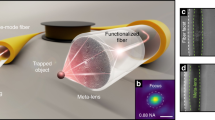
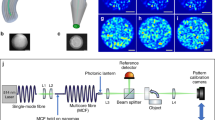
Abstract
Holographic optical tweezers (HOT) hold great promise for many applications in biophotonics, allowing the creation and measurement of minuscule forces on biomolecules, molecular motors and cells. Geometries used in HOT currently rely on bulk optics, and their exploitation in vivo is compromised by the optically turbid nature of tissues. We present an alternative HOT approach in which multiple three-dimensional (3D) traps are introduced through a high-numerical-aperture multimode optical fibre, thus enabling an equally versatile means of manipulation through channels having cross-section comparable to the size of a single cell. Our work demonstrates real-time manipulation of 3D arrangements of micro-objects, as well as manipulation inside otherwise inaccessible cavities. We show that the traps can be formed over fibre lengths exceeding 100 mm and positioned with nanometric resolution. The results provide the basis for holographic manipulation and other high-numerical-aperture techniques, including advanced microscopy, through single-core-fibre endoscopes deep inside living tissues and other complex environments.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ashkin, A., Dziedzic, J. M., Bjorkholm, J. E. & Chu, S. Observation of a single-beam gradient force optical trap for dielectric particles. Opt. Lett. 11, 288–290 (1986).
Fazal, F. M. & Block, S. M. Optical tweezers study life under tension. Nat. Photon. 5, 318–321 (2011).
Svoboda, K. & Block, S. M. Force and velocity measured for single kinesin molecules. Cell 77, 773–784 (1994).
Bustamante, C., Macosko, J. C. & Wuite, G. J. L. Grabbing the cat by the tail: manipulating molecules one by one. Nat. Rev. Mol. Cell. Biol. 1, 130–136 (2000).
Bustamante, C., Bryant, Z. & Smith, S. B. Ten years of tension: single-molecule DNA mechanics. Nature 421, 423–427 (2003).
Grier, D. G. A revolution in optical manipulation. Nature 424, 810–816 (2003).
Dholakia, K. & Reece, P. Optical micromanipulation takes hold. Nano Today 1, 18–27 (February, 2006).
Padgett, M. & Di Leonardo, R. Holographic optical tweezers and their relevance to lab on chip devices. Lab Chip 11, 1196–1205 (2011).
Denk, W., Strickler, J. & Webb, W. Two-photon laser scanning fluorescence microscopy. Science 248, 73–76 (1990).
Hell, S. W. & Wichmann, J. Breaking the diffraction resolution limit by stimulated emission: stimulated-emission-depletion fluorescence microscopy. Opt. Lett. 19, 780–782 (1994).
Dickson, R. M., Cubitt, A. B., Tsien, R. Y. & Moerner, W. E. On/off blinking and switching behaviour of single molecules of green fluorescent protein. Nature 388, 355–358 (1997).
Betzig, E. et al. Imaging intracellular fluorescent proteins at nanometer resolution. Science 313, 1642–1645 (2006).
Jun, Y., Tripathy, S. K., Narayanareddy, B. R., Mattson-Hoss, M. K. & Gross, S. P. Calibration of optical tweezers for in vivo force measurements: how do different approaches compare? Biophys. J. 107, 1474–1484 (2014).
Zhong, M.-C., Wei, X.-B., Zhou, J.-H., Wang, Z.-Q. & Li, Y.-M. Trapping red blood cells in living animals using optical tweezers. Nat. Commun. 4, 1768 (2013).
Bambardekar, K., Clément, R., Blanc, O., Chardès, C. & Lenne, P.-F. Direct laser manipulation reveals the mechanics of cell contacts in vivo. Proc. Natl Acad. Sci. USA 112, 1416–1421 (2015).
Constable, A., Kim, J., Mervis, J., Zarinetchi, F. & Prentiss, M. Demonstration of a fiber-optical light-force trap. Opt. Lett. 18, 1867–1869 (1993).
Ribeiro, R. S. R., Soppera, O., Oliva, A. G., Guerreiro, A. & Jorge, P. A. S. New trends on optical fiber tweezers. J. Lightw. Technol. 33, 3394–3405 (2015).
Liberale, C. et al. Miniaturized all-fibre probe for three-dimensional optical trapping and manipulation. Nat. Photon. 1, 723–727 (2007).
Berthelot, J. et al. Three-dimensional manipulation with scanning near-field optical nanotweezers. Nat. Nanotech. 9, 295–299 (2014).
Guck, J., Ananthakrishnan, R., Moon, T. J., Cunningham, C. C. & Käs, J. Optical deformability of soft biological dielectrics. Phys. Rev. Lett. 84, 5451–5454 (2000).
Guck, J. et al. Optical deformability as an inherent cell marker for testing malignant transformation and metastatic competence. Biophys. J. 88, 3689–3698 (2005).
Kreysing, M. K. et al. The optical cell rotator. Opt. Express. 16, 16984–16992 (2008).
Kreysing, M. et al. Dynamic operation of optical fibres beyond the single-mode regime facilitates the orientation of biological cells. Nat. Commun. 5, 5481 (2014).
Mosk, A. P., Lagendijk, A., Lerosey, G. & Fink, M. Controlling waves in space and time for imaging and focusing in complex media. Nat. Photon. 6, 283–292 (2012).
Vellekoop, I. M. & Mosk, A. P. Focusing coherent light through opaque strongly scattering media. Opt. Lett. 32, 2309–2311 (2007).
Bertolotti, J. et al. Non-invasive imaging through opaque scattering layers. Nature 491, 232–234 (2012).
Čižmár, T., Mazilu, M. & Dholakia, K. In situ wavefront correction and its application to micromanipulation. Nat. Photon. 4, 388–394 (2010).
Di Leonardo, R. & Bianchi, S. Hologram transmission through multi-mode optical fibers. Opt. Express. 19, 247–254 (2011).
Čižmár, T. & Dholakia, K. Shaping the light transmission through a multimode optical fibre: complex transformation analysis and applications in biophotonics. Opt. Express 19, 18871–18884 (2011).
Bianchi, S. & Di Leonardo, R. A multi-mode fiber probe for holographic micromanipulation and microscopy. Lab Chip 12, 635–639 (2012).
Čižmár, T. & Dholakia, K. Exploiting multimode waveguides for pure fibre-based imaging. Nat. Commun. 3, 1027 (2012).
Choi, Y. et al. Scanner-free and wide-field endoscopic imaging by using a single multimode optical fiber. Phys. Rev. Lett. 109, 203901 (2012).
Papadopoulos, I. N., Farahi, S., Moser, C. & Psaltis, D. High-resolution, lensless endoscope based on digital scanning through a multimode optical fiber. Biomed. Opt. Express 4, 260–270 (2013).
Papadopoulos, I. N., Farahi, S., Moser, C. & Psaltis, D. Focusing and scanning light through a multimode optical fiber using digital phase conjugation. Opt. Express 20, 10583–10590 (2012).
Plöschner, M., Tyc, T. & Čižmár, T. Seeing through chaos in multimode fibres. Nat. Photon. 9, 529–535 (2015).
Morales-Delgado, E. E., Psaltis, D. & Moser, C. Two-photon imaging through a multimode fiber. Opt. Express 23, 32158–32170 (2015).
Plöschner, M. et al. Multimode fibre: light-sheet microscopy at the tip of a needle. Sci. Rep. 5, 18050 (2015).
Amitonova, L. V. et al. High-resolution wavefront shaping with a photonic crystal fiber for multimode fiber imaging. Opt. Lett. 41, 497–500 (2016).
Papadopoulos, I. N., Farahi, S., Moser, C. & Psaltis, D. Increasing the imaging capabilities of multimode fibers by exploiting the properties of highly scattering media. Opt. Lett. 38, 2776–2778 (2013).
Choi, Y., Yoon, C., Kim, M., Yang, J. & Choi, W. Disorder-mediated enhancement of fiber numerical aperture. Opt. Lett. 38, 2253–2255 (2013).
Bianchi, S. et al. Focusing and imaging with increased numerical apertures through multimode fibers with micro-fabricated optics. Opt. Lett. 38, 4935–4937 (2013).
Wadsworth, W. J. et al. Very high numerical aperture fibers. IEEE Photon. Technol. Lett. 16, 843–845 (2004).
Popoff, S. M. et al. Measuring the transmission matrix in optics: an approach to the study and control of light propagation in disordered media. Phys. Rev. Lett. 104, 100601 (2010).
Popoff, S. M., Lerosey, G., Fink, M., Boccara, A. C. & Gigan, S. Image transmission through an opaque material. Nat. Commun. 1, 1–5 (2010).
Gloge, D. Weakly guiding fibers. Appl. Opt. 10, 2252–2258 (1971).
Issa, N. A. High numerical aperture in multimode microstructured optical fibers. Appl. Opt. 43, 6191–6197 (2004).
Ho, K.-P. & Kahn, J. M. Mode-dependent loss and gain: statistics and effect on mode-division multiplexing. Opt. Express 19, 16612–16635 (2011).
Neuman, K. C. & Block, S. M. Optical trapping. Rev. Sci. Instrum. 75, 2787–2809 (2004).
Jiang, X. et al. Single-mode hollow-core photonic crystal fiber made from soft glass. Opt. Express 19, 15438–15444 (2011).
Jiang, X. et al. Supercontinuum generation in ZBLAN glass photonic crystal fiber with six nanobore cores. Opt. Lett. 41, 4245–4248 (2016).
Richards, B. & Wolf, E. Electromagnetic diffraction in optical systems. II. Structure of the image field in an aplanatic system. Proc. R. Soc. A 253, 358–379 (1959).
Novotny, L. & Hecht, B. Principles of Nano-Optics (Cambridge Univ. Press, Cambridge, 2006).
Kramers, H. Brownian motion in a field of force and the diffusion model of chemical reactions. Physica 7, 284–304 (1940).
Acknowledgements
I.T.L. and S.T. acknowledge funding from the People Programme (Marie Curie Actions) of the European Union’s Seventh Framework Programme (FP7/2007–2013) under REA grant agreement no. 608144. I.T.L., S.T. and T.Č. also acknowledge financial support from the Thüringer Ministerium für Wirtschaft, Wissenschaft und Digitale Gesellschaft, the Thüringer Aufbaubank and the Federal Ministry of Education and Research, Germany (BMBF). M.Š. and T.Č. acknowledge support from the European Regional Development Fund through project no. CZ.02.1.01/0.0/0.0/15003/0000476. T.Č. also acknowledges the University of Dundee and SUPA (Scottish Universities Physics Alliance; PaLS initiative) for financial support. X.J. and P.St.J.R. thank F. Babic for assistance with drawing the fibres. The authors also thank K. Wilcox and Elliot Scientific Ltd for lending equipment used in the experiments.
Author information
Authors and Affiliations
Contributions
I.T.L., S.T. and T.Č. performed all experiments. X.J. and P.St.J.R. designed and manufactured the optical fibres. M.Š. modelled the optical tweezers. I.T.L., A.C. and T.Č. analysed the results. T.Č. led the project. I.T.L. and T.Č. wrote the manuscript with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary methods and results. This file was missing when the Article was first published; it was uploaded 14 December 2017.
Videos
Supplementary Video 1
Dynamic holographic optical tweezers (HOT) manipulation of eight particles in a rotating cube arrangement.
Supplementary Video 2
Dynamic HOT manipulation of six particles illustrating Bohr’s model of the He atom.
Supplementary Video 3
Dynamic HOT axial manipulation of two particles.
Supplementary Video 4
Dynamic HOT manipulation inside a semi-opaque cavity.
Supplementary Video 5
Stability of a static HOT while bending the multimode fibre.
Supplementary Video 6
Simulation of focus fine positioning.
Rights and permissions
About this article
Cite this article
Leite, I.T., Turtaev, S., Jiang, X. et al. Three-dimensional holographic optical manipulation through a high-numerical-aperture soft-glass multimode fibre. Nature Photon 12, 33–39 (2018). https://doi.org/10.1038/s41566-017-0053-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41566-017-0053-8
This article is cited by
-
AI-driven projection tomography with multicore fibre-optic cell rotation
Nature Communications (2024)
-
Large-volume focus control at 10 MHz refresh rate via fast line-scanning amplitude-encoded scattering-assisted holography
Nature Communications (2024)
-
Synchronization of spin-driven limit cycle oscillators optically levitated in vacuum
Nature Communications (2023)
-
Single multimode fibre for in vivo light-field-encoded endoscopic imaging
Nature Photonics (2023)
-
110 μm thin endo-microscope for deep-brain in vivo observations of neuronal connectivity, activity and blood flow dynamics
Nature Communications (2023)