Abstract
Background/Objectives
Vismodegib, a hedgehog pathway inhibitor, has been used in the management of locally advanced basal cell carcinoma (BCC) not suitable for surgery or radiation therapy. We report our experience using neoadjuvant vismodegib for locally advanced periocular BCC, followed by surgical excision. Our aim was to assess the effect on the extent of surgical excision and histological response.
Subjects/Methods
A retrospective case series of patients treated with neoadjuvant vismodegib, for the management of locally advanced periocular BCC prior to surgical excision, with intraoperative margin control. Patients were treated until a maximum clinical response was seen. The difference between the estimated surgical margins prior to vismodegib and the eventual margins used was compared. Fine (1 mm) vertical sections through the excised tumour were performed to assess the histological response and look for a multifocal tumour.
Results
Eight Caucasian patients had neoadjuvant treatment with vismodegib for a median duration of 6 months. Some clinical response was seen in all cases but was only partial in 6/8 patients. Histological evaluation of the excised specimen showed residual BCC in 6/8 cases and thus 2/8 showed complete histological regression. Two cases showed squamous differentiation. Side effects occurred in 7/8 patients all which resolved on cessation of therapy. The mean follow-up was 13.4 ± 5.2 months.
Conclusions
Neoadjuvant treatment of periocular BCC showed a mixed clinical and histological response. Final surgical excision achieved clear margins in all patients with no recurrence at 13 months and a reduction in predicted defect size, but possible squamous differentiation in two cases.
Similar content being viewed by others
Introduction
Basal cell carcinoma (BCC) is the most common skin cancer, with Australia recording the highest incidence of up to 2% per year [1, 2]. Vismodegib (Erivedge, Genentech) a hedgehog pathway inhibitor, has been used in the management of locally advanced basal cell carcinoma (BCC) not suitable for surgery or radiation therapy and for metastatic BCC [3,4,5]. It acts on the hedgehog signalling pathway by inhibiting the signalling activity of smoothened homologue (SMO) [3]. Partial response rates are high, but drug-related adverse events are commonly reported and are higher with longer duration of treatment [3,4,5,6]. Vismodegib has been used in the management of locally advanced periocular BCC with similar rates of response [7,8,9,10,11,12].
The treatment has come under recent scrutiny in some countries with the National Institute for Health and Care Excellence (NICE) withdrawing its recommendation for the use of vismodegib in the management of BCC in the United Kingdom (UK). However, in Australia vismodegib remains licensed for histologically confirmed locally advanced BCC where the patient’s condition is inappropriate for surgery, where curative resection is unlikely following recurrence after two or more surgical procedures, or where substantial morbidity or deformity is anticipated due to loss of the eye or eyelid.
The role of vismodegib as a neoadjuvant treatment option has been investigated obviating the need for orbital exenteration or other major disfiguring surgery in the periocular area [9, 10, 13]. The purpose of this study is to report our experience with the use of neoadjuvant vismodegib for locally advanced periocular BCC, followed by surgical excision with intraoperative margin control. In particular to assess the effect of surgical margins and the histological response.
Materials (subjects) and methods
All consecutive patients with locally advanced periocular BCC who were treated with neoadjuvant vismodegib, for the management of locally advanced periocular BCC, prior to surgical excision at two tertiary centres in Adelaide, Australia, under the care of the senior author (DS), between 2017 and 2020. Institutional review board approval was obtained and the study adhered to the tenets of the declaration of Helsinki.
Baseline data collected included age, sex, ethnicity, tumour location and subtype. Lesions were staged according to the American Joint Committee on Cancer (AJCC) 8th edition cancer staging manual for eyelid carcinoma [14]. All patients received oral vismodegib 150 mg daily until one of the following conditions were met: a complete clinical response was reached; partial clinical response was seen and a plateau was reached with no further improvement seen in the clinical appearance of the tumour over two consecutive months, or the patient was intolerant of the medication.
The duration of vismodegib treatment and drug-related adverse events experienced were collected. The maximum diameter of the tumour with a surrounding 3 mm margin before treatment with vismodegib was recorded. This was to simulate the planned tumour excision without treatment. In addition, mapping biopsies were taken from the same sites before and after treatment with vismodegib, prior to definitive surgical excision. Photographs were used to guide areas being biopsied. The maximum diameter of the final excised area at the time of surgery was recorded. The time to surgical intervention, histopathological findings and duration of follow-up were also collected.
Results
Demographics
Eight Caucasian patients (five male, three female), of mean age 57.6 years (range 24–81), were identified in this review. The patients are detailed individually in Table 1 and summarised in Table 2.
Tumour characteristics
The tumour was located at the medial canthus in five patients, the lower eyelid in two patients and at the lateral canthus in one patient. Six patients had primary tumours and two had recurrent tumours. Both recurrent tumours had been managed surgically previously. No patients had prior radiotherapy. The histological subtype was nodular in four cases, infiltrative in three cases and mixed superficial/nodular in one case. The TNM staging was T4 in one case, T3a in four cases, T2b in three cases.
Treatment response
All patients had treatment with vismodegib at a dose of 150 mg once daily, for a median duration of 6 months (mean 5.1 ± 1.9, range 3–8 months). Some clinical response was seen in all cases but was only partial in 6/8 patients.
Drug-related adverse events
Side effects occurred in 7/8 patients (87.5%) with muscle cramps (5/8), hair loss (5/8) and dysgeusia (5/8) the most common. All side effects are detailed in Table 3. All side effects resolved on cessation of therapy. No cases of death were recorded.
Surgical management
Patients proceeded to surgical excision at a mean interval of 5.4 ± 3.7 weeks following completion of vismodegib treatment. 5/8 patients underwent excision with intraoperative margin control via frozen section analysis and 3/8 underwent MOH’s micrographic surgery. The mean maximum diameter of the tumour, with a surrounding 3 mm margin simulating the intended tissue excision without the use of vismodegib, was 27.3 ± 8.6 mm prior to treatment and the mean maximum diameter of the final excised area at the time of definitive surgical management was 19.1 ± 8.5 mm. Two patients had a reduction in maximum diameter of ≤3 mm.
Clear margins were achieved in all cases. Histological evaluation of the excised specimen showed residual BCC in 6/8 cases and with 2/8 showing no evidence of residual tumour and thus demonstrated complete histological regression. In addition, the excised specimens generally were comprised of fibrosis and a lymphohistiocytic inflammatory infiltrate, whether the residual tumour was present or not. Two cases demonstrated squamous differentiation on histological evaluation of the final excised specimen. No cases of tumour multifocality were seen in those with residual BCC. No patients proceeded to exenteration within the duration of follow-up of the study.
The mean follow-up following definitive surgical excision was 13.4 ± 5.2 months. No tumour recurrence was seen during this time.
Two case examples are illustrated in Figs. 1 and 2.
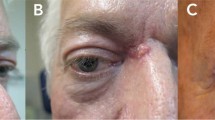
A Appearance of a mixed superficial/nodular BCC prior to treatment with vismodegib. B Clinical appearance of the same area following 6 months treatment with vismodegib with areas of mapping biopsies marked, which demonstrated no residual tumour. C Deformity following excision of the final area, which also demonstrated no residual tumour of fine vertical histological sectioning. D Appearance 3 months following reconstruction of the area with the aid of a sliding tarso-conjunctival flap and full-thickness skin graft.
A Appearance of a recurrent nodular lower lid BCC with mapping biopsies marked prior to treatment with vismodegib. B Clinical appearance of the same area 8 months following treatment with vismodegib, demonstrating a partial clinical response only. C Deformity following excision of the final area, which showed a residual tumour on fine vertical histological sectioning. Clear margins were achieved.
Discussion
We report our experience with the use of vismodegib as a neoadjuvant treatment in the management of patients with locally advanced periocular BCC prior to definitive surgical excision with a partial response in 6/8 patients and complete response in 2/8 patients.
A number of studies have investigated the use of vismodegib in the management of locally advanced periocular BCC and have reported complete response rates from 25 to 67% [9,10,11,12]. In addition, its use as a neoadjuvant treatment has been described prior to globe-sparing surgery [9, 10, 13]. We report a lower complete response rate of 25%, however, this may be explained in that using the treatment in a neoadjuvant fashion prior to surgical excision, the patients received the treatment for a shorter duration (median 6 months). Sagiv et al. reported a 63% complete clinical response rate with the treatment given for a median duration of 14 months, whereas Oliphant et al. reported a similar response to the present study, with 38% of patients demonstrating a complete clinical response in patients treated for a median duration of 7 months.
As all patients in the present study proceeded to definitive surgical excision, this complete response was confirmed histologically, with no residual tumour seen in these two patients. Interestingly, neither of these tumours where of infiltrative BCC on initial or repeat mapping biopsies, with one case a nodular BCC and the other a mixed superficial/nodular BCC. Furthermore, the two patients who showed the poorest response were those with recurrent tumours. One of these was a 24-year-old male with a recurrent lower eyelid BCC, and given the very young age of the patient, the genetic makeup of this tumour may vary from those in the more typically affected age group and may explain the limited clinical response. We also attempted to demonstrate tissue preservation by measuring the maximum diameter of the tumour with a surrounding 3 mm margin, simulating the intended area to be excised without vismodegib treatment and comparing this to the area excised at the time of definitive surgical management. The mean maximum diameter reduced from 27.3 to 19.1 mm suggesting some tissue preservation occurred.
A number of questions remain unanswered regarding patient selection for the use of vismodegib as a neoadjuvant in the management of locally advanced periocular BCC. It remains difficult to predict which patients are likely to respond completely versus those who show only a partial response or indeed do not respond at all. This is an important consideration if tissue preservation is one of the goals of treatment following tumour clearance. It remains uncertain if certain factors play a role in the likelihood of response to vismodegib such as BCC subtype, primary or recurrent tumours or indeed what other genetic or environmental factors play a role. Sekulic et al. [4] reported comparable objective response rates across histological subtypes although lower response rates in patients with larger tumours.
In addition, a number of considerations regarding the histological response to vismodegib treatment also remain. One such concern relates to the pattern of tumour regression. If reducing the tumour size prior to surgical excision is a goal of treatment in an effort to preserve tissue, it would be important to know the tumour is regressing homogeneously throughout its margin in an effort to excise any residual tumour as a single focus. A previous report demonstrated a case of tumour regression in a multifocal fashion, resulting in multiple foci of tumour in a patient with a partial response to treatment [9]. This would have implications for definitive surgical excision, with the potential for missing a focus of residual tumour and the possibility of recurrent disease.
Furthermore, the risk of squamous differentiation within basal cell carcinoma following treatment with vismodegib has been reported [15]. Moreover, the development of squamous cell carcinoma (SCC) has also been reported [16,17,18,19]. However, other authors have reported no subsequent risk of developing SCC [20]. In the present series, 2/8 patients showed squamous differentiation within the final excised basal cell carcinoma. Whilst it is not possible to say this was not present within some part of the tumour prior to vismodegib treatment, multiple initial mapping biopsies did not demonstrate the presence of this in either patient.
In both the ERIVANCE and STEVIE trials, patients with locally advanced BCC received vismodegib for a median 12.7 months and 39.7 weeks respectively [4, 21]. Sagiv et al. [9] reported a median of 14 months neoadjuvant treatment prior to surgical intervention. In the present study, patients were on treatment for a much shorter duration (median 6 months) and proceeded to surgical intervention shortly after (5.4 weeks). The optimal duration of vismodegib treatment prior to surgical intervention and the interval between cessation of treatment and surgery have yet to be evaluated [21]. Complete response was seen in as little as 3 months in one case in this series, similar to that reported by Sagiv et al. [9] who also noted a significant clinical response within the first month of all patients treated with vismodegib. By contrast, Ally et al. [22] stated that vismodegib was not effective in patients treated for <3 months. It was our intention to minimise the delay between completion of treatment and definitive surgical excision as clinical recurrence was noted in one case who waited 10 weeks to have surgery. In this case, one area biopsied following completion of vismodegib was shown to be free of tumour, but on the day of surgery appeared to have recurred clinically in this area and indeed was involved with tumour on final histological evaluation. It is our recommendation to proceed to definitive surgical intervention as soon as possible following completion of vismodegib treatment to minimise the risk of recurrence within this window
As similarly reported in other studies, drug-related adverse events were frequently experienced but resolved on cessation of treatment [4, 21]. Data from the ERIVANCE trial suggest a higher incidence of drug-related adverse events when vismodegib treatment lasted more than 12 months [4]. Ben Ishai et al. reported the meantime to first moderate or severe adverse event as 24.1 weeks [21]. In the present study, patients were on treatment for a median of 6 months, similar to their reported time to a first adverse event, and proceeded to surgical intervention shortly after (5.4 weeks). With tumour recurrence reported on cessation of therapy necessitating a return to vismodegib therapy for patients not suitable for surgery, a planned course of neoadjuvant therapy prior to surgical excision may make drug-related adverse events less frequent and perhaps more tolerable if taken for a shorter period of time.
We acknowledge a number of limitations of the study including its retrospective nature, the small number of patients and limited duration of follow-up.
We report our experience on the use of vismodegib as a neoadjuvant prior to surgical excision in the management of locally advanced periocular BCC. Whilst response to treatment was seen in most patients and the treatment may limit the degree of disfiguring surgery, or obviate the need for orbital exenteration in selected patients, it is difficult to predict clinical and histological responses and the role of vismodegib as a neoadjuvant in this setting remains uncertain and requires further investigation.
Summary
What was known before
-
Vismodegib has been used in the management of locally advanced BCC not suitable for surgery or radiation therapy.
-
The role of vismodegib as a neoadjuvant treatment option has been investigated, obviating the need for orbital exenteration or other major disfiguring surgery in the periocular area.
What this study adds
-
We report a mixed response to neoadjuvant vismodegib.
-
Response to the treatment was seen in most patients which may limit the degree of disfiguring surgery or obviate the need for orbital exenteration in selected patients. However, it is difficult to predict clinical and histological responses and the role of vismodegib as a neoadjuvant in this setting remains uncertain and requires further investigation.
References
Rubin AI, Chen EH, Désirée R. Basal-cell carcinoma. N. Engl J Med. 2005;353:2262–9.
Prabhakaran VC, Gupta A, Huilgol SC, Selva D. Basal cell carcinoma of the eyelids. 2007;8:1–14.
Sekulic A, Migden MR, Oro AE, Dirix L, Lewis KD, Hainsworth JD, et al. Efficacy and safety of vismodegib in advanced basal-cell carcinoma. N. Engl J Med. 2012;366:2171–9. http://www.nejm.org/doi/abs/10.1056/NEJMoa1113713.
Sekulic A, Migden MR, Basset-Seguin N, Garbe C, Gesierich A, Lao CD, et al. Long-term safety and efficacy of vismodegib in patients with advanced basal cell carcinoma: final update of the pivotal ERIVANCE BCC study. BMC Cancer. 2017;17:1–10.
Jacobsen AA, Aldahan AS, Hughes OB, Shah VV, Strasswimmer J. Hedgehog pathway inhibitor therapy for locally advanced and metastatic basal cell carcinoma a systematic review and pooled analysis of interventional studies. JAMA Dermatol. 2016;152:816–24.
Migden MR, Guminski A, Gutzmer R, Dirix L, Lewis KD, Combemale P, et al. Treatment with two different doses of sonidegib in patients with locally advanced or metastatic basal cell carcinoma (BOLT): A multicentre, randomised, double-blind phase 2 trial. Lancet Oncol. 2015;16:716–28.
Demirci H, Worden F, Nelson C, Elner V, Kahana A. Efficacy of vismodegib (ErivedgeTM) for basal cell carcinoma involving the orbit and periocular area. Ophthalmic Plast Reconstr Surg. 2015;31:463–6.
Gill HS, Moscato EE, Chang ALS, Soon S, Silkiss RZ. Vismodegib for periocular and orbital basal cell carcinoma. JAMA Ophthalmol. 2013;131:1591–4.
Sagiv O, Nagarajan P, Ferrarotto R, Kandl TJ, Thakar SD, Glisson BS, et al. Ocular preservation with neoadjuvant vismodegib in patients with locally advanced periocular basal cell carcinoma. Br. J. Ophthalmol. 2018;103:775–80.
Eiger-Moscovich M, Reich E, Tauber G, Berliner O, Priel A, Ben Simon G, et al. Efficacy of vismodegib for the treatment of orbital and advanced periocular basal cell carcinoma. Am J Ophthalmol 2019;207:62–70.
Wong KY, Fife K, Lear JT, Price RD, Durrani AJ. Vismodegib for locally advanced periocular and orbital basal cell carcinoma: a review of 15 consecutive cases. Plastr Reconstr Surg. 2017;5:1–6.
Oliphant H, Laybourne J, Chan K, Haridas A, Edmunds MR, Morris D, et al. Vismodegib for periocular basal cell carcinoma: an international multicentre case series. Eye. 2020;34:2076–81. https://doi.org/10.1038/s41433-020-0778-3.
Kahana A, Worden FP, Elner VM. Vismodegib as eye-sparing adjuvant treatment for orbital basal cell carcinoma. JAMA Ophthalmol. 2013;131:3–5.
Esmaeli B, Dutton J, Graue G. Eyelid carcinoma. In: Amin M, Edge S, Greene F, editors. AJCC cancer staging manual. Ch 64, 8th ed. Switzerland: Springer International Publishing; 2017. p. 779–85.
Feigenbaum L, Scott B, Moye M, Nijhawan R. Development of basal cell carcinoma with squamous differentiation during vismodegib treatment. Dermatologic Surg. 2017;43:989–91.
Mohan SV, Chang J, Li S, Solomon Henry A, Wood DJ, Chang ALS. Increased risk of cutaneous squamous cell carcinoma after vismodegib therapy for basal cell carcinoma. JAMA Dermatol. 2016;152:527–32.
Saintes C, Saint-Jean M, Brocard A, Peuvrel L, Renaut JJ, Khammari A, et al. Development of squamous cell carcinoma into basal cell carcinoma under treatment with vismodegib. J Eur Acad Dermatol Venereol 2015;29:1006–9.
Iarrobino A, Messina JL, Kudchadkar R, Sondak VK. Emergence of a squamous cell carcinoma phenotype following treatment of metastatic basal cell carcinoma with vismodegib. J. Am. Acad. Dermatol. 2013;69:e33–4.
Orouji A, Goerdt S, Utikal J, Leverkus M. Multiple highly and moderately differentiated squamous cell carcinomas of the skin during vismodegib treatment of inoperable basal cell carcinoma. Br. J. Dermatol. 2014;171:431–3.
Bhutani T, Abrouk M, Sima CS, Sadetsky N, Hou J, Caro I, et al. Risk of cutaneous squamous cell carcinoma after treatment of basal cell carcinoma with vismodegib. J Am Acad Dermatol 2017;77:713–8. https://doi.org/10.1016/j.jaad.2017.03.038.
Ben Ishai M, Tiosano A, Fenig E, Ben Simon G, Yassur I. Outcomes of vismodegib for periocular locally advanced basal cell carcinoma from an open-label trial. JAMA Ophthalmol. 2020;138:749–55.
Ally MS, Aasi S, Wysong A, Teng C, Anderson E, Bailey-Healy I, et al. An investigator-initiated open-label clinical trial of vismodegib as a neoadjuvant to surgery for high-risk basal cell carcinoma. J. Am. Acad. Dermatol. 2014;71:904–11.e1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Curragh, D.S., Huilgol, S.C. & Selva, D. Neoadjuvant vismodegib in the management of locally advanced periocular basal cell carcinoma. Eye 35, 2740–2745 (2021). https://doi.org/10.1038/s41433-020-01291-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-020-01291-2
This article is cited by
-
Mikroskopisch kontrolliert exzidieren
Deutsche Dermatologie (2024)