Abstract
As one of the promising next-generation probiotics (NGPs), Akkermansia muciniphila, a well-known mucin-degrading bacterium, has been proven to be closely related to the metabolic diseases of its human host. However, the role of A. muciniphila in the host’s intestinal health remains ambiguous. Here, we comprehensively summarize and discuss the characteristics, the distribution, and the colonization of A. muciniphila in the human gastrointestinal tract (GIT). We propose that the application of A. muciniphila as a biomarker for longevity, for diagnostics and prognostics of intestinal diseases, or for intestinal health should be cautiously considered. Precise dietary regulation can mediate the treatment of intestinal diseases by altering the abundance of A. muciniphila. Although the beneficial role of A. muciniphila and its component in intestinal inflammation has been discovered, in gnotobiotic mice with specific gut microbiota, certain genotype, and colorectal cancer, or in animal models infected with a specific pathogen, A. muciniphila may be related to the occurrence and development of intestinal diseases. Genomic analysis, emphasizing the strain-level phylogenetic differences of A. muciniphila, indicates that a clear description and discussion of each strain is critical before its practical application. Our review provides much needed insight for the precise application of A. muciniphila.
Similar content being viewed by others
Introduction
As a mucin utilizing specialist1, Akkermansia muciniphila has been highly considered as one of the next-generation probiotics (NGPs) and is regarded to play an important role in the maintenance of the intestinal epithelial barrier. A typical cycle of intestinal inflammation is driven by abnormal interactions among genetic risk factors, environmental triggers (microbiota), modifiers, and the host’s immune system2. Akkermansia muciniphila widely exists in the GIT of multiple animals including humans, mice3, cattle4, guinea pigs5, swine6, rabbits7, ostriches8 and chickens9. In infants and healthy adults, A. muciniphila can account for 1~3% of total fecal cells10, during which the excessive degradation of mucin allows pathogens to invade the sloughed intestinal mucosa11. In such cases, supplement with adequate numbers of A. muciniphila, or heat-killed A. muciniphila may safely improve the intestinal barrier in obese humans12 and mice fed high-fat diets13,14. However, an excessive enrichment of A. muciniphila in mice with a specific intestinal environment may lead to the aggravation of intestinal inflammation caused by epithelial barrier damage15,16,17. Although the effect of A. muciniphila on intestinal inflammation has been gradually studied, how it works is still unclear. Meanwhile, factors including host, intestinal segmentation, age, intestinal disease, and diet, affecting the distribution of A. muciniphila in the GIT and how A. muciniphila interacts with the host to maintain intestinal health is mainly unknown. In this review, we bring together the latest research to comprehensively discuss the potential of A. muciniphila as a NGP to intervene in the intestinal homeostasis in humans and animals.
The characteristics and safety of A. muciniphila in the GIT
Belonging to the phylum Verrucomicrobia, A. muciniphila has been described as an oval-shaped, non-mobile, Gram-negative, non-spore forming, and strictly anaerobic bacterium. However, more than 90% number of A. muciniphila ATCC BAA-835 can survive in 95% oxygen and 5% CO2 for 1 h18. Different strains and phylogroups of A. muciniphila differ in their sensitivity to oxygen19, and most of the known A. muciniphila strains can utilize mucin as the sole carbon and nitrogen sources. The bacterium can grow on the Brain Heart Infusion (BHI) and Columbia medium, and mucin-derived monosaccharides, such as fucose, galactose, and N-acetylglucosamine, can also be used by A. muciniphila as growth substrates20.
The complete genome of type strain, A. muciniphila ATCC BAA-835, is 2,664,102 bp long, and has 2,176 predicted protein-coding genes, which suggest it can metabolize different kinds of carbohydrates and mucin21. Phylogenetic analysis of A. muciniphila classified it into three22 or four23 species-level phylogroups. Akkermansia muciniphila MucT strain is resistant to several antibiotics, such as chloramphenicol, clindamycin, streptomycin, erythromycin, vancomycin, and metronidazole24,25. The MucT strain is also abundantly colonized in the GIT of individuals treated with broad-spectrum antibiotics25, which may be due to the fact that A. muciniphila is an open-pangenome microorganism that can continually acquire genes from other bacteria via lateral gene transfer22.
Nowadays, A. muciniphila is widely studied as a promising probiotic to improve metabolic syndrome and obesity. However, its safety and toxicity are a growing concern. Long-term oral high-dosage of A. muciniphila, or pasteurized A. muciniphila (1010 bacteria per day), are safe and well tolerated in overweight and obese individuals12. The bacterial reverse mutation, in vitro mammalian cell micronucleus test, and a subchronic toxicity test (lasting 90 days in rat), show that pasteurized A. muciniphila has no-adverse effects26. Recently, pasteurized A. muciniphila has been recognized as a new food by the European Union27. Based on these findings and policies, the utilization of A. muciniphila in metabolic syndrome and in healthy individuals may be safe. However, whether A. muciniphila treatment is safe, when intestinal diseases occur, still needs to be confirmed.
The colonization and abundance of A. muciniphila in GIT
Location-dependent colonization of A. muciniphila in the GIT
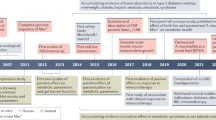
The abundance of A. muciniphila in the GIT seems to be location-dependent. Bacteria, belonging to the phylum Verrucomicrobia, not specified to Akkermansia, can be detected in human duodenal biopsies (0.0688%) and mucus (0.0387%)28. Akkermansia species, with an average relative abundance of 0.01%, are also found in the jejunal content of humans29, but the abundance of Verrucomicrobia-related bacteria can make up 5% of the bacteria density in the distal ileum of humans, and as much as 6% and 9% in the ascending colon and rectum mucosal biopsies, respectively30. Compared to the small intestine, the passage time of chyme is much longer (9–46 h)31 and the mucosal layer is thicker in the large intestine, which is presumed to provide multiple substrates for A. muciniphila32. As a mucin-degrading bacterium, A. muciniphila is abundantly found in the mucin-rich intra-intestinal location33, for which it is positively correlated with the concentration of mucin34. For instance, inoculated A. muciniphila is found to efficiently colonize (13.08% of total microbes) in the caecum of chickens9. In humans, there are approximate 1.45 × 104 cells of A. muciniphila per gram of ascending, or sigmoid colonic mucosal biopsies35. Moreover, the proportion of Akkermanisa in the lumen (0.57%) is found higher than that in colonic mucosa of healthy individuals (0.21%)36. The pH value may be another factor affecting the distribution of A. muciniphila in different intestinal segments. The pH value of the small and large intestine is 6.6~7.5 and 6.4~7.0, respectively37 (Fig. 1a). Using a model of the human digestive system, Simulator of the Human Intestinal Microbial Ecosystem (SHIME), when the pH value of the distal colon is 6.6~6.9, the abundance of A. muciniphila is at its highest38.
a The distribution of A. muciniphila along the GIT (small and large intestine) and in the lumen and mucosal layer. b The schematic diagram of A. muciniphila abundance changing with age in the human and mouse GIT. c The mechanisms of A. muciniphila ameliorating aging in mice. All figures are created with Biorender.com.
Aging-dependent colonization of A. muciniphila in GIT
Tracking the fecal bacterial composition in 98 infants from birth to 12 months old shows a gradual increase (0% to 0.57%) in the relative abundance of A. muciniphila39. Similar increase (0.14% to 4.25%) is found in children aged from 1 to 4 years40. In addition, the abundance of A. muciniphila is high in long-living Chinese people (≥90 years old)41. Moreover, the abundance of A. muciniphila is especially higher in the gut of older populations aged from 105 to 109 years old, compared to other age groups42 (Fig. 1b). These findings43,44,45,46 (Table 1) give rise to the consideration of exogenous A. muciniphila inoculation to alleviate the negative effects of aging47,48 (Fig. 1c). However, opposite results are found in studies using rodents. The abundance of Akkermansia appears to be decreaed in aged mice or rats3,49,50. Although A. muciniphila may be a potential biomarker of longevity in humans, mice may not be a natural research model to study this relationship in humans.
Factors influencing the colonization and abundance of A. muciniphila in the GIT
The abundance of A. muciniphila related to different intestinal diseases
Inflammatory bowel disease (IBD), including Crohn’s disease (CD) and ulcerative colitis (UC), is a known risk factor for the development of colorectal cancer (CRC), like colitis-associated colorectal cancer (CAC)51, the third leading cause of cancer-related death in humans52. The number of A. muciniphila in healthy individuals is higher than that in IBD patients53,54 (Supplementary Table 1), especially in the hindgut55. The relative abundance of A. muciniphila can be as high as 2.9% in healthy populations, but is found to sharply decline in noninflamed UC (0.03%), inflamed UC (0.02%), noninflamed CD (0.62%), and inflamed CD (0.20%) patients56. Moreover, A. muciniphila are more abundant in CD patients than in UC patients54,56.
However, the higher abundance of A. muciniphila may not be negatively correlated with IBD. A surprising result shows in both CRC patients and CRC mice, the abundance of A. muciniphila is higher than that in healthy people57,58 (Supplementary Table 1). Moreover, A. muciniphila is enriched in the early stage of CRC59. The abundance of A. muciniphila may also be increased by pathogenic infection60,61 (Supplementary Table 1).
Diet and lifestyle can regulate the abundance of A. muciniphila
Diet is an important factor that cannot be ignored to shape the gut microbiota62,63. We summarized previous studies and focused on the relationship between the abundance of A. muciniphila and dietary ingredients, which are associated with host health and intestinal diseases. The high-concentration of cellulose in the diet can relieve the inflammation of dextran sodium sulfate (DSS)-induced mice, while increasing the abundance of A. muciniphila64. A diet enriched with rye bran and wheat aleurone is reported to increase the relative abundance of Akkermansia in C57BL/6 J mice, accompanied by changes in glycine betaine metabolism65. Both sugarcane bagasse, a water-soluble fiber, and xylo-oligosaccharide can also increase the abundance of Akkermansia in Fischer 344 rats66. Milk and its products, for example, breast milk can promote the growth of A. muciniphila in mice transplanted with microbiota from infant67, which may be triggered by galacto-N-biose68. Another study revealed that the consumption of cheese is negatively associated with the abundance of A. muciniphila69. The increase of A. muciniphila by dietary supplement of polyphenol containing grape proanthocyanidin, chlorogenic acid, and resveratrol is accompanied by the improvement of metabolic profile and anti-inflammatory activities of host, especially in mice with DSS-induced colitis70,71,72. Interestingly, grape proanthocyanidin may indirectly induce the intestinal bloom of A. muciniphila, in vivo, in mice, but shows no effect on the quantity of A. muciniphila in vitro70. Probiotics, such as Lactobacillus fermentum and Bacillus subtilis, can alleviate DSS-induced colits in mice and increase the abundance of Akkermansia73,74. In contrast, other probiotics, such as Bifidobacteria adolescentis, is found to inhibit the excessive growth of A. muciniphila during the therapy of DSS-induced chronic colitis75. Similarly, Pediococcus pentosaceus and Lactobacillus coryniformis can ameliorate CRC in mice via regulating gut microbiota, including increasing the abundance of A. muciniphila76,77. Particular dietary patterns, such as low-calorie diet, ketogenic diet, and fasting, are reported to increase the abundance of A. muciniphila in healthy individuals, or IBD patients78,79,80,81. It is worth noting that gut microbial composition can be influenced by many factors, especially stool consistency and fecal transit time, which are closely connected with the abundance of A. muciniphila82,83. To summarize, A. muciniphila may participate in the effect of diet on IBD, but whether the change of A. muciniphila abundance is the cause, or result, remains to be determined.
A. muciniphila and intestinal homeostasis of host
A. muciniphila and the intestinal physical barrier of host
Live A. muciniphila bacteria have been repeatedly confirmed to be related to the improvement of the intestinal barrier. Oral gavage with live A. muciniphila can increase the expression of tight junction proteins (TJs), such as zonula occludens (ZO-1) and occludin, in DSS-induced mice84. In vitro, active A. muciniphila bacteria are also found to increase the transepithelial electrical resistance (TER), a recognized parameter to reflect the cell integrity of the cell membrane85 of cocultured Caco-2 cells after 24 or 48 h18,86. Particularly, some cellular components of A. muciniphila have also been shown to improve the intestinal permeability. One of them is extracellular vesicles (AmEVs), the lipid bilayer secreted by A. muciniphila. Compared to obese mice induced by high-fat diet, or lipopolysaccharide (LPS)-induced Caco-2 cell, the expression of occludin, ZO-1, and claudin-5 is enhanced (in vivo and in vitro) by activating the adenosine monophosphate (AMP)-activated protein kinase (AMPK) pathway in a dose-dependent manner with oral administration of 10 μg AmEVs87. Moreover, after pasteurization88, a stable outer membrane of A. muciniphila, Amuc_1100, has been shown to increase the TER in vitro86 and the expression of TJ genes in the small intestine of obese mice induced by high-fat diet in vivo14. Amuc-1100 belongs to a gene cluster related to the formation of pilus86 and was recently used in mice with metabolic and intestinal diseases14,88.
As a mucin-specialist, the abundance of A. muciniphila is closely related to the thickness of the intestinal mucosa. A similar result is found in Apoe−/− mice fed western-diet89. Goblet cells, a specialized epithelial cell that secretes mucins, have attracted much attention because of their important role in maintaining the integrity of the inner mucus layer90. A gavage with 1.0 × 108 CFU/day of A. muciniphila (DSM 22959) can increase the density of goblet cells in the ileum of mice with a long-term feeding of high-fat diet91. Similarly, A. muciniphila bacteria are believed to increase the number of goblet cells and up-regulate the expression of Mucin 2 (MUC2) and trefoil factor 2 (Tff2) in Salmonella pullorum-infected chickens92. A genome-wide association study (GWAS) based on 288 pigs revealed a correlation between the relative abundance of A. muciniphila and a gene encoding carbohydrate sulfotransferase 1293, a required gene for the biosynthesis of glycosaminoglycan and the formation of mucin94,95. It should be highlighted that the genome of A. muciniphila (ATCC BAA-835) lacks mucus-binding domains21, which is verified by an in vitro study that A. muciniphila can barely adhere to the mucus18. These results describe the protective effect of A. muciniphila on intestinal mucosa, which may be related to the increase of goblet cells (Fig. 2).
A. muciniphila and the intestinal immunity of the host
The intestinal inflammation involves the complex interaction of host genes, host immunity, microbiota and environmental factors (Fig. 3a). As a mucin-degrader in the gut, A. muciniphila can easily induce the immune response of the host due to its frequent communication with intestinal epithelial cells (IECs) (Fig. 3b and c). For instance, A. muciniphila increases the expression of genes encoding 2-oleoylglycerol, 2-arachidonoylglycerol and 2-palmitoylglycerol in the ileum of mice13, which are associated with the endocannabinoid system involving intestinal homeostasis and improved intestinal barriers96. When A. muciniphila is present in the intestine of specified pathogen free (SPF) mice, T cells response to A. muciniphila are localized to the Peyer’s patches (PPs), large intestine, small intestine lamina propria and mesenteric lymph nodes (mLNs), which is regulated by the outer membrane proteins Amuc_RS03735 and Amuc_RS0374097. In mice with oral treatment of live A. muciniphila, the differentiation of peripheral regulatory T cells (pTregs), the proliferation of residual thymus-derived Tregs (tTregs) in the colon (which reprogramed by epitope 2 C.1 from A. muciniphila98), and the differentiation of Foxp3+ Treg from CD4+ T cells in MLNs are found to be promoted99. Akkermansia muciniphila is also found to be positively correlated with TLR4 receptor and against TLR4−/− induced colitis in mice by increasing the proportion of RORγt+ Treg cells that enhances the immune response100. Whereas, in altered Schaedler flora (ASF) mice, the treatment of A. muciniphila specifically impacted the number of T follicular helper (TFH) cells only in the Peyer’s patches (PPs)97. As the TFH cells are important for the secretion of immunoglobulins (e.g. IgA), the variation in the quantity of these cells may help to slow down the advanced-stage intestinal inflammation101. Besides the proliferation, the development of immune cells is also involved in the abundance of A. muciniphila. In addition, both pasteurised A. muciniphila and Amuc_1100 can decrease the colonic infiltration of CD8+ cytotoxic T lymphocytes (CTLs), which aggravates colitis by mediating the production of cytokines102,103, and can suppress the proliferation of proinflammatory CD16/32+ macrophages in the MLNs and decrease the mRNA level of pro-inflammatory cytokines in mice with DSS-induced colitis88. In a mice model with CRC, pasteurised A. muciniphila and Amuc_1100 increased the activation of CTLs in the MLN and the proportion of tumor necrosis factor-alpha (TNF-α)+ CTLs to promote the apoptosis of tumor cells. Meanwhile, the proportion of PD-1+ CTLs in MLN can be decreased to suppress the growth of tumor88. Another protein of A. muciniphila, Amuc_1434, an aspartic protease can degrade MUC2 in vitro104, can inhibit the proliferation of LS174T cells and block the G0/G1 phase of cell cycle of LS174T cells by increasing the expression of tumor protein 53 (p53) in vitro105. Further, Amuc_1434* treatment promotes the apoptosis of LS174T cells and increases the level of mitochondrial reactive oxygen species (ROS) by upregulating tumor-necrosis-factor-related apoptosis-inducing ligand (TRAIL)105. The concentration of inflammatory cytokines can be used as an important indicator to assess the severity of intestinal inflammation. The pretreatment of A. muciniphila was found to suppress the expression of pro-inflammatory cytokines, such as interferon gamma (IFN-γ), interleukin-17 (IL-17), TNF-α, interleukin-1beta (IL-1β) and nitric oxide synthase 2 (NOS2), in the colon of mice with DSS-induced colitis106. Similarly, the mRNA level of pro-inflammatory cytokines, TNF-α, IFN-γ, IL-1β, IL-6, IL-18 and IL-33, in the colon of mice with DSS-induced colitis can be also decreased by the treatment of pasteurised A. muciniphila (1.5×108 CFU) or 3 µg of Amuc_110088. In vitro, the level of IL-6 in colonic epithelial cells (CT26), challenged by E. coli-derived extracellular vesicle, can be reduced by the pre-treatment of AmEVs in a dose-dependent manner107. Adiacyl phosphatidylethanolamine, with two branched chains (a15:0-i15:0 PE), isolated from A. muciniphila can cause the release of specific inflammatory cytokines by acting on the non-classical TLR2-TLR1 heterodimer, and at low doses, can blunt the activation threshold of immune cells108.
The production of antigen-specific T cell-dependent IgA and IgG1 in the serum of ASF mice is reported to be induced by acquiring A. muciniphila vertically from mothers97. Live A. muciniphila bacteria markedly increases the expression of regenerating islet derived 3-gamma (Reg3g)13, a lectin protecting the intestinal mucosa against the invasion of pathogens109, in the colon of mice fed high-fat diet. In contrast, both live and pasteurized A. muciniphila improved the expression of lysozyme C-1 (Lyz1) in the small intestine of obese mice induced by high-fat diet14 (Fig. 3b and c).
The interaction between A. muciniphila and the intestinal epithelium
A few studies suggest a direct effect of A. muciniphila on IECs. A linear discriminate analysis clearly shows the enrichment of A. muciniphila in the early regenerative mucosa of mice. The intrarectal administration of active A. muciniphila remarkably facilitate the closure of injured mucosa (from 43.7% to 74.14%) in mice by promoting the proliferation and migration of intestinal stem cells (ISCs) and accelerating the regeneration of the wound in SK-CO15 monolayers in vitro110. This requires the participation of formyl peptide receptor 1 (FPR1) and neutrophilic NADPH oxidase (NOX1) to increase ROS in the wound edge and the phosphorylation of extracellular-signal-regulated kinase (ERK) in colonic epithelial cells. In addition, the gavage of AmEVs isolated from A. muciniphila can alleviate dysplasia in C57BL/6 mice induced by 2% DSS107. Amuc_1100 (3 μg) can also relieve the shortening of colon and the histological injuries in the proximal colon in mice with DSS-induced colitis88, indicating an alleviation or even the repair of injured intestinal epithelium by A. muciniphila, or its derivatives.
The steady renewal of the IECs is fueled by ISCs lying at the basilar part of crypts111, which is particularly important in case of disrupted intestinal homeostasis. The colonization of A. muciniphila in the chicken colon is found to regulate the proliferation of ISCs though the classical Wnt/β-catenin signaling pathway92. In addition, A. muciniphila can closely bind to laminin18, one of the important components of extracellular matrix which can regulate the migration, differentiation and anti-inflammatory responses of IECs112,113,114. A GWAS based study showed a strong connection between laminin β1 chain encoding gene and the susceptibility of UC115, and showed the laminin γ1 chain encoding gene as a susceptible locus of IBD116. However, the interaction between A. muciniphila and laminin is still poorly understood. Therefore, as a bacterium that is directly communicated with intestinal mucosa, A. muciniphila displays an intervention in the proliferation and/or differentiation of IECs and ISCs, which represents a very complex cross-talk to be further discussed.
Relationship between A. muciniphila and other intestinal bacteria during intestinal inflammation
Although A. muciniphila is found to negatively correlate with total mucin-degrading bacteria, its decreased number may result in the proliferation of mucin-associated bacteria when intestinal inflammation occurs56. This can reduce the degradation of mucus and maintain a relatively stable intestinal barrier56. Several studies provide direct evidence for such interaction between A. muciniphila and other mucosa-associated bacteria. When cocultured with mucolytic bacteria like Bacteroides vulgatus, Ruminococcus gnavus, or Ruminococcus torques, in a defined medium with MUC2 as sole carbon source, the growth of A. muciniphila is inhibited while the growth of other bacteria is promoted56,117. On the other hand, A. muciniphila may influence the intestinal microbiota by regulating the intestinal immunity of the host13,118. Akkermansia muciniphila treatment accelerates the normalization of the microbial community in mice with colitis, and reverses the decreased ratio of Firmicutes/Bacteroidetes bacteria in the cecum caused by high-fat diet119. A correlation between the abundance of A. muciniphila and Faecalibacterium prausnitzii is also confirmed in the feces of CD patients120. Moreover, six genera (Prevotella, Sutterella, Klebsiella, Dorea, Parabacteroides, and Akkermansia) are found to flourish in CD patients with remission121. Furthermore, both A. muciniphila-F. prausnitzii, and A. muciniphila-Bacteroides thetaiotaomicron, in IBD patients, are lower than in healthy individuals54, suggesting a relationship between mutualistic symbiosis of mucolytic bacteria and IBD.
The negative effect of A. muciniphila in specific GIT environment
In several cases, A. muciniphila may have a negative impact on intestinal health (Table 2). Specifically, in a gnotobiotic C3H mouse model with eight bacterial species normally found in humans, the infection of Salmonella typhimurium with the pro-colonization of A. muciniphila makes the former a dominant bacterium in this limited microbiota accompanied by more severe intestinal inflammation16. Another study shows that A. muciniphila is able to induce colitis in specific-pathogen-free and germ-free Il10−/− mice and its colonization is mediated by Nod-like receptor 617. Low-fiber diet promotes expansion of A. muciniphila and other mucus-degrading bacteria in mice colonizing with a synthetic human gut microbiota, which promotes the degradation of the mucus layer and increases the colitis caused by Citrobacter rodentium infection15. In CRC mice transplanted with the fecal microbiota from CRC patients, Akkermansia bacteria are positively correlated with increased tumor burden122. In addition, gavage of A. muciniphila into intestine-specific Apc mutant mice (FabplCre; Apc15lox/+) aggravates the development of colorectal cancer by increasing the number of tumors123. In conclusion, A. muciniphila may be at risk of exacerbating pathogenic infections and inflammation of intestine, which is a common problem to be considered in mucin-degrading bacteria124.
The inspiration of precise application: strain-specific role of A. muciniphila on host intestinal health associated with its genetic and phenotypic properties
The role of probiotics largely depends on the bacterial strains used, which is essential for their clinical effects125. Different bacterial strains have distinct genomic homology leading to discrepant function126,127, which makes it reasonable to consider the practical application of different strains. A total of 106 A. muciniphila metagenome-assembled genomes (MAGs) have been reconstructed based on the available metagenomic datasets of human, mouse and pig, which revealed three phylogroups of A. muciniphila, AmI, AmII and AmIII with different relative abundance22. Based on the whole-genome shotgun sequencing of 39 isolates of A. muciniphila, from human and mouse feces, three A. muciniphila phylogroups (AmI, AmII and AmIII) are identified and the functional annotation shows their distinct metabolic and functional features22. The comparative genomic analysis based on 35 metagenome-assembled genomes (MAGs) and 40 publicly available genomes further reveals at least four phylogroups of A. muciniphila (AmI to AmIV) and some strains in specific phylogroup have the genes and ability to vitamin B12 biosynthesis23. A study including genomic analysis and phenotypic test shows distinct characteristics of these phylogroups, including oxygen tolerance, cell adherence, the activation of toll-like receptor 2, sulfur acquisition and the colonization of the bacterium in GIT19. A large-scale metagenomic-based genomic analysis further confirms that the genomic difference may diversify the effect of A. muciniphila strains on host health128,129, and results of in vivo and in vitro studies support this hypothesis. In mice with chronic colitis, A. muciniphila strain ATCC 835 presents better anti-inflammatory properties than strain 13999. Of 11 human-derived A. muciniphila strains, only the supernatant from a culture of the AK32 strain can increase the size of small intestine-derived organoids in vitro130. It can be assumed that the function of different A. muciniphila strains may be various, possibly due to the diversity in their cellular components and metabolites, although most related studies focus on A. muciniphila ATCC 835. Moreover, function-specific component of different A. muciniphila strains, or their metabolites may be mass produced or recombined to investigate and reveal the effects and mechanism of A. muciniphila targeting diseases (Fig. 4). Based on the understanding of functional characterization of A. muciniphila strains, studies on the phenotypes of A. muciniphila in vitro and its effect on the host are required for the precise application of A. muciniphila in disease treatment.
In summary, regardless of host animal species, A. muciniphila is found to be more abundant in the hindgut. The abundance of A. muciniphila in the human GIT increases with age, which is contrary to that in mice. Types of intestinal diseases, dietary supplements, as well as other mucus-associated microbes can influence the abundance of A. muciniphila, but cautious consideration should be given to A. muciniphila as a biomarker for indicating an intestinal health risk. Akkermansia muciniphila may safety be administered in healthy individuals or those with metabolic syndrome (excess fat around the waist, high blood sugar, increased blood pressure, and abnormal cholesterol levels). Akkermansia muciniphila may also be beneficial to the maintenance of intestinal homeostasis of the host. However, in some cases, such as the lack of dietary fiber, pathogenic infection, or specific host genotypes, the accumulation of A. muciniphila in the GIT may exacerbate the damage of the intestinal epithelium, indicating that A. muciniphila may have a double-edged effect on the intestinal health of the host. In view of the strain-specific genome and phenotype of A. muciniphila, a clear description and discussion of each strain is critical before its practical application.
References
Derrien, M., Vaughan, E. E., Plugge, C. M. & de Vos, W. M. Akkermansia muciniphila gen. nov., sp. nov., a human intestinal mucin-degrading bacterium. Int. J. Syst. Evolut. Microbiol. 54, 1469–1476 (2004).
Sheehan, D. & Shanahan, F. The Gut Microbiota in Inflammatory Bowel Disease. Gastroenterol. Clin. North Am. 46, 143–154 (2017).
Alam, M. S., Gangiredla, J., Hasan, N. A., Barnaba, T. & Tartera, C. Aging-Induced Dysbiosis of Gut Microbiota as a Risk Factor for Increased Listeria monocytogenes Infection. Front. Immunol. 12, 672353 (2021).
Dowd, S. E. et al. Evaluation of the bacterial diversity in the feces of cattle using 16S rDNA bacterial tag-encoded FLX amplicon pyrosequencing (bTEFAP. 8, 125 (2008). BMC Microbiol. 8, 125 (2008).
Hildebrand, F. et al. A comparative analysis of the intestinal metagenomes present in guinea pigs (Cavia porcellus) and humans (Homo sapiens). BMC Genomics 13, 514 (2012).
McCormack, U. M. et al. Exploring a Possible Link between the Intestinal Microbiota and Feed Efficiency in Pigs. Appl. Environ. Microbiol. 83, e00380–17 (2017).
Fang, S. et al. Faecal microbiota and functional capacity associated with weaning weight in meat rabbits. Microb. Biotechnol. 12, 1441–1452 (2019).
Videvall, E. et al. Major shifts in gut microbiota during development and its relationship to growth in ostriches. Mol. Ecol. 28, 2653–2667 (2019).
Kubasova, T. et al. Gut Anaerobes Capable of Chicken Caecum Colonisation. Microorganisms 7, 597 (2019).
Derrien, M., Collado, M. C., Ben-Amor, K., Salminen, S. & de Vos, W. M. The Mucin degrader Akkermansia muciniphila is an abundant resident of the human intestinal tract. Appl. Environ. Microbiol. 74, 1646–1648 (2008).
Lindén, S. K., Florin, T. H. J. & McGuckin, M. A. Mucin dynamics in intestinal bacterial infection. PloS one 3, e3952 (2008).
Depommier, C. et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat. Med. 25, 1096–1103 (2019).
Everard, A. et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA 110, 9066–9071 (2013).
Plovier, H. et al. A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice. Nat. Med. 23, 107–113 (2017).
Desai, M. S. et al. A Dietary Fiber-Deprived Gut Microbiota Degrades the Colonic Mucus Barrier and Enhances Pathogen Susceptibility. Cell 167, 1339–1353 (2016).
Ganesh, B. P., Klopfleisch, R., Loh, G. & Blaut, M. Commensal Akkermansia muciniphila exacerbates gut inflammation in Salmonella Typhimurium-infected gnotobiotic mice. PloS one 8, e74963 (2013).
Seregin, S. S. et al. NLRP6 Protects Il10−/− Mice from Colitis by Limiting Colonization of Akkermansia muciniphila. Cell Rep. 19, 733–745 (2017).
Reunanen, J. et al. Akkermansia muciniphila Adheres to Enterocytes and Strengthens the Integrity of the Epithelial Cell Layer. Appl. Environ. Microbiol. 81, 3655–3662 (2015).
Becken, B. et al. Genotypic and Phenotypic Diversity among Human Isolates of Akkermansia muciniphila. mBio 12, e00478–21 (2021).
Ottman, N. et al. Genome-Scale Model and Omics Analysis of Metabolic Capacities of Akkermansia muciniphila Reveal a Preferential Mucin-Degrading Lifestyle. Appl. Environ. Microbiol. 83, e01014–e01017 (2017).
van Passel, M. W. J. et al. The genome of Akkermansia muciniphila, a dedicated intestinal mucin degrader, and its use in exploring intestinal metagenomes. PloS one 6, e16876 (2011).
Guo, X. et al. Genome sequencing of 39 Akkermansia muciniphila isolates reveals its population structure, genomic and functional diverisity, and global distribution in mammalian gut microbiotas. BMC genomics 18, 800 (2017).
Kirmiz, N. et al. Comparative Genomics Guides Elucidation of Vitamin B12 Biosynthesis in Novel Human-Associated Akkermansia Strains. Appl. Environ. Microbiol. 86, e02117–e02119 (2020).
Cozzolino, A. et al. Preliminary Evaluation of the Safety and Probiotic Potential of Akkermansia muciniphila DSM 22959 in Comparison with Lactobacillus rhamnosus GG. Microorganisms 8, 189 (2020).
Dubourg, G. et al. High-level colonisation of the human gut by Verrucomicrobia following broad-spectrum antibiotic treatment. Int. J. antimicrobial agents 41, 149–155 (2013).
Druart, C. et al. Toxicological safety evaluation of pasteurized Akkermansia muciniphila. J. Appl. Toxicol.: JAT 41, 276–290 (2021).
Turck, D. et al. Safety of pasteurised Akkermansia muciniphila as a novel food pursuant to Regulation (EU) 2015/2283. EFSA J. Eur. Food Saf. Auth. 19, e06780 (2021).
Li, G. et al. Diversity of Duodenal and Rectal Microbiota in Biopsy Tissues and Luminal Contents in Healthy Volunteers. J. Microbiol. Biotechnol. 25, 1136–1145 (2015).
Rogers, M. B. et al. Disturbances of the Perioperative Microbiome Across Multiple Body Sites in Patients Undergoing Pancreaticoduodenectomy. Pancreas 46, 260–267 (2017).
Wang, M., Ahrné, S., Jeppsson, B. & Molin, G. Comparison of bacterial diversity along the human intestinal tract by direct cloning and sequencing of 16S rRNA genes. FEMS Microbiol. Ecol. 54, 219–231 (2005).
Madsen, J. L. Effects of gender, age, and body mass index on gastrointestinal transit times. Digestive Dis. Sci. 37, 1548–1553 (1992).
Johansson, M. E. V. et al. Composition and functional role of the mucus layers in the intestine. Cell. Mol. life Sci.: CMLS 68, 3635–3641 (2011).
Derrien, M. et al. Modulation of Mucosal Immune Response, Tolerance, and Proliferation in Mice Colonized by the Mucin-Degrader Akkermansia muciniphila. Front. Microbiol. 2, 166 (2011).
van den Abbeele, P. et al. Arabinoxylans and inulin differentially modulate the mucosal and luminal gut microbiota and mucin-degradation in humanized rats. Environ. Microbiol. 13, 2667–2680 (2011).
Lyra, A. et al. Comparison of bacterial quantities in left and right colon biopsies and faeces. World J. Gastroenterol. 18, 4404–4411 (2012).
Ringel, Y. et al. High throughput sequencing reveals distinct microbial populations within the mucosal and luminal niches in healthy individuals. Gut microbes 6, 173–181 (2015).
Evans, D. F. et al. Measurement of gastrointestinal pH profiles in normal ambulant human subjects. Gut 29, 1035–1041 (1988).
van Herreweghen, F. et al. In vitro colonisation of the distal colon by Akkermansia muciniphila is largely mucin and pH dependent. Beneficial microbes 8, 81–96 (2017).
Bäckhed, F. et al. Dynamics and Stabilization of the Human Gut Microbiome during the First Year of Life. Cell host microbe 17, 690–703 (2015).
Guo, M. et al. Developmental differences in the intestinal microbiota of Chinese 1-year-old infants and 4-year-old children. Sci. Rep. 10, 19470 (2020).
Kong, F. et al. Gut microbiota signatures of longevity. Curr. Biol.: CB 26, R832–R833 (2016).
Biagi, E. et al. Gut Microbiota and Extreme Longevity. Curr. Biol.: CB 26, 1480–1485 (2016).
Bárcena, C. et al. Healthspan and lifespan extension by fecal microbiota transplantation into progeroid mice. Nat. Med. 25, 1234–1242 (2019).
Salazar, N. et al. Age-Associated Changes in Gut Microbiota and Dietary Components Related with the Immune System in Adulthood and Old Age: A Cross-Sectional Study. Nutrients 11, 1765 (2019).
Kim, B.-S. et al. Comparison of the Gut Microbiota of Centenarians in Longevity Villages of South Korea with Those of Other Age Groups. J. Microbiol. Biotechnol. 29, 429–440 (2019).
Rampelli, S. et al. Shotgun Metagenomics of Gut Microbiota in Humans with up to Extreme Longevity and the Increasing Role of Xenobiotic Degradation. mSystems 5, e00124–20 (2020).
van der Lugt, B. et al. Akkermansia muciniphila ameliorates the age-related decline in colonic mucus thickness and attenuates immune activation in accelerated aging Ercc1-/Δ7 mice. Immun. Ageing.: I A 16, 6 (2019).
Bodogai, M. et al. Commensal bacteria contribute to insulin resistance in aging by activating innate B1a cells. Sci. Transl. Med. 10, aat4271 (2018).
van der Lugt, B. et al. Integrative analysis of gut microbiota composition, host colonic gene expression and intraluminal metabolites in aging C57BL/6J mice. Aging 10, 930–950 (2018).
Zhang, X. et al. Age-related compositional changes and correlations of gut microbiome, serum metabolome, and immune factor in rats. GeroScience 43, 709–725 (2021).
Grivennikov, S. I. Inflammation and colorectal cancer: colitis-associated neoplasia. Semin. Immunopathol. 35, 229–244 (2013).
Jemal, A. et al. Cancer Statistics, 2007. CA: A Cancer J. Clinicians 57, 43–66 (2007).
Vigsnæs, L. K., Brynskov, J., Steenholdt, C., Wilcks, A. & Licht, T. R. Gram-negative bacteria account for main differences between faecal microbiota from patients with ulcerative colitis and healthy controls. Beneficial microbes 3, 287–297 (2012).
Zhang, T. et al. Alterations of Akkermansia muciniphila in the inflammatory bowel disease patients with washed microbiota transplantation. Appl. Microbiol. Biotechnol. 104, 10203–10215 (2020).
Earley, H. et al. The abundance of Akkermansia muciniphila and its relationship with sulphated colonic mucins in health and ulcerative colitis. Sci. Rep. 9, 15683 (2019).
Png, C. W. et al. Mucolytic bacteria with increased prevalence in IBD mucosa augment in vitro utilization of mucin by other bacteria. Am. J. Gastroenterol. 105, 2420–2428 (2010).
Song, C.-H. et al. Changes in Microbial Community Composition Related to Sex and Colon Cancer by Nrf2 Knockout. Front. Cell. Infect. Microbiol. 11, 636808 (2021).
Lang, M. et al. Crypt residing bacteria and proximal colonic carcinogenesis in a mouse model of Lynch syndrome. Int. J. cancer 147, 2316–2326 (2020).
Han, S. et al. Adequate Lymph Node Assessments and Investigation of Gut Microorganisms and Microbial Metabolites in Colorectal Cancer. OncoTargets Ther. 13, 1893–1906 (2020).
Vakili, B., Fateh, A., Asadzadeh Aghdaei, H., Sotoodehnejadnematalahi, F. & Siadat, S. D. Characterization of Gut Microbiota in Hospitalized Patients with Clostridioides difficile Infection. Curr. Microbiol. 77, 1673–1680 (2020).
Borton, M. A. et al. Chemical and pathogen-induced inflammation disrupt the murine intestinal microbiome. Microbiome 5, 47 (2017).
Bolte, L. A. et al. Long-term dietary patterns are associated with pro-inflammatory and anti-inflammatory features of the gut microbiome. Gut 70, 1287–1298 (2021).
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, https://doi.org/10.1038/nature12820 (2014).
Kim, Y. et al. Dietary cellulose prevents gut inflammation by modulating lipid metabolism and gut microbiota. Gut microbes 11, 944–961 (2020).
Koistinen, V. M. et al. Contribution of gut microbiota to metabolism of dietary glycine betaine in mice and in vitro colonic fermentation. Microbiome 7, 103 (2019).
Pelpolage, S. W. et al. Colonic fermentation of water soluble fiber fraction extracted from sugarcane (Sacchurum officinarum L.) bagasse in murine models. Food Chem. 292, 336–345 (2019).
Li, N. et al. Human milk and infant formula modulate the intestinal microbiota and immune systems of human microbiota-associated mice. Food Funct. 12, 2784–2798 (2021).
Rubio-Del-Campo, A. et al. Infant gut microbiota modulation by human milk disaccharides in humanized microbiome mice. Gut microbes 13, 1–20 (2021).
Partula, V. et al. Associations between usual diet and gut microbiota composition: results from the Milieu Intérieur cross-sectional study. Am. J. Clin. Nutr. 109, 1472–1483 (2019).
Zhang, L. et al. Grape proanthocyanidin-induced intestinal bloom of Akkermansia muciniphila is dependent on its baseline abundance and precedes activation of host genes related to metabolic health. J. nutritional Biochem. 56, 142–151 (2018).
Zhang, Z. et al. Chlorogenic Acid Ameliorates Experimental Colitis by Promoting Growth of Akkermansia in Mice. Nutrients 9, 677 (2017).
Chen, M. et al. Resveratrol attenuates high-fat diet-induced non-alcoholic steatohepatitis by maintaining gut barrier integrity and inhibiting gut inflammation through regulation of the endocannabinoid system. Clin. Nutr. (Edinb., Scotl.) 39, 1264–1275 (2020).
Jang, Y. J., Kim, W.-K., Han, D. H., Lee, K. & Ko, G. Lactobacillus fermentum species ameliorate dextran sulfate sodium-induced colitis by regulating the immune response and altering gut microbiota. Gut microbes 10, 696–711 (2019).
Liu, Y. et al. Long-term and continuous administration of Bacillus subtilis during remission effectively maintains the remission of inflammatory bowel disease by protecting intestinal integrity, regulating epithelial proliferation, and reshaping microbial structure and function. Food Funct. 12, 2201–2210 (2021).
Fan, L. et al. B. adolescentis ameliorates chronic colitis by regulating Treg/Th2 response and gut microbiota remodeling. Gut microbes 13, 1–17 (2021).
Chung, Y. et al. A synthetic probiotic engineered for colorectal cancer therapy modulates gut microbiota. Microbiome 9, 122 (2021).
Wang, T. et al. Lactobacillus coryniformis MXJ32 administration ameliorates azoxymethane/dextran sulfate sodium-induced colitis-associated colorectal cancer via reshaping intestinal microenvironment and alleviating inflammatory response. European journal of nutrition, https://doi.org/10.1007/s00394-021-02627-8 (2021).
Wu, X., Unno, T., Kang, S. & Park, S. A Korean-Style Balanced Diet Has a Potential Connection with Ruminococcaceae Enterotype and Reduction of Metabolic Syndrome Incidence in Korean Adults. Nutrients 13, 495 (2021).
Li, L. et al. The effects of daily fasting hours on shaping gut microbiota in mice. BMC Microbiol. 20, 65 (2020).
Zheng, J. et al. Dietary inflammatory potential in relation to the gut microbiome: results from a cross-sectional study. Br. J. Nutr. 124, 931–942 (2020).
Kong, C. et al. Ketogenic diet alleviates colitis by reduction of colonic group 3 innate lymphoid cells through altering gut microbiome. Signal Transduct. Target. Ther. 6, 154 (2021).
Vandeputte, D. et al. Stool consistency is strongly associated with gut microbiota richness and composition, enterotypes and bacterial growth rates. Gut 65, 57–62 (2016).
Asnicar, F. et al. Blue poo: impact of gut transit time on the gut microbiome using a novel marker. Gut 70, 1665–1674 (2021).
Bian, X. et al. Administration of Akkermansia muciniphila Ameliorates Dextran Sulfate Sodium-Induced Ulcerative Colitis in Mice. Front. Microbiol. 10, 2259 (2019).
Blikslager, A. T., Moeser, A. J., Gookin, J. L., Jones, S. L. & Odle, J. Restoration of barrier function in injured intestinal mucosa. Physiological Rev. 87, 545–564 (2007).
Ottman, N. et al. Pili-like proteins of Akkermansia muciniphila modulate host immune responses and gut barrier function. PloS one 12, e0173004 (2017).
Chelakkot, C. et al. Akkermansia muciniphila-derived extracellular vesicles influence gut permeability through the regulation of tight junctions. Exp. Mol. Med. 50, e450 (2018).
Wang, L. et al. A purified membrane protein from Akkermansia muciniphila or the pasteurised bacterium blunts colitis associated tumourigenesis by modulation of CD8+ T cells in mice. Gut 69, 1988–1997 (2020).
Li, J., Lin, S., Vanhoutte, P. M., Woo, C. W. & Xu, A. Akkermansia Muciniphila Protects Against Atherosclerosis by Preventing Metabolic Endotoxemia-Induced Inflammation in Apoe−/− Mice. Circulation 133, 2434–2446 (2016).
Hansson, G. C. & Johansson, M. E. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Gut microbes 1, 51–54 (2010).
Shin, J. et al. Elucidation of Akkermansia muciniphila Probiotic Traits Driven by Mucin Depletion. Front. Microbiol. 10, 1137 (2019).
Zhu, L. et al. Akkermansia muciniphila protects intestinal mucosa from damage caused by S. pullorum by initiating proliferation of intestinal epithelium. Vet. Res. 51, 34 (2020).
Crespo-Piazuelo, D. et al. Association between the pig genome and its gut microbiota composition. Sci. Rep. 9, 8791 (2019).
Hiraoka, N. et al. Molecular cloning and expression of two distinct human chondroitin 4-O-sulfotransferases that belong to the HNK-1 sulfotransferase gene family. J. Biol. Chem. 275, 20188–20196 (2000).
Ouwerkerk, J. P., Vos, W. M. de & Belzer, C. Glycobiome: bacteria and mucus at the epithelial interface. Best. Pract. Res. Clin. Gastroenterol. 27, 25–38 (2013).
Muccioli, G. G. et al. The endocannabinoid system links gut microbiota to adipogenesis. Mol. Syst. Biol. 6, 392 (2010).
Ansaldo, E. et al. Akkermansia muciniphila induces intestinal adaptive immune responses during homeostasis. Sci. (N. Y.) 364, 1179–1184 (2019).
Kuczma, M. P. et al. Commensal epitopes drive differentiation of colonic Tregs. Sci. Adv. 6, eaaz3186 (2020).
Zhai, R. et al. Strain-Specific Anti-inflammatory Properties of Two Akkermansia muciniphila Strains on Chronic Colitis in Mice. Front. Cell. Infect. Microbiol. 9, 239 (2019).
Liu, Y. et al. TLR4 regulates RORγt+ regulatory T-cell responses and susceptibility to colon inflammation through interaction with Akkermansia muciniphila. Microbiome 10, 98 (2022).
Craft, J. E. Follicular helper T cells in immunity and systemic autoimmunity. Nat. Rev. Rheumatol. 8, 337–347, https://doi.org/10.1038/nrrheum.2012.58 (2012).
Lee, J. C. et al. Gene expression profiling of CD8+ T cells predicts prognosis in patients with Crohn disease and ulcerative colitis. J. Clin. Investig. 121, 4170–4179 (2011).
Bunker, J. J. et al. Innate and Adaptive Humoral Responses Coat Distinct Commensal Bacteria with Immunoglobulin A. Immunity 43, 541–553 (2015).
Meng, X. et al. A Purified Aspartic Protease from Akkermansia Muciniphila Plays an Important Role in Degrading Muc2. Int. J. Mol. Sci. 21, 72 (2019).
Meng, X., Zhang, J., Wu, H., Yu, D. & Fang, X. Akkermansia muciniphila Aspartic Protease Amuc_1434* Inhibits Human Colorectal Cancer LS174T Cell Viability via TRAIL-Mediated Apoptosis Pathway. Int. J. Mol. Sci. 21, 3385 (2020).
Gobert, A. P. et al. The human intestinal microbiota of constipated-predominant irritable bowel syndrome patients exhibits anti-inflammatory properties. Sci. Rep. 6, 39399 (2016).
Kang, C.-S. et al. Extracellular vesicles derived from gut microbiota, especially Akkermansia muciniphila, protect the progression of dextran sulfate sodium-induced colitis. PloS one 8, e76520 (2013).
Bae, M. et al. Akkermansia muciniphila phospholipid induces homeostatic immune responses. Nature, https://doi.org/10.1038/s41586-022-04985-7 (2022).
Vaishnava, S. et al. The antibacterial lectin RegIIIgamma promotes the spatial segregation of microbiota and host in the intestine. Sci. (N. Y.) 334, 255–258 (2011).
Alam, A. et al. The microenvironment of injured murine gut elicits a local pro-restitutive microbiota. Nat. Microbiol. 1, 15021 (2016).
Weichselbaum, L. & Klein, O. D. The intestinal epithelial response to damage. Sci. China Life Sci. 61, 1205–1211 (2018).
Lotz, M. M. et al. Intestinal epithelial restitution. Involvement of specific laminin isoforms and integrin laminin receptors in wound closure of a transformed model epithelium. Am. J. Pathol. 150, 747–760 (1997).
Lepage, M., Seltana, A., Thibault, M.-P., Tremblay, É. & Beaulieu, J.-F. Knockdown of laminin α5 stimulates intestinal cell differentiation. Biochemical biophysical Res. Commun. 495, 1510–1515 (2018).
Coskun, M. et al. Regulation of Laminin γ2 Expression by CDX2 in Colonic Epithelial Cells Is Impaired During Active Inflammation. J. Cell. Biochem. 118, 298–307 (2017).
Thompson, A. I. & Lees, C. W. Genetics of ulcerative colitis. Inflamm. bowel Dis. 17, 831–848 (2011).
Barrett, J. C. et al. Genome-wide association study of ulcerative colitis identifies three new susceptibility loci, including the HNF4A region. Nat. Genet. 41, 1330–1334 (2009).
Pichler, M. J. et al. Butyrate producing colonic Clostridiales metabolise human milk oligosaccharides and cross feed on mucin via conserved pathways. Nat. Commun. 11, 3285 (2020).
Hänninen, A. et al. Akkermansia muciniphila induces gut microbiota remodelling and controls islet autoimmunity in NOD mice. Gut 67, 1445–1453 (2018).
Kim, S. et al. Akkermansia muciniphila Prevents Fatty Liver, Decreases Serum Triglycerides, and Maintains Gut Homeostasis. Appl. Environ. Microbiol. 86, e03004–e03019 (2020).
Lopez-Siles, M. et al. Alterations in the Abundance and Co-occurrence of Akkermansia muciniphila and Faecalibacterium prausnitzii in the Colonic Mucosa of Inflammatory Bowel Disease Subjects. Front. Cell. Infect. Microbiol. 8, 281 (2018).
Dunn, K. A. et al. Early Changes in Microbial Community Structure Are Associated with Sustained Remission After Nutritional Treatment of Pediatric Crohn’s Disease. Inflamm. bowel Dis. 22, 2853–2862 (2016).
Baxter, N. T., Zackular, J. P., Chen, G. Y. & Schloss, P. D. Structure of the gut microbiome following colonization with human feces determines colonic tumor burden. Microbiome 2, 20 (2014).
Dingemanse, C. et al. Akkermansia muciniphila and Helicobacter typhlonius modulate intestinal tumor development in mice. Carcinogenesis 36, 1388–1396 (2015).
Bornet, E. & Westermann, A. J. The ambivalent role of Bacteroides in enteric infections. Trends Microbiol. 30, 104–108 (2022).
Koretz, R. L. Response to Dr. Baldassarre. Am. J. Gastroenterol. 113, 1561–1562 (2018).
Zhang, C. & Zhao, L. Strain-level dissection of the contribution of the gut microbiome to human metabolic disease. Genome Med. 8, 41 (2016).
Filippis, F. de et al. Distinct Genetic and Functional Traits of Human Intestinal Prevotella copri Strains Are Associated with Different Habitual Diets. Cell host microbe 25, 444–453.e3 (2019).
Karcher, N. et al. Genomic diversity and ecology of human-associated Akkermansia species in the gut microbiome revealed by extensive metagenomic assembly. Genome Biol. 22, 209 (2021).
Lv, Q.-B. et al. A thousand metagenome-assembled genomes of Akkermansia reveal new phylogroups and geographical and functional variations in human gut. (2020).
Kim, S. et al. Mucin degrader Akkermansia muciniphila accelerates intestinal stem cell-mediated epithelial development. Gut microbes 13, 1–20 (2021).
Argüello, H. et al. Early Salmonella Typhimurium infection in pigs disrupts Microbiome composition and functionality principally at the ileum mucosa. Sci. Rep. 8, 7788 (2018).
Zackular, J. P. et al. The gut microbiome modulates colon tumorigenesis. mBio 4, e00692–13 (2013).
Acknowledgements
This work is supported by the National Natural Science Foundation of China (grant numbers 31872369 and 32072743). The authors thank Yifan Bao (Department of Physiological Chemistry, University of Vienna) to help draw figures.
Author information
Authors and Affiliations
Contributions
Y.L., C.L., H.L. and Q.O. collected the references, conceived and wrote the manuscript and share equal contribution; F.K. and A.W. helped to carry out pan genomic analysis. Z.R., G.T. J.C., and A.D.W. helped to collect references and revise the manuscript; B.Y. and J.H. helped to prepare and organize the tables and figures. All authors have read the completed version of the manuscript and agreed to its publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Luo, Y., Lan, C., Li, H. et al. Rational consideration of Akkermansia muciniphila targeting intestinal health: advantages and challenges. npj Biofilms Microbiomes 8, 81 (2022). https://doi.org/10.1038/s41522-022-00338-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41522-022-00338-4
This article is cited by
-
Multi-omics analysis of the gut microbiome and metabolites associated with the psychoneurological symptom cluster in children with cancer receiving chemotherapy
Journal of Translational Medicine (2024)
-
Microbiome variations among age classes and diets of captive Asian elephants (Elephas maximus) in Thailand using full-length 16S rRNA nanopore sequencing
Scientific Reports (2023)
-
Colonization with ubiquitous protist Blastocystis ST1 ameliorates DSS-induced colitis and promotes beneficial microbiota and immune outcomes
npj Biofilms and Microbiomes (2023)
-
Aerobic exercise training and gut microbiome-associated metabolic shifts in women with overweight: a multi-omic study
Scientific Reports (2023)
-
Resistant starches from dietary pulses modulate the gut metabolome in association with microbiome in a humanized murine model of ageing
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.