DNA (i). Cryo-EM densities for catalytic residues D620, D730 and E984 as well as the two bound Ca2+ ions in the active site center are indicated. j, Representative DNA regions of the cryo-EM maps (shown in mesh) superimposed to the final models with the phosphate backbones in sticks and bases in cartoons. The regions include positions −4 to +8 of 12-RSS whose sequences are shown in Supplementary Fig. 1b. Left: melted RSS from the 4.0 Å-resolution averaged map with one RSS melted and the other RSS unmelted; middle: unmelted RSS from the 3.9 Å-resolution C2-symmetrized map with unmelted RSSs; right: nicked RSS from the 3.0 Å-resolution C2-symmetrized map with nicked RSSs. Cryo-EM densities for the three nucleotides CAC in the heptamer are indicated.
Supplementary Figure 6 Comparison of the DNAs and proteins at different states.
a,b, Detailed interactions of the RNH domain (yellow) from the 1st RAG1 molecule with unmelted (a) and melted (b) RSS (pale cyan) with heptamer highlighted in salmon. The 1st RAG1 is defined as the RAG1 molecule that performs catalysis on the bound RSSs. The main chain or side chain of the residues in RNH that have potential interaction with DNA are shown as sticks (light blue). The nucleotide to be nicked is highlighted in cyan. c, Active site in the RNH region (shown in yellow) for nicked RSS (shown as sticks). The heptamers and coding flanks are shown in salmon and light cyan, respectively. The nucleotides to be nicked are highlighted in cyan. The metal ions (A and B) in the active centers are shown as orange spheres and residues that coordinate the metal ions are highlighted in light blue. Dashed black lines indicate potential coordination between metal ions and ligands. d, Superposition of the RNH domains that are bound to melted and nicked RSSs showing that the RNH active sites remain the same whether they are poised for nicking or hairpin formation. Bound RSSs are omitted. The zoom-in view displays the active site residues and metal ions.
Supplementary Figure 7 Detailed interactions of the ID with unmelted and melted DNAs in the coding flank–RSS junction.
a, Superposition of the 1st RAG1 that binds unmelted (colored as in Fig. 1a), melted (green) and nicked (cyan) RSSs respectively, and of Apo-RAG (gray). The 1st RAG1 is defined as the RAG1 molecule that performs catalysis on the bound RSSs. The α15 helices are indicated by black arrows. b,c, Detailed interactions of the ID (magenta) of the 1st RAG with unmelted RSS (pale cyan) (b) and melted RSS (pale cyan) (c) with heptamer highlighted in salmon. The main chain or side chain of the residues in ID that have potential interaction with DNA are shown as sticks (light blue). The nucleotide that is potentially recognized specifically by RAG is highlighted in red.
Supplementary Figure 8 Interactions of the β4–β5 loop with different DNAs.
a,b, Detailed interactions of the β4-β5 loop (lemon) from the 2nd RAG1 with melted DNA (a) and nicked DNA (b), respectively. The 2nd RAG1 is defined as the RAG1 molecule that only mediates the binding of the RSS but not catalysis. Heptamers are highlighted in salmon. The main chains or side chains of residues in the β4-β5 loop that have potential interactions with the DNA are shown as sticks (light blue). The nucleotides to be nicked or cleaved are highlighted in cyan.
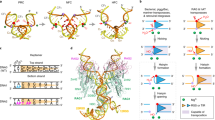
Supplementary Figure 9 Structural and mechanistic implications for DNA transposases.
a, Superposition of the apo-Hermes dimer (2BW3) and the Hermes dimer bound to inverted repeats (light blue) (4D1Q) by aligning one Hermes molecule (left). Only the RNH and ID domains in Hermes are shown. The RNH domains in apo-Hermes and Hermes bound to DNA are shown in pale green and yellow, respectively, and the ID domains in apo-Hermes and Hermes bound to DNA are shown in gray and pink, respectively. The difference at the second Hermes monomer between the apo form and the synaptic complex form is evident (red arrow). b, Superposition of the inactive Tn5 dimer (1B7E) and the Tn5 dimer bound to hairpin mimic DNA (light blue) (1MUH) by aligning one Tn5 molecule (left). Only the RNH and ID domains in Tn5 are shown. The RNH domains in apo-Tn5 and Tn5 bound to hairpin mimic DNA are shown in pale green and yellow, respectively, and the ID domains in apo-Tn5 and Tn5 bound to hairpin mimic DNA are shown in gray and pink, respectively. The difference at the second Tn5 monomer between the apo form and the synaptic complex form is evident (red arrow). c, Examples of the signal or inverted repeat sequences in RAG and other DNA transposases and integrases. The flanking sequences are shown in lower case and the signal or IR sequences are shown in upper case. The conserved di-nucleotides CA and GT around the cleavage sites are highlighted in red. MuA: transposase of the bacteriophage Mu; Mos1: transposase in the Mariner family; PFV: prototype foamy virus integrase.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9, Supplementary Note and Supplementary Table 1
Supplementary Video 1
A rotational view of RAG in complex with one unmelted RSS and one melted RSS. The RAG structure in complex with melted 12-RSS and unmelted 23-RSS (PDB ID 6DBV) is shown rotating around a vertical axis that is close to the 2-fold axis of the core region of the RAG1–RAG2 complex.
Supplementary Video 2
A composite morph from apo-RAG to RAG in complex with doubly unmelted RSS, to RAG in complex with one melted RSS and one unmelted RSS, and to RAG in complex with doubly melted RSS. The zebrafish apo-RAG structure was modeled from PDB ID 4WWX, and PDB IDs for zebrafish RAG structures with doubly unmelted RSS, one melted RSS and one unmelted RSS, and doubly melted RSS are 6DBT, 6DBV, and 6DBL respectively.
Rights and permissions
About this article
Cite this article
Ru, H., Mi, W., Zhang, P. et al. DNA melting initiates the RAG catalytic pathway. Nat Struct Mol Biol 25, 732–742 (2018). https://doi.org/10.1038/s41594-018-0098-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41594-018-0098-5
This article is cited by
-
The role of chromatin loop extrusion in antibody diversification
Nature Reviews Immunology (2022)
-
Structural insights into the evolution of the RAG recombinase
Nature Reviews Immunology (2022)
-
Clinical Manifestations, Mutational Analysis, and Immunological Phenotype in Patients with RAG1/2 Mutations: First Cases Series from Mexico and Description of Two Novel Mutations
Journal of Clinical Immunology (2021)
-
Identification of RAG-like transposons in protostomes suggests their ancient bilaterian origin
Mobile DNA (2020)
-
How mouse RAG recombinase avoids DNA transposition
Nature Structural & Molecular Biology (2020)