Abstract
Background
Oral antiandrogen therapies are predominantly used in older men, but real-life studies evaluating the impact of age on pharmacokinetic exposure are lacking. This study aims to evaluate the impact of age on the pharmacokinetic profiles of abiraterone acetate and enzalutamide in clinical practice.
Patients and methods
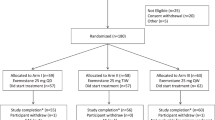
Retrospective observational study to evaluate the impact of age on the first steady-state sample of patients treated with abiraterone acetate or enzalutamide in routine daily clinical practice. The effect of age on target attainment was assessed.
Results
For abiraterone acetate and enzalutamide, 71 and 64 patients were included, respectively. Baseline patients’ characteristics and administered doses were not age-dependent. No age-related differences were observed in exposure to the main metabolites of abiraterone acetate, except for active metabolite Δ(4)-Abiraterone (D4A) with a median plasma concentration of 2.5 × 10−3 mg/L in the oldest versus 1.3 × 10−3 mg/L in the youngest age quartile (coefficient of variation, CV, 72%, p = 0.03). For enzalutamide, no significant differences in exposure were found, except for carboxylic acid enzalutamide, having a median plasma concentration of 5.8 mg/L versus 3.9 mg/L in the oldest versus the youngest age quartile (CV 66%, p = 0.03). However, this was driven by one patient aged 99 years old. Age had no significant influence on target attainment of either compound.
Conclusions
This study showed no significant impact of age on the pharmacokinetic profiles of abiraterone acetate and enzalutamide, except for the active metabolite D4A and the inactive metabolite carboxylic acid enzalutamide, both having significantly higher exposure in older males. Target attainments of abiraterone and enzalutamide were not significantly affected by age, which suggests that age has no clinically relevant impact on exposure to these oral antiandrogen therapies. However, the clinical impact of higher exposure to D4A in older males remains undetermined.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Fitzmaurice C, Allen C, Barber RM, Barregard L, Bhutta ZA, Brenner H, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990–2015: a systematic analysis for the Global Burden of Disease Study Global Burden. JAMA Oncol. 2017;3:524–48.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7–30.
American Cancer Society. Global Cancer Facts & Figures 3rd Edition. Am. Cancer Soc. 2015. https://doi.org/10.1002/ijc.27711.
US Food and Drug Administration (FDA). Clinical Pharmacology and Biopharmaceutics Review: Zytiga (abiraterone acetate). MD: Silver Spring; 2010. p. 1–86.
Scher HI, Fizazi K, Saad F, Taplin M-E, Sternberg CN, Miller K, et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med. 2012;367:1187–97.
Beer TM, Armstrong AJ, Rathkopf DE, Loriot Y, Sternberg CN, Higano CS, et al. Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med. 2014;371:424–33.
de Bono JS, Logothetis CJ, Molina A, Fizazi K, North S, Chu L, et al. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med. 2011;364:1995–2005.
Ryan CJ, Smith MR, de Bono JS, Molina A, Logothetis CJ, de Souza P, et al. Abiraterone in metastatic prostate cancer without previous chemotherapy. N Engl J Med. 2013;368:138–48.
Fizazi K, Scher HI, Molina A, Logothetis CJ, Chi KN, Jones RJ, et al. Abiraterone acetate for treatment of metastatic castration-resistant prostate cancer: final overall survival analysis of the COU-AA-301 randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 2012;13:983–92.
Szmulewitz RZ, Peer CJ, Ibraheem A, Martinez E, Kozloff MF, Carthon B, et al. Prospective international randomized phase ii study of low-dose abiraterone with food versus standard dose abiraterone in castration-resistant prostate cancer. J Clin Oncol. 2018;36:1389–95.
Acharya M, Gonzalez M, Mannens G, De Vries R, Lopez C, Griffin T, et al. A phase I, open-label, single-dose, mass-balance study of 14C-labeled abiraterone acetate in healthy male subjects. Xenobiotica. 2013;43:379–89.
Li Z, Bishop AC, Alyamani M, Garcia JA, Dreicer R, Bunch D, et al. Conversion of abiraterone to D4A drives antitumour activity in prostate cancer. Nature. 2015;523:347–51.
Emamekhoo H, Li Z, Sharifi N. Clinical significance of D4A in prostate cancer therapy with abiraterone. Cell Cycle. 2015;14:3213–4.
Carton E, Noe G, Huillard O, Golmard L, Giroux J, Cessot A, et al. Relation between plasma trough concentration of abiraterone and prostate-specific antigen response in metastatic castration-resistant prostate cancer patients. Eur J Cancer. 2017;72:54–61.
US Food and Drug Administration. Prescribing information: Xtandi (enzalutamide). Maryland: Silver Spring, US Food and Drug Administration; 2012.
US Food and Drug Administration. Clinical Pharmacology and Biopharmaceutics Review: Xtandi (Enzalutamide). MD: Silver Spring; 2012. p. 1–75.
Scher HI, Beer TM, Higano CS, Anand A, Taplin M-E, Efstathiou E, et al. Antitumour activity of MDV3100 in castration-resistant prostate cancer: a phase 1–2 study. Lancet. 2010;375:1437–46.
Groenland SL, van Nuland M, Verheijen RB, Schellens JHM, Beijnen JH, Huitema ADR et al. Therapeutic drug monitoring of oral anti-hormonal drugs in oncology. Clin Pharmacokinet 2018. https://doi.org/10.1007/s40262-018-0683-0.
Smith MR, Rathkopf DE, Mulders PFA, Carles J, Van Poppel H, Li J, et al. Efficacy and safety of abiraterone acetate in elderly (75 years or older) chemotherapy-naive patients with metastatic castration-resistant prostate cancer. J Urol. 2015;194:1277–84.
Mulders PFA, Molina A, Marberger M, Saad F, Higano CS, Chi KN, et al. Efficacy and safety of abiraterone acetate in an elderly patient subgroup (aged 75 and older) with metastatic castration-resistant prostate cancer after docetaxel-based chemotherapy. Eur Urol. 2014;65:875–83.
Siemens DR, Klotz L, Heidenreich A, Chowdhury S, Villers A, Baron B, et al. Efficacy and safety of enzalutamide vs bicalutamide in younger and older patients with metastatic castration-resistant prostate cancer in the TERRAIN trial. J Urol. 2018;199:147–54.
Graff JN, Baciarello G, Armstrong AJ, Higano CS, Iversen P, Flaig TW, et al. Efficacy and safety of enzalutamide in patients 75 years or older with chemotherapy-naive metastatic castration-resistant prostate cancer: results from PREVAIL. Ann Oncol. 2016;27:286–94.
Sternberg CN, de Bono JS, Chi KN, Fizazi K, Mulders P, Cerbone L, et al. Improved outcomes in elderly patients with metastatic castration-resistant prostate cancer treated with the androgen receptor inhibitor enzalutamide: results from the phase III AFFIRM trial. Ann Oncol. 2014;25:429–34.
Dawson A, Dennison E. Measuring the musculoskeletal aging phenotype. Maturitas. 2016;93:13–7.
Seripa D, Panza F, Daragjati J, Paroni G, Pilotto A. Measuring pharmacogenetics in special groups: geriatrics. Expert Opin Drug Metab Toxicol. 2015;11:1073–88.
Van Nuland M, Hillebrand MJX, Rosing H, Schellens JHM, Beijnen JH Development and validation of an LC-MS/MS method for the simultaneous quantification of abiraterone, enzalutamide, and their major metabolites in human plasma. Ther Drug Monit. 2017; 39. https://doi.org/10.1097/FTD.0000000000000387.
European Medicines Agency. European Public Assessment Report (EPAR). London: Zytiga (Abiraterone Acetate); 2016.
Acharya M, Gonzalez M, Mannens G, De Vries R, Lopez C, Griffin T, et al. A phase I, open-label, single-dose, mass-balance study of 14C-labeled abiraterone acetate in healthy male subjects. Xenobiotica. 2012;43:1–11.
Wang Y, Chia YL, Nedelman J, Schran H, Mahon F-X, Molimard M. A therapeutic drug monitoring algorithm for refining the imatinib trough level obtained at different sampling times. Ther Drug Monit. 2009;31:579–84.
Jones GRD, Lim. E-M. The National Kidney Foundation Guideline on estimation of the glomerular filtration rate. Clin Biochem Rev. 2003;24:95–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Crombag, MR.B.S., van Nuland, M., Bergman, A.M. et al. Impact of age on exposure to oral antiandrogen therapies in clinical practice. Prostate Cancer Prostatic Dis 22, 168–175 (2019). https://doi.org/10.1038/s41391-018-0096-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-018-0096-z
This article is cited by
-
The impact of patient characteristics on enzalutamide pharmacokinetics and how this relates to treatment toxicity and efficacy in metastatic prostate cancer patients
Cancer Chemotherapy and Pharmacology (2020)