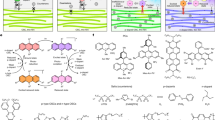
Abstract
High-efficiency blue phosphorescence emission is essential for organic optoelectronic applications. However, synthesizing heavy-atom-free organic systems having high triplet energy levels and suppressed non-radiative transitions—key requirements for efficient blue phosphorescence—has proved difficult. Here we demonstrate a simple chemical strategy for achieving high-performance blue phosphors, based on confining isolated chromophores in ionic crystals. Formation of high-density ionic bonds between the cations of ionic crystals and the carboxylic acid groups of the chromophores leads to a segregated molecular arrangement with negligible inter-chromophore interactions. We show that tunable phosphorescence from blue to deep blue with a maximum phosphorescence efficiency of 96.5% can be achieved by varying the charged chromophores and their counterions. Moreover, these phosphorescent materials enable rapid, high-throughput data encryption, fingerprint identification and afterglow display. This work will facilitate the design of high-efficiency blue organic phosphors and extend the domain of organic phosphorescence to new applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Source data are provided with this paper. The remaining data supporting the findings of this study are available within the paper and its Supplementary Information files and are available from the corresponding authors upon reasonable request.
The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Data Centre under deposition numbers 1888289, 1888291–1888293, 2011667–2011675, 2015867 and 2024673. These data can be obtained free of charge from the Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
References
Xu, H. et al. Recent progress in metal–organic complexes for optoelectronic applications. Chem. Soc. Rev. 43, 3259–3302 (2014).
Pan, M., Liao, W., Yin, S., Sun, S. & Su, C. Single-phase white-light-emitting and photoluminescent color-tuning coordination assemblies. Chem. Rev. 118, 8889–8935 (2018).
Zhang, K. et al. Long-lived emissive probes for time-resolved photoluminescence bioimaging and biosensing. Chem. Rev. 118, 1770–1839 (2018).
Deng, W. et al. 2D Ruddlesden–Popper perovskite nanoplate based deep-blue light-emitting diodes for light communication. Adv. Funct. Mater. 29, 1903861–1903869 (2019).
Nanishi, Y. The birth of the blue LED. Nat. Photon. 8, 884–886 (2014).
Li, Y., Gecevicius, M. & Qiu, J. Long persistent phosphors—from fundamentals to applications. Chem. Soc. Rev. 45, 2090–2136 (2016).
Baldo, M. et al. Highly efficient phosphorescent emission from organic electroluminescent devices. Nature 395, 151–154 (1998).
Hirata, S. et al. Highly efficient blue electroluminescence based on thermally activated delayed fluorescence. Nat. Mater. 14, 330–336 (2015).
Yang, J. et al. The influence of the molecular packing on the room temperature phosphorescence of purely organic luminogens. Nat. Commun. 9, 840 (2018).
Shoji, Y. et al. Unveiling a new aspect of simple arylboronic esters: long-lived room-temperature phosphorescence from heavy-atom-free molecules. J. Am. Chem. Soc. 139, 2728–2733 (2017).
Yang, Z. et al. Intermolecular electronic coupling of organic units for efficient persistent room-temperature phosphorescence. Angew. Chem. Int. Ed. 55, 2181–2185 (2016).
An, Z. et al. Stabilizing triplet excited states for ultralong organic phosphorescence. Nat. Mater. 14, 685–690 (2015).
Lucenti, E. et al. Cyclic triimidazole derivatives: intriguing examples of multiple emissions and ultralong phosphorescence at room temperature. Angew. Chem. Int. Ed. 56, 16302–16307 (2017).
Bolton, O., Lee, K., Kim, H., Lin, K. & Kim, J. Activating efficient phosphorescence from purely organic materials by crystal design. Nat. Chem. 3, 205–210 (2011).
Wei, J. et al. Induction of strong long-lived room-temperature phosphorescence of N-phenyl-2-naphthylamine molecules by confinement in a crystalline dibromobiphenyl matrix. Angew. Chem. Int. Ed. 55, 15589–15593 (2016).
Ma, X., Xu, C., Wang, J. & Tian, H. Amorphous pure organic polymers for heavy-atom-free efficient room-temperature phosphorescence emission. Angew. Chem. Int. Ed. 57, 10854–10858 (2018).
Cai, S. et al. Enabling long-lived organic room temperature phosphorescence in polymers by subunit interlocking. Nat. Commun. 10, 4247 (2019).
Kabe, R. & Adachi, C. Organic long persistent luminescence. Nature 550, 384–387 (2017).
Su, Y. et al. Ultralong room temperature phosphorescence from amorphous organic materials toward confidential information encryption and decryption. Sci. Adv. 4, 9732–9743 (2018).
Zhang, G., Palmer, G., Dewhirst, M. & Fraser, C. A dual-emissive-materials design concept enables tumour hypoxia imaging. Nat. Mater. 8, 747–751 (2009).
Gao, R., Mei, X., Yan, D., Liang, R. & Wei, M. Nano-photosensitizer based on layered double hydroxide and isophthalic acid for singlet oxygenation and photodynamic therapy. Nat. Commun. 9, 2798 (2018).
He, Z. et al. Achieving persistent, efficient, and robust room-temperature phosphorescence from pure organics for versatile applications. Adv. Mater. 31, 1807222 (2019).
Hirata, S. Recent advances in materials with room-temperature phosphorescence: photophysics for triplet exciton stabilization. Adv. Optical Mater. 5, 1700116 (2017).
Kenry, C. C. & Liu, B. Enhancing the performance of pure organic room-temperature phosphorescent luminophores. Nat. Commun. 10, 2111 (2019).
Baryshnikov, G., Minaev, B. & Agren, H. Theory and calculation of the phosphorescence phenomenon. Chem. Rev. 117, 6500–6537 (2017).
Kwon, M. et al. Suppressing molecular motions for enhanced room-temperature phosphorescence of metal-free organic materials. Nat. Commun. 6, 8947 (2015).
Sternlicht, H., Nieman, G. & Robinson, G. Triplet–triplet annihilation and delayed fluorescence in molecular aggregates. J. Chem. Phys. 38, 1326–1335 (1963).
Schulman, E. & Parker, R. Room temperature phosphorescence of organic compounds. The effects of moisture, oxygen, and the nature of the support-phosphor interaction. J. Phys. Chem. 81, 1932–1939 (1977).
Schulman, E. & Walling, C. Phosphorescence of adsorbed ionic organic molecules at room temperature. Science 178, 53–54 (1972).
Braga, D., Grepioni, F. & Desiraju, G. Crystal engineering and organometallic architecture. Chem. Rev. 98, 1375–1405 (1998).
Gu, L. et al. Colour-tunable ultra-long organic phosphorescence of a single-component molecular crystal. Nat. Photon. 13, 406–411 (2019).
Acknowledgements
This work is supported by the National Key R&D Program of China (2020YFA0709900), the National Natural Science Foundation of China (21875104, 21975120, 51673095, 21973043, 91833304 and 91833302), the Natural Science Fund for Distinguished Young Scholars of Jiangsu Province (BK20180037) and the Postgraduate Research & Practice Innovation Program of Jiangsu Province (KYCX21_1098). We are grateful to the High-Performance Computing Center of Nanjing Tech University for technical support.
Author information
Authors and Affiliations
Contributions
W. Ye, H.S., H.M., Z.A., X.L. and W.H. conceived the experiments and wrote the paper. W. Ye, H.W., L.B., C.M., W.J., J.Z., J.L., Z.S. and X.H. were primarily responsible for the experiments. S.C., S.L., C.D. and H.Z. performed the lifetime measurements. K. Ling, M.Z., W. Yao, Z.Z. and K.S. conducted the single-crystal measurement and analysis. X.Y., Yanyun Zhang, K. Liu and Yujian Zhang measured the quantum efficiency. A.L., M.G. and H.M. contributed to the time-dependent density functional theory calculations. C.G., Y.M. and Y. Zhou programmed the codes for applications. All authors contributed to data analyses.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Materials thanks Jinsang Kim and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–40, Tables 1–12 and Discussion.
Supplementary Video 1
Blue luminescence and afterglow emission from TSP, TTP, HSM and HPM crystals upon UV excitation.
Supplementary Video 2
A quick response code was printed on a student ID card by inkjet printing, and revealed after removing UV irradiation.
Supplementary Video 3
Afterglow display device, powered by direct current. Numbers gradually disappear within 1 s after the power source is disconnected.
Supplementary Video 4
Afterglow display showing different paths from A to B slowly fading after d.c. excitation.
Supplementary Video 5
Afterglow display used as radar scan operating at various frequencies.
Supplementary Data 1
Crystallographic data for TSP.
Supplementary Data 2
Crystallographic data for DSP.
Supplementary Data 3
Crystallographic data for TPP.
Supplementary Data 4
Crystallographic data for HSM.
Supplementary Data 5
Crystallographic data for HPM.
Supplementary Data 6
Crystallographic data for TNP.
Supplementary Data 7
Crystallographic data for TMP.
Supplementary Data 8
Crystallographic data for diaminopyridine biphenyldicarboxylate (DAB).
Supplementary Data 9
Crystallographic data for diaminopyridine bipyridinedicarboxylate (DAP).
Supplementary Data 10
Crystallographic data for tetrasodium naphthalenetetracarboxylate (TSN).
Supplementary Data 11
Crystallographic data for tetraaminopyridine naphthalenetetracarboxylate (TAN).
Supplementary Data 12
Crystallographic data for tetradimethylaminopyridine naphthalenetetracarboxylate (TDN).
Supplementary Data 13
Crystallographic data for tetraaminopyridine dibromonaphthalenetetracarboxylate (TAB).
Supplementary Data 14
Crystallographic data for tetradimethylaminopyridine dibromonaphthalenetetracarboxylate (TDB)
Supplementary Data 15
Crystallographic data for tetraammonium dibromonaphthalenetetracarboxylate (TNB).
Supplementary Data 16
CheckCIF file for TSP.
Supplementary Data 17
CheckCIF file for DSP.
Supplementary Data 18
CheckCIF file for TPP.
Supplementary Data 19
CheckCIF file for HSM.
Supplementary Data 20
CheckCIF file for HPM.
Supplementary Data 21
CheckCIF file for TNP.
Supplementary Data 22
CheckCIF file for TMP.
Supplementary Data 23
CheckCIF file for DAB.
Supplementary Data 24
CheckCIF file for DAP.
Supplementary Data 25
CheckCIF file for TSN.
Supplementary Data 26
CheckCIF file for TAN.
Supplementary Data 27
CheckCIF file for TDN.
Supplementary Data 28
CheckCIF file for TAB.
Supplementary Data 29
CheckCIF file for TDB.
Supplementary Data 30
CheckCIF file for TNB.
Source data
Source Data Fig. 2
Unprocessed source data used to generate Fig. 2.
Source Data Fig. 3
Unprocessed source data used to generate Fig. 3.
Source Data Fig. 4
Unprocessed source data used to generate Fig. 4.
Rights and permissions
About this article
Cite this article
Ye, W., Ma, H., Shi, H. et al. Confining isolated chromophores for highly efficient blue phosphorescence. Nat. Mater. 20, 1539–1544 (2021). https://doi.org/10.1038/s41563-021-01073-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-021-01073-5
This article is cited by
-
Charge trapping for controllable persistent luminescence in organics
Nature Photonics (2024)
-
Boosting organic phosphorescence in adaptive host-guest materials by hyperconjugation
Nature Communications (2024)
-
Finely manipulating room temperature phosphorescence by dynamic lanthanide coordination toward multi-level information security
Nature Communications (2024)
-
Management of triplet excitons transition: fine regulation of Förster and dexter energy transfer simultaneously
Light: Science & Applications (2024)
-
Nucleic-acid-base photofunctional cocrystal for information security and antimicrobial applications
Nature Communications (2024)