Abstract
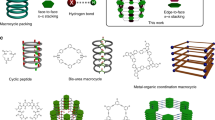
Although the principles of noncovalent bonding are well understood and form the basis for the syntheses of many intricate supramolecular structures, supramolecular noncovalent synthesis cannot yet achieve the levels of precision and complexity that are attainable in organic and/or macromolecular covalent synthesis. Here we show the stepwise synthesis of block supramolecular polymers from metal–porphyrin derivatives (in which the metal centre is Zn, Cu or Ni) functionalized with fluorinated alkyl chains. These monomers first undergo a one-dimensional supramolecular polymerization and cyclization process to form a toroidal structure. Subsequently, successive secondary nucleation, elongation and cyclization steps result in two-dimensional assemblies with concentric toroidal morphologies. The site selectivity endowed by the fluorinated chains, reminiscent of regioselectivity in covalent synthesis, enables the precise control of the compositions and sequences of the supramolecular structures, as demonstrated by the synthesis of several triblock supramolecular terpolymers.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within this paper and its Supplementary Information. Source data are provided with this paper.
References
Whitesides, G. M., Mathias, J. P. & Seto, C. T. Molecular self-assembly and nanochemistry: a chemical strategy for the synthesis of nanostructures. Science 254, 1312–1319 (1991).
Lehn J.-M. Supramolecular Chemistry: Concepts and Perspectives (Wiley, 1995).
Service, R. F. How far can we push chemical self-assembly? Science 309, 95 (2005).
Wehner, M. & Würthner, F. Supramolecular polymerization through kinetic pathway control and living chain growth. Nat. Rev. Chem. 4, 38–53 (2020).
Hartlieb, M., Mansfield, E. D. H. & Perrier, S. A guide to supramolecular polymerizations. Polym. Chem. 11, 1083–1110 (2020).
Ogi, S., Sugiyasu, K., Manna, S., Samitsu, S. & Takeuchi, M. Living supramolecular polymerization realized through a biomimetic approach. Nat. Chem. 6, 188–195 (2014).
Kang, J. et al. A rational strategy for the realization of chain-growth supramolecular polymerization. Science 347, 646–651 (2015).
Ogi, S., Stepanenko, V., Sugiyasu, K., Takeuchi, M. & Würthner, F. Mechanism of self-assembly process and seeded supramolecular polymerization of perylene bisimide organogelator. J. Am. Chem. Soc. 137, 3300–3307 (2015).
Kemper, B. et al. Kinetically controlled stepwise self-assembly of AuI-metallopeptides in water. J. Am. Chem. Soc. 140, 534–537 (2018).
Besenius, P. Controlling supramolecular polymerization through multicomponent self-assembly. J. Polym. Sci., Part A: Polym. Chem. 55, 34–78 (2017).
Vantomme, G. & Meijer, E. W. The construction of supramolecular systems. Science 363, 1396–1397 (2019).
Dong, Y. et al. Multistep molecular and macromolecular assembly for the creation of complex nanostructures. Chem. Phys. Rev. 3, 021305 (2022).
Zhang, W. et al. Supramolecular linear heterojunction composed of graphite-like semiconducting nanotubular segments. Science 334, 340–343 (2011).
Korevaar, P. A. et al. Pathway complexity in supramolecular polymerization. Nature 481, 492–496 (2012).
Adelizzi, B., Van Zee, N. J., de Windt, L. N. J., Palmans, A. R. A. & Meijer, E. W. Future of supramolecular copolymers unveiled by reflecting on covalent copolymerization. J. Am. Chem. Soc. 141, 6110–6121 (2019).
Jung, S. H., Bochicchio, D., Pavan, G. M., Takeuchi, M. & Sugiyasu, K. A block supramolecular polymer and its kinetically enhanced stability. J. Am. Chem. Soc. 140, 10570–10577 (2018).
Wagner, W., Wehner, M., Stepanenko, V. & Würthner, F. Supramolecular block copolymers by seeded living polymerization of perylene bisimides. J. Am. Chem. Soc. 141, 12044–12054 (2019).
Sarkar, A., Sasmal, R., Das, A., Agasti, S. S. & George, S. J. Kinetically controlled synthesis of supramolecular block copolymers with narrow dispersity and tunable block lengths. Chem. Commun. 57, 3937–3940 (2021).
Sakamoto, J., van Heijst, J., Lukin, O. & Schlüter, A. D. Two-dimensional polymers: just a dream of synthetic chemists? Angew. Chem. Int. Ed. 48, 1030–1069 (2009).
Payamyar, P., King, B. T., Öttinger, H. C. & Schlüter, A. D. Two-dimensional polymers: concepts and perspectives. Chem. Commun. 52, 18–34 (2016).
Zheng, Y. et al. Supramolecular thiophene nanosheets. Angew. Chem. Int. Ed. 52, 4845–4848 (2013).
Ghosh, S., Philips, D. S., Saeki, A. & Ajayaghosh, A. Nanosheets of an organic molecular assembly from aqueous medium exhibit high solid-state emission and anisotropic charge-carrier mobility. Adv. Mater. 29, 1605408 (2017).
Lin, Y. et al. Residue-specific solvation-directed thermodynamic and kinetic control over peptide self-assembly with 1D/2D structure selection. ACS Nano 13, 1900–1909 (2019).
Insua, I. & Montenegro, J. 1D to 2D self-assembly of cyclic peptides. J. Am. Chem. Soc. 142, 300–307 (2020).
Fukui, T. et al. Control over differentiation of a metastable supramolecular assembly in one and two dimensions. Nat. Chem. 9, 493–499 (2017).
Sasaki, N., Yuan, J., Fukui, T., Takeuchi, M. & Sugiyasu, K. Control over the aspect ratio of supramolecular nanosheets by molecular design. Chem. Eur. J. 26, 7840–7846 (2020).
Sasaki, N. et al. Supramolecular double-stranded Archimedean spirals and concentric toroids. Nat. Commun. 11, 3578 (2020).
Brendel, J. C. & Schacher, F. H. Block copolymer self-assembly in solution—Quo Vadis? Chem. Asian J. 13, 230–239 (2018).
Karayianni, M. & Pispas, S. Block copolymer solution self-assembly: recent advances, emerging trends, and applications. J. Polym. Sci. 59, 1874–1898 (2021).
Wang, X. et al. Cylindrical block copolymer micelles and co-micelles of controlled length and architecture. Science 317, 644–647 (2007).
Gilroy, J. B. et al. Monodisperse cylindrical micelles by crystallization-driven living self-assembly. Nat. Chem. 2, 566–570 (2010).
Ganda, S. & Stenzel, M. H. Concepts, fabrication methods and applications of living crystallization-driven self-assembly of block copolymers. Prog. Polym. Sci. 101, 101195 (2020).
Rupar, P. A., Chabanne, L., Winnik, M. A. & Manners, I. Non-centrosymmetric cylindrical micelles by unidirectional growth. Science 337, 559–562 (2012).
Hudson, Z. M., Lunn, D. J., Winnik, M. A. & Manners, I. Colour-tunable fluorescent multiblock micelles. Nat. Commun. 5, 3372 (2014).
Qiu, H. et al. Uniform patchy and hollow rectangular platelet micelles from crystallizable polymer blends. Science 352, 697–701 (2016).
Merg, A. D. et al. Seeded heteroepitaxial growth of crystallizable collagen triple helices: engineering multifunctional two-dimensional core-shell nanostructures. J. Am. Chem. Soc. 141, 20107–20117 (2019).
Yang, S., Kang, S.-Y. & Choi, T.-L. Semi-conducting 2D rectangles with tunable length via uniaxial living crystallization-driven self-assembly of homopolymer. Nat. Commun. 12, 2602 (2021).
Kitamoto, Y. et al. One-shot preparation of topologically chimeric nanofibers via a gradient supramolecular copolymerization. Nat. Commun. 10, 4578 (2019).
Li, Y. et al. Corner-, edge-, and facet-controlled growth of nanocrystals. Sci. Adv. 7, eabf1410 (2021).
Datta, S. et al. Self-assembled poly-catenanes from supramolecular toroidal building blocks. Nature 583, 400–405 (2020).
Datta, S., Takahashi, S. & Yagai, S. Nanoengineering of curved supramolecular polymers: toward single-chain mesoscale materials. Acc. Mater. Res. 3, 259–271 (2022).
Pearce, A. K., Wilks, T. R., Arno, M. C. & O’Reilly, R. K. Synthesis and applications of anisotropic nanoparticles with precisely defined dimensions. Nat. Rev. Chem. 5, 21–45 (2021).
MacFarlane, L. R. et al. Functional nanoparticles through π-conjugated polymer self-assembly. Nat. Rev. Mater. 6, 7–26 (2021).
Helmich, F. et al. Dilution-induced self-assembly of porphyrin aggregates: a consequence of coupled equilibria. Angew. Chem. Int. Ed. 49, 3939–3942 (2010).
Hirose, T., Helmich, F. & Meijer, E. W. Photocontrol over cooperative porphyrin self-assembly with phenylazopyridine ligands. Angew. Chem. Int. Ed. 52, 304–309 (2013).
Cremers, J. et al. Nanorings with copper(II) and zinc(II) centers: forcing copper porphyrins to bind axial ligands in heterometallated oligomers. Chem. Sci. 7, 6961–6968 (2016).
Thies, S. et al. Coordination-induced spin crossover (CISCO) through axial bonding of substituted pyridines to nickel-porphyrins: σ-donor versus π–acceptor effects. Chem. Eur. J. 16, 10074–10083 (2010).
Ando, T., Uchihashi, T. & Scheuring, S. Filming biomolecular processes by high-speed atomic force microscopy. Chem. Rev. 114, 3120–3188 (2014).
Fukui, T. et al. Direct observation and manipulation of supramolecular polymerization by high-speed atomic force microscopy. Angew. Chem. Int. Ed. 57, 15465–15470 (2018).
Bochicchio, D., Salvalaglio, M. & Pavan, G. M. Into the dynamics of a supramolecular polymer at submolecular resolution. Nat. Commun. 8, 147 (2017).
Yamamoto, K., Higuchi, M., Shiki, S., Tsuruta, M. & Chiba, H. Stepwise radial complexation of imine groups in phenylazomethine dendrimers. Nature 415, 509–511 (2002).
Rousseaux, S. A. L. et al. Self-assembly of Russian doll concentric porphyrin nanorings. J. Am. Chem. Soc. 137, 12713–12718 (2015).
Kondratuk, D. V. et al. Supramolecular nesting of cyclic polymers. Nat. Chem. 7, 317–322 (2015).
Acknowledgements
This work was supported by Ministry of Education, Culture, Sports, Science and Technology (MEXT): Grants-in-Aid for Scientific Research (JP19K05592 and JP22H02134 for K.S.), a Grant-in-Aid for Scientific Research on Innovative Areas ‘Soft Crystals’ (20H04682 for K.S.; 20H04669 for T.U.) and a Grant-in-Aid for Transformative Research Areas (A) ‘Condensed Conjugation’ (JP20H05868 for M.T.), and Data Creation and Utilization-Type Material Research and Development Project (JPMXP1122714694). Financial support from the Izumi Science and Technology Foundation, the Iketani Science and Technology Foundation, the Murata Science Foundation, the Sekisui Chemical Grant Program and the Mitsubishi Foundation is also acknowledged.
Author information
Authors and Affiliations
Contributions
N.S. and K.S. conceived the project. N.S. and H.I. synthesized the molecules and investigated their self-assembly. J.K. conducted the STEM measurements. Y.I. and T.U. performed the high-speed AFM measurements. All of the authors discussed the results. N.S. and K.S. wrote the manuscript with input from all of the authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Subi George and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–33 and Tables 1–4.
Supplementary Video 1
Real-time observation of depolymerization of SCTs induced by DMAP using high-speed AFM.
Supplementary Data 1
Source data for Supplementary Fig. 14.
Supplementary Data 2
Source data for Supplementary Fig. 15.
Supplementary Data 3
Source data for Supplementary Fig. 16.
Supplementary Data 4
Source data for Supplementary Fig. 17.
Supplementary Data 5
Source data for Supplementary Fig. 18.
Supplementary Data 6
Source data for Supplementary Fig. 19.
Supplementary Data 7
Source data for Supplementary Fig. 20.
Supplementary Data 8
Source data for Supplementary Fig. 21.
Supplementary Data 9
Source data for Supplementary Fig. 22.
Supplementary Data 10
Source data for Supplementary Fig. 23.
Supplementary Data 11
Source data for Supplementary Fig. 24.
Supplementary Data 12
Source data for Supplementary Fig. 25.
Supplementary Data 13
Source data for Supplementary Fig. 26.
Supplementary Data 14
Source data for Supplementary Fig. 27.
Supplementary Data 15
Source data for Supplementary Fig. 29.
Supplementary Data 16
Source data for Supplementary Fig. 30.
Supplementary Data 17
Source data for Supplementary Fig. 31.
Supplementary Data 18
Source data for Supplementary Fig. 33.
Source data
Source Data Fig. 3
Time-dependent absorption spectral changes of SCTs. Statistical source data of external circumference, internal circumference and area of SCTs.
Source Data Fig. 4
Elemental mapping profiles obtained across the ABC type and ACB type triblock SCTs.
Source Data Fig. 5
Changes in the absorbance observed during the multistep noncovalent synthesis.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sasaki, N., Kikkawa, J., Ishii, Y. et al. Multistep, site-selective noncovalent synthesis of two-dimensional block supramolecular polymers. Nat. Chem. 15, 922–929 (2023). https://doi.org/10.1038/s41557-023-01216-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-023-01216-y
This article is cited by
-
Noncovalent synthesis of homo and hetero-architectures of supramolecular polymers via secondary nucleation
Nature Communications (2024)
-
Site selectivity steps in
Nature Chemistry (2023)