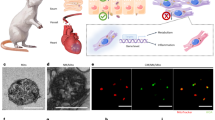
Abstract
Background
Bariatric surgeries have been shown to be effective in reversing damaged pulmonary function in individuals suffering from obesity and type 2 diabetes mellitus, whereas its underlying mechanisms remain largely unknown.
Methods
Sleeve gastrectomy (SG) was performed on obese and diabetic Wistar rats, and their pulmonary function and lung tissues were compared to sham-operated (SH) obese and diabetic rats, and age-matched healthy controls (C) to explore the improvements in microstructures and expression of surfactant protein (SP)-A and -C at postoperative 4th, 8th, and 12th week.
Result
Apart from the profound metabolic changes and improvement in pulmonary function, lung volume was restored along with an improved diffusion capacity noted by thinned capillary basement membrane and decreased harmonic mean length of diffusion barrier in SG rats. The digital slices of light microscope showed the general changes brought on by the SG, including normalized basic structures, ameliorated inflammatory status, as well as reduced lipid deposition, where the hydroxyproline (HYP), triglyceride (TG) assays, and electron microscope further suggested that the improvement in alveolar structures lies in reduced collagen fibers, lipids and septal tissues, increased capillary blood, and normalized alveolar type 2 (AT2) cells. Besides, disrupted SP-A and SP-C expression were also normalized after SG.
Conclusion
The improvement of lung function after SG is related to the ameliorated alveolar structures, and surface protein expression induced by weight loss and improved glucose metabolism.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Research data of current study is available at R.R., J.L., Q.X., M.Z., Y.X., Z.Y., J.Z., Y.C., Z.M., S.H., G.Z. (2020), “Data for: Sleeve gastrectomy ameliorates alveolar structures and surfactant protein expression in lungs of obese and diabetic rats”, Mendeley Data, V2, https://doi.org/10.17632/5nn793z99j.2.
References
Prentki M, Nolan CJ. Islet β cell failure in type 2 diabetes. J Clin Invest. 2006;116:1802–12.
Harding HP, Ron D. Endoplasmic reticulum stress and the development of diabetes. a review. Diabetes. 2002;51(Suppl 3):S455–61.
Sandler M. Is the lung a ‘target organ’ in diabetes mellitus? JAMA Int Med. 1990;150:1385–8.
Conway BN, May ME, Fischl A, Frisbee J, Han X, Blot WJ. Cause-specific mortality by race in low-income Black and White people with Type 2 diabetes. Diabet Med. 2015;32:33–41.
van den Borst B, Gosker HR, Zeegers MP, Schols AMWJ. Pulmonary function in diabetes: a metaanalysis. Chest. 2010;138:393–406.
Chatila WM, Thomashow BM, Minai OA, Criner GJ, Make BJ. Comorbidities in chronic obstructive pulmonary disease. Proc Am Thorac. Soc. 2008;5:549–55.
Chance WW, Rhee C, Yilmaz C, Dane DM, Pruneda ML, Raskin P, et al. Diminished alveolar microvascular reserves in type 2 diabetes reflect systemic microangiopathy. Diabetes Care. 2008;31:1596–601.
Hsia CC, Raskin P. The diabetic lung: relevance of alveolar microangiopathy for the use of inhaled insulin. Am J Med. 2005;118:205–11.
Niranjan V, McBrayer DG, Ramirez LC, Raskin P, Hsia CC. Glycemic control and cardiopulmonary function in patients with insulin-dependent diabetes mellitus. Am J Med. 1997;103:504–13.
Schuyler MR, Niewoehner DE, Inkley SR, Kohn R. Abnormal lung elasticity in juvenile diabetes mellitus. Am Rev Respir. Dis. 1976;113:37–41.
Sandler M, Bunn AE, Stewart RI. Cross-section study of pulmonary function in patients with insulin-dependent diabetes mellitus. Am Rev Respir Dis. 1987;135:223–9.
Goldman MD. Lung dysfunction in diabetes. Diabetes Care. 2003;26:1915–8.
Yeh H-C, Punjabi NM, Wang N-Y, Pankow JS, Duncan BB, Cox CE, et al. Cross-sectional and prospective study of lung function in adults with type 2 diabetes: the Atherosclerosis Risk in Communities (ARIC) studys. Diabetes Care. 2008;31:741–6.
Weynand B, Jonckheere A, Frans A, Rahier J. Diabetes mellitus induces a thickening of the pulmonary basal lamina. Respiration. 1999;66:14–9.
López-Cano C, Lecube A, García-Ramírez M, Muñoz X, Sánchez E, Seminario A, et al. Serum surfactant protein D as a biomarker for measuring lung involvement in obese patients with type 2 diabetes. J Clin Endocrinol Metab. 2017;102:4109–16.
Coya JM, Akinbi HT, Sáenz A, Yang L, Weaver TE, Casals C. Natural anti-infective pulmonary proteins: in vivo cooperative action of surfactant protein SP-A and the lung antimicrobial peptide SP-BN. J Immunol. 2015;195:1628–36.
Kadoya C, Lee B-W, Ogami A, Oyabu T, Nishi K-i, Yamamoto M, et al. Analysis of pulmonary surfactant in rat lungs after inhalation of nanomaterials: Fullerenes, nickel oxide and multi-walled carbon nanotubes. Nanotoxicology. 2016;10:194–203.
Foster DJ, Ravikumar P, Bellotto DJ, Unger RH, Hsia CC. Fatty diabetic lung: altered alveolar structure and surfactant protein expression. Am J Physiol Lung Cell Mol Physiol. 2010;298:L392–403.
Hsia CC, Raskin P. Lung function changes related to diabetes mellitus. Diabetes Technol Ther. 2007;9 Suppl 1:S73–82.
Sjostrom L, Narbro K, Sjostrom CD, Karason K, Larsson B, Wedel H, et al. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007;357:741–52.
Campos EC, Peixoto-Souza FS, Alves VC, Basso-Vanelli R, Barbalho-Moulim M, Laurino-Neto RM, et al. Improvement in lung function and functional capacity in morbidly obese women subjected to bariatric surgery. Clinics (Sao Paulo). 2018;73:e20.
Onofre T, Carlos R, Oliver N, Felismino A, Fialho D, Corte R, et al. Effects of a physical activity program on cardiorespiratory fitness and pulmonary function in obese women after bariatric surgery: a pilot study. Obes Surg. 2017;27:2026–33.
Arismendi E, Rivas E, Vidal J, Barreiro E, Torralba Y, Burgos F, et al. Airway hyperresponsiveness to mannitol in obesity before and after bariatric surgery. Obes Surg. 2015;25:1666–71.
van Huisstede A, Rudolphus A, Castro Cabezas M, Biter LU, van de Geijn GJ, Taube C, et al. Effect of bariatric surgery on asthma control, lung function and bronchial and systemic inflammation in morbidly obese subjects with asthma. Thorax. 2015;70:659–67.
Gheibi S, Kashfi K, Ghasemi A. A practical guide for induction of type-2 diabetes in rat: Incorporating a high-fat diet and streptozotocin. Biomed Pharmacother. 2017;95:605–13.
Unger RH. Lipotoxic diseases. Annu Rev Med. 2002;53:319–36.
Yilmaz C, Ravikumar P, Bellotto DJ, Unger RH, Hsia CC. Fatty diabetic lung: functional impairment in a model of metabolic syndrome. J Appl Physiol. 2010;109:1913–9.
Winck AD, Heinzmann-Filho JP, Soares RB, da Silva JS, Woszezenki CT, Zanatta LB. Effects of obesity on lung volume and capacity in children and adolescents: a systematic review. Rev Paul Pediatr. 2016;34:510–7.
Santos A, Rivas E, Rodríguez-Roisin R, Sánchez M, Ruiz-Cabello J, Arismendi E, et al. Lung tissue volume is elevated in obesity and reduced by bariatric surgery. Obes Surg. 2016;26:2475–82.
Carlson EC, Audette JL, Veitenheimer NJ, Risan JA, Laturnus DI, Epstein PN. Ultrastructural morphometry of capillary basement membrane thickness in normal and transgenic diabetic mice. Anat Rec A Discov Mol Cell Evol Biol. 2003;271:332–41.
Lash JM, Sherman WM, Hamlin RL. Capillary basement membrane thickness and capillary density in sedentary and trained obese Zucker rats. Diabetes. 1989;38:854–60.
Chakrabarti S, Sima AA. Effect of aldose reductase inhibition and insulin treatment on retinal capillary basement membrane thickening in BB rats. Diabetes. 1989;38:1181–6.
Burri PH, Dbaly J, Weibel ER. The postnatal growth of the rat lung. I. Morphometry. Anat Rec. 1974;178:711–30.
Zavorsky GS, Kim DJ, Sylvestre JL, Christou NV. Alveolar-membrane diffusing capacity improves in the morbidly obese after bariatric surgery. Obes Surg. 2008;18:256–63.
Unger RH, Orci L. Lipotoxic diseases of nonadipose tissues in obesity. Int J Obes Relat Metab Disord. 2000;24 Suppl 4:S28–32.
Mason RJ. Biology of alveolar type II cells. Respirology. 2006;11:S12–5.
Brown LA, Longmore WJ. Altered phospholipid secretion in type II pneumocytes isolated from streptozotocin-diabetic rats. Biochim Biophys Acta. 1986;878:258–65.
Sugahara K, Ushijima K, Morioka T, Usuku G. Studies of the lung in diabetes mellitus. I. Ultrastructural studies of the lungs in alloxan-induced diabetic rats. Virchows Arch A Pathol Anat Histol. 1981;390:313–24.
Boehme DS, Roberts CM, Bessman SP. High-performance liquid chromatographic analysis of lung phospholipids and their precursors in the offspring of diabetic rats. Biochem Med Metab Biol. 1990;44:29–36.
Moore TR. A comparison of amniotic fluid fetal pulmonary phospholipids in normal and diabetic pregnancy. Am J Obstet Gynecol. 2002;186:641–50.
Seki Y, Kasama K, Haruta H, Watanabe A, Yokoyama R, Porciuncula JPC, et al. Five-year-results of laparoscopic sleeve gastrectomy with duodenojejunal bypass for weight loss and type 2 diabetes mellitus. Obes Surg. 2017;27:795–801.
Camastra S, Vitali A, Anselmino M, Gastaldelli A, Bellini R, Berta R, et al. Muscle and adipose tissue morphology, insulin sensitivity and beta-cell function in diabetic and nondiabetic obese patients: effects of bariatric surgery. Sci Rep. 2017;7:9007.
Acknowledgements
This project was supported by the National Natural Science Foundation of China (NSFC, Grant Nos 81370496 and 81873647) and Youth Program of National Natural Science Foundation of China (Grant No. 81600059)
Author information
Authors and Affiliations
Contributions
G.Z. and J.L. conceived the experiments. G.Z., Z.Y., and J.Z. designed the experiment. Q.X. and M.Z. did the literature research. R.R. and Y.X. carried out the experiment. Y.C. and Z.M. performed the statistical analysis. Manuscript was prepared by R.R. S.H. and G.Z. had the final approval of the submitted and published version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All study protocols were approved by the Animal Care and Utilization Committee of Shandong University.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Ruze, R., Li, J., Xu, Q. et al. Sleeve gastrectomy ameliorates alveolar structures and surfactant protein expression in lungs of obese and diabetic rats. Int J Obes 44, 2394–2404 (2020). https://doi.org/10.1038/s41366-020-0647-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-020-0647-y
This article is cited by
-
Ectopic and visceral fat deposition in aging, obesity, and idiopathic pulmonary fibrosis: an interconnected role
Lipids in Health and Disease (2023)
-
Multiple Beneficial Effects of Laparoscopic Sleeve Gastrectomy for Patients with Obesity, Type 2 Diabetes Mellitus, and Restrictive Ventilatory Dysfunction
Obesity Surgery (2022)