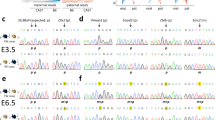
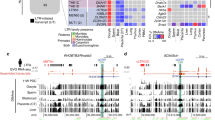
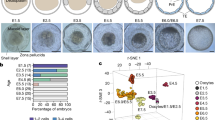
Abstract
The imprinted isoform of the Mest gene in mice is involved in key mammalian traits such as placental and fetal growth, maternal care and mammary gland maturation. The imprinted isoform has a distinct differentially methylated region (DMR) at its promoter in eutherian mammals but in marsupials, there are no differentially methylated CpG islands between the parental alleles. Here, we examined similarities and differences in the MEST gene locus across mammals using a marsupial, the tammar wallaby, a monotreme, the platypus, and a eutherian, the mouse, to investigate how imprinting of this gene evolved in mammals. By confirming the presence of the short isoform in all mammalian groups (which is imprinted in eutherians), this study suggests that an alternative promoter for the short isoform evolved at the MEST gene locus in the common ancestor of mammals. In the tammar, the short isoform of MEST shared the putative promoter CpG island with an antisense lncRNA previously identified in humans and an isoform of a neighbouring gene CEP41. The antisense lncRNA was expressed in tammar sperm, as seen in humans. This suggested that the conserved lncRNA might be important in the establishment of MEST imprinting in therian mammals, but it was not imprinted in the tammar. In contrast to previous studies, this study shows that MEST is not imprinted in marsupials. MEST imprinting in eutherians, therefore must have occurred after the marsupial-eutherian split with the acquisition of a key epigenetic imprinting control region, the differentially methylated CpG islands between the parental alleles.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Raw RNA-seq data set of tammar adult testis (GEO accession: DRP001145) is publicly available on NCBI SRA (https://www.ncbi.nlm.nih.gov/sra).
References
Ager E, Suzuki S, Pask A, Shaw G, Ishino F, Renfree MB (2007) Insulin is imprinted in the placenta of the marsupial, Macropus eugenii. Dev Biol 309:317–328. https://doi.org/10.1016/j.ydbio.2007.07.025
Bonthuis PJ, Steinwand S, Stacher Hörndli CN, Emery J, Huang WC, Kravitz S et al. (2022) Noncanonical genomic imprinting in the monoamine system determines naturalistic foraging and brain-adrenal axis functions. Cell Rep 38. https://doi.org/10.1016/j.celrep.2022.110500
Cao W, Douglas KC, Samollow PB, VandeBerg JL, Wang X, Clark AG (2023) Origin and evolution of marsupial-specific imprinting clusters through lineage-specific gene duplications and acquisition of promoter differential methylation. Mol Biol Evol 40. https://doi.org/10.1093/molbev/msad022
Cooper D, Johnston P, Vandeberg J, Robinson E (1989) X-chromosome inactivation in marsupials. Aust J Zool 37:411. https://doi.org/10.1071/ZO9890411
Cooper DW (1971) Directed genetic change model for X chromosome inactivation in eutherian mammals. Nature 230:292–294. https://doi.org/10.1038/230292a0
Cooper DW, VandeBerg JL, Sharman GB, Poole WE (1971) Phosphoglycerate kinase polymorphism in kangaroos provides further evidence for paternal X inactivation. Nat N. Biol 230:155–157. https://doi.org/10.1038/newbio230155a0
Couldrey C, Moitra J, Vinson C, Anver M, Nagashima K, Green J (2002) Adipose tissue: a vital in vivo role in mammary gland development but not differentiation. Dev Dyn 223:459–468. https://doi.org/10.1002/dvdy.10065
Das R, Anderson N, Koran MI, Weidman JR, Mikkelsen TS, Kamal M et al. (2012) Convergent and divergent evolution of genomic imprinting in the marsupial Monodelphis domestica. BMC Genomics 16:394. https://doi.org/10.1186/1471-2164-13-394
Douglas KC, Wang X, Jasti M, Wolff A, VandeBerg JL, Clark AG et al. (2014) Genome-wide histone state profiling of fibroblasts from the opossum, Monodelphis domestica, identifies the first marsupial-specific imprinted gene. BMC Genomics 15:89. https://doi.org/10.1186/1471-2164-15-89
Edwards CA, Mungall AJ, Matthews L, Ryder E, Gray DJ, Pask AJ et al. (2008) The evolution of the DLK1-DIO3 imprinted domain in mammals. PLoS Biol 6:1292–1305. https://doi.org/10.1371/journal.pbio.0060135
Edwards CA, Takahashi N, Corish JA, Ferguson-Smith AC (2019) The origins of genomic imprinting in mammals. Reprod Fertil Dev 31:1203. https://doi.org/10.1071/RD18176
Eggermann T, Spengler S, Begemann M, Binder G, Buiting K, Albrecht B et al. (2012) Deletion of the paternal allele of the imprinted MEST/PEG1 region in a patient with Silver-Russell syndrome features. Clin Genet 81:298–300. https://doi.org/10.1111/j.1399-0004.2011.01719.x
Ferguson-Smith AC (2011) Genomic imprinting: the emergence of an epigenetic paradigm. Nat Rev Genet 12:565–575. https://doi.org/10.1038/nrg3032
Grant J, Mahadevaiah SK, Khil P, Sangrithi MN, Royo H, Duckworth J et al. (2012) Rsx is a metatherian RNA with Xist-like properties in X-chromosome inactivation. Nature 487:254–258. https://doi.org/10.1038/nature11171
Green B, Griffiths M, MC Leckie R (1983) Qualitative and quantitative changes in milk fat during lactation in the tammar wallaby (Macropus eugenii). Aust J Biol Sci 36:455. https://doi.org/10.1071/BI9830455
Green SW, Renfree MB(1982) Changes in the milk proteins during lactation in the tammar wallaby, Macropus eugenii. Aust J Bioi Sci 35(2):145–152. https://doi.org/10.1071/bi9820145
Haig D (2000) The kinship theory of genomic imprinting. Annu Rev Ecol Syst 31:9–32. https://doi.org/10.1146/annurev.ecolsys.31.1.9
Hiramuki Y, Sato T, Furuta Y, Surani MA, Sehara-Fujisawa A (2015) Mest but not MIR-335 affects skeletal muscle growth and regeneration. PLoS One 10:1–15. https://doi.org/10.1371/journal.pone.0130436
Holm TM, Jackson-Grusby L, Brambrink T, Yamada Y, Rideout WM, Jaenisch R (2005) Global loss of imprinting leads to widespread tumorigenesis in adult mice. Cancer Cell 8:275–285. https://doi.org/10.1016/j.ccr.2005.09.007
Huntriss JD, Hemmings KE, Hinkins M, Rutherford AJ, Sturmey RG et al. (2013) Variable imprinting of the MEST gene in human preimplantation embryos. Eur J Hum Genet 21:40–47. https://doi.org/10.1038/ejhg.2012.102
Ishihara T, Griffith OW, Suzuki S, Renfree MB (2022a) Placental imprinting of SLC22A3 in the IGF2R imprinted domain is conserved in therian mammals. Epigenetics Chromatin 15. https://doi.org/10.1186/s13072-022-00465-4
Ishihara T, Hickford D, Fenelon JC, Griffith OW, Suzuki S, Renfree MB (2022b) Evolution of the short form of DNMT3A, DNMT3A2, occurred in the common ancestor of mammals. Genome Biol Evol 14. https://doi.org/10.1093/gbe/evac094
Kamei Y, Suganami T, Kohda T, Ishino F, Yasuda K, Miura S et al. (2007) Peg1/Mest in obese adipose tissue is expressed from the paternal allele in an isoform-specific manner. FEBS Lett 581:91–96. https://doi.org/10.1016/j.febslet.2006.12.002
Killian JK, Byrd JC, Jirtle JV, Munday BL, Stoskopf MK, MacDonald RG et al. (2000) M6P/IGF2R imprinting evolution in mammals. Mol Cell 5:707–716. https://doi.org/10.1016/S1097-2765(00)80249-X
Kim D, Paggi JM, Park C, Bennett C, Salzberg SL (2019) Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat Biotechnol 37:907–915. https://doi.org/10.1038/s41587-019-0201-4
Kosaki K, Kosaki R, Craigen WJ, Matsuo N (2000) Isoform-specific imprinting of the human PEG1/MEST gene. Am J Hum Genet 66:309–312. https://doi.org/10.1086/302712
Lefebvre L, Viville S, Barton SC, Ishino F, Keverne EB, Azim Surani M (1998) Abnormal maternal behaviour and growth retardation associated with loss of the imprinted gene Mest. Nat Genet 20:163–169. https://doi.org/10.1038/2464
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G et al. (2009) The sequence alignment/ap format and SAMtools. Bioinformatics 25:2078–2079. https://doi.org/10.1093/bioinformatics/btp352
Li L-C, Dahiya R (2002) MethPrimer: designing primers for methylation PCRs. Bioinformatics 18:1427–1431. https://doi.org/10.1093/bioinformatics/18.11.1427
Li T, Vu TH, Lee KO, Yang Y, Nguyen CV, Bui HQ et al. (2002) An imprinted PEG1/MEST antisense expressed predominantly in human testis and in mature spermatozoa. J Biol Chem 277:13518–13527. https://doi.org/10.1074/jbc.M200458200
Li X, Song N, Wang D, Han X, Lv Q, Ouyang H et al. (2015) Isoform-specific imprinting of the MEST gene in porcine parthenogenetic fetuses. Gene 558:287–290. https://doi.org/10.1016/j.gene.2015.01.031
Mahadevaiah SK, Sangrithi MN, Hirota T, Turner JMA (2020) A single-cell transcriptome atlas of marsupial embryogenesis and X inactivation. Nature 586:612–617. https://doi.org/10.1038/s41586-020-2629-6
Mayer W, Hemberger M, Frank HG, Grümmer R, Winterhager E, Kaufmann P et al. (2000) Expression of the imprinted genes MEST/Mest in human and murine placenta suggests a role in angiogenesis. Dev Dyn 217:1–10. https://doi.org/10.1002/(SICI)1097-0177(200001)217:1<1::AID-DVDY1>3.0.CO;2-4
Nakabayashi K (2002) Identification and characterization of an imprinted antisense RNA (MESTIT1) in the human MEST locus on chromosome 7q32. Hum Mol Genet 11:1743–1756. https://doi.org/10.1093/hmg/11.15.1743
National Health and Medical Research Council (Australia) (2013) Australian code for the care and use of animals for scientific purposes.
Nicholas K, Sharp J, Watt A, Wanyonyi S, Crowley T, Gillespie M et al. (2012) The tammar wallaby: a model system to examine domain-specific delivery of milk protein bioactives. Semin Cell Dev Biol. https://doi.org/10.1016/j.semcdb.2012.03.016
O’Neill MJ, Ingram RS, Vrana PB, Tilghman SM (2000) Allelic expression of IGF2 in marsupials and birds. Dev Genes Evol 210:18–20. https://doi.org/10.1007/PL00008182
Pask AJ, Papenfuss AT, Ager EI, McColl KA, Speed TP, Renfree MB (2009) Analysis of the platypus genome suggests a transposon origin for mammalian imprinting. Genome Biol 10:R1. https://doi.org/10.1186/gb-2009-10-1-r1
Patten MM, Ross L, Curley JP, Queller DC, Bonduriansky R, Wolf JB (2014) The evolution of genomic imprinting: theories, predictions and empirical tests. Heredity (Edinb). https://doi.org/10.1038/hdy.2014.29
Reik W, Walter J (2001) Evolution of imprinting mechanisms: the battle of the sexes begins in the zygote. Nat Genet 27:255–256. https://doi.org/10.1038/85804
Renfree MB (2010) Marsupials: placental mammals with a difference. Placenta 31:S21–S26. https://doi.org/10.1016/j.placenta.2009.12.023
Renfree MB (1973a) Proteins in the uterine secretions of the marsupial Macropus eugenii. Dev Biol 32:41–49. https://doi.org/10.1016/0012-1606(73)90218-2
Renfree MB (1973b) The composition of fetal fluids of the marsupial Macropus eugenii. Dev Biol 33:62–79. https://doi.org/10.1016/0012-1606(73)90165-6
Renfree MB, Hore TA, Shaw G, Marshall Graves JA, Pask AJ (2009) Evolution of genomic imprinting: Insights from marsupials and monotremes. Annu Rev Genomics Hum Genet 10:241–262. https://doi.org/10.1146/annurev-genom-082908-150026
Reule M, Krause R, Hemberger M, Fundele R (1998) Analysis of Peg1/Mest imprinting in the mouse. Dev Genes Evol 208:161–163. https://doi.org/10.1007/s004270050168
Riesewijk AM, Hu L, Schulz U, Tariverdian G, Höglund P, Kere J et al. (1997) Monoallelic expression of human PEG1/MEST is paralleled by parent-specific methylation in fetuses. Genomics 42:236–244. https://doi.org/10.1006/geno.1997.4731
Robinson JT, Thorvaldsdóttir H, Winckler W, Guttman M, Lander ES, Getz G et al. (2011) Integrative genomics viewer. Nat Biotechnol 29:24–26. https://doi.org/10.1038/nbt.1754
Sahakyan A, Yang Y, Plath K (2018) The role of Xist in X-chromosome dosage compensation. Trends Cell Biol. https://doi.org/10.1016/j.tcb.2018.05.005
Santini L, Halbritter F, Titz-Teixeira F, Suzuki T, Asami M, Ma X, Ramesmayer J et al. (2021) Genomic imprinting in mouse blastocysts is predominantly associated with H3K27me3. Nat Commun 12:3804. https://doi.org/10.1038/s41467-021-23510-4
Smits G, Mungall AJ, Griffiths-Jones S, Smith P, Beury D, Matthews L et al. (2008) Conservation of the H19 noncoding RNA and H19-IGF2 imprinting mechanism in therians. Nat Genet 40:971–976. https://doi.org/10.1038/ng.168
Stringer JM, Pask AJ, Shaw G, Renfree MB (2014) Post-natal imprinting: evidence from marsupials. Heredity (Edinb) 113:145–155. https://doi.org/10.1038/hdy.2014.10
Stringer JM, Suzuki S, Pask AJ, Shaw G, Renfree MB (2012) Selected imprinting of INS in the marsupial. Epigenetics Chromatin 5:14. https://doi.org/10.1186/1756-8935-5-14
Suzuki S, Ono R, Narita T, Pask AJ, Shaw G, Wang C et al. (2007) Retrotransposon Silencing by DNA methylation can drive mammalian genomic imprinting. PLoS Genet 3:e55. https://doi.org/10.1371/journal.pgen.0030055
Suzuki S, Renfree MB, Pask AJ, Shaw G, Kobayashi S, Kohda T et al. (2005) Genomic imprinting of IGF2, p57KIP2 and PEG1/MEST in a marsupial, the tammar wallaby. Mech Dev 122:213–222. https://doi.org/10.1016/j.mod.2004.10.003
Suzuki S, Shaw G, Renfree MB (2018) Identification of a novel antisense noncoding RNA, ALID, transcribed from the putative imprinting control region of marsupial IGF2R. Epigenetics Chromatin 11:55. https://doi.org/10.1186/s13072-018-0227-8
Takahashi M, Kamei Y, Ezaki O, Takahashi Y, Kamei OE (2005) Mest/Peg1 imprinted gene enlarges adipocytes and is a marker of adipocyte size. Am J Physiol Endocrinol Metab 288:117–124. https://doi.org/10.1152/ajpendo
Thorvaldsdóttir H, Robinson JT, Mesirov JP (2013) Integrative Genomics Viewer (IGV): high-performance genomics data visualization and exploration. Brief Bioinform 14:178–192. https://doi.org/10.1093/bib/bbs017
Trott JF, Simpson KJ, Moyle RLC, Hearn CM, Shaw G, Nicholas KR et al. (2003) Maternal regulation of milk composition, milk production, and pouch young development during lactation in the tammar wallaby (Macropus eugenii). Biol Reprod 68:929–936. https://doi.org/10.1095/biolreprod.102.005934
Wang X, Clark AG (2014) Using next-generation RNA sequencing to identify imprinted genes. Heredity (Edinb) 113:156–166. https://doi.org/10.1038/hdy.2014.18
Wang X, Soloway PD, Clark AG (2011) A survey for novel imprinted genes in the mouse placenta by mRNA-seq. Genetics 189:109–122. https://doi.org/10.1534/genetics.111.130088
Yamada T, Kayashima T, Yamasaki K, Ohta T, Yoshiura KI, Matsumoto N et al. (2002) The gene TSGA14, adjacent to the imprinted gene MEST, escapes genomic imprinting. Gene 288:57–63. https://doi.org/10.1016/S0378-1119(02)00428-6
Yonekura S, Ohata M, Tsuchiya M, Tokita H, Mizusawa M, Tokutake Y (2019) Peg1/Mest, an imprinted gene, is involved in mammary gland maturation. J Cell Physiol 234:1080–1087. https://doi.org/10.1002/jcp.27219
Acknowledgements
We thank Corinne van den Hoek for assistance with the animals and all members of the wallaby research group for help with the tammar wallabies and the dissections in Melbourne. We thank Professor Peter Temple-Smith and Dr Kath Handasyde for help with the collection of platypus material. TI acknowledges support provided by a writing up award from the Albert Shimmins fund.
Author information
Authors and Affiliations
Contributions
TI, SS, JCF, GS, OWG and MBR discussed and designed the research; TI performed most of the research; TAN performed allelic expression analysis on PY tissues; JCF, GS and MBR collected platypus tissues; MBR, GS, TAN and TI collected the tammar tissues; JCF extracted RNA from platypus tissue; SS, OWG and MBR provided supervision; MBR and GS (and a University of Melbourne Scholarship) provided the funding. TI, SS, JCF, OWG, GS and MBR discussed the data and TI, SS and MBR wrote the paper. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Associate editor: Aurora Ruiz-Herrera.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ishihara, T., Suzuki, S., Newman, T.A. et al. Marsupials have monoallelic MEST expression with a conserved antisense lncRNA but MEST is not imprinted. Heredity 132, 5–17 (2024). https://doi.org/10.1038/s41437-023-00656-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41437-023-00656-z