Abstract
An element-block is defined as a minimum functional unit composed of heteroatoms. The concept of ‘element-block material’ for designing advanced materials was proposed in 2015. In this review, the recent progress in material designs based on this concept is described. From our research, several element-blocks as representative examples are selected, and their roles in each material are explained. Initially, research on the development of ‘designable hybrids’ by employing polyhedral oligomeric silsesquioxane (POSS) is illustrated. Multiple functions of hybrids composed of modified POSS and the significant roles of POSS in bio-related polymeric materials for solving critical problems in conventional 19F MR probes and for realizing new sensing technologies are demonstrated. Next, solid-state luminescent materials containing o-carborane and group 13 element-blocks are discussed. The materials’ highly efficient emission in the solid state and their origins are explained. Furthermore, stimuli-responsive luminescent chromism is summarized. The sensing mechanisms and the application of these materials as sensors are presented in this study. In the last part, we mention optically active element-blocks containing chiral P-stereogenic phosphorus. In this review, the characteristics originating from each element-block are explained.
Similar content being viewed by others
Introduction
A new concept for material designs as an ‘element-block material’ was proposed in 2015.1 The minimum functional unit containing heteroatoms is defined as an ‘element-block’, and the creation of advanced ‘organic–inorganic hybrids’ has been attempted by linking, combining and assembling element-blocks. The production of functional devices was also proposed by the organization of ‘element-block materials’ according to the preprogrammed design. The discovery of brilliant new functions exceeding each component and the establishment of tactics for material designs based on inorganic polymers are expected as research progresses. Indeed, a wide variety of elements in the periodic table have been applied in the development of element-blocks, and superior properties to conventional materials have been obtained. In this review, we introduce recent progress in the development of element-block materials. Several characteristic examples are demonstrated, mainly from our work. As representative element-blocks, we select polyhedral oligomeric silsesquioxane (POSS), o-carborane, group 13 element complexes and chiral phosphine compounds and illustrate their roles in the material properties (Figure 1). Initially, the development of ‘designable hybrids’ employing POSS is presented. By regarding POSS as an inorganic element-block, polymer hybrids can be prepared simply by mixing with POSS derivatives and polymers. Based on this idea, we successfully produced refractive-index materials keeping the trade-off relationship at a high level in the material properties and created new hybrids possessing different thermal behaviors. The next topic is concerned with solid-state luminescent materials containing o-carborane and group 13 element-blocks. It was found that several conjugated compounds involving these element-blocks showed bright solid-state emission. Furthermore, these luminescent properties were drastically influenced by external stimuli. The property mechanisms and applications as a stimuli-responsive luminescent chromic material are presented here. We also mention chiral element-blocks, and the characteristics originating from these structural features are explained.
POSS element-blocks for realizing designable hybrids
From recent advances in synthetic chemistry, various applications of organic molecules to electronic devices have been realized. For example, conjugated polymers have been recognized as a key material in organic opto-electronic devices and applied for fabricating film-type photovoltaic cells and flexible electroluminescent displays.2 In addition, because of their applicability to printing methods, low-cost fabrication processes have been explored based on these functional organic polymers.3 However, there remain large requirements for improving the durability of these functional polymers because of intrinsic stabilities lower than those of inorganics. By mixing polymers and inorganic components at the nanoscale, organic–inorganic polymer hybrids can be obtained.4 These materials commonly showed thermal stability and toughness that originated from the inorganic components, as well as the intrinsic characteristics of the polymers. Therefore, hybridization is a valid strategy for improving the robustness of conventional polymers. Based on these advantages, various types of functional hybrids have been prepared such as white-light emissive materials,5 environmental-resistant electric conductors,6 heat generators toward microwave irradiation,7, 8 molecular carriers with a light-driven releasing ability9 and oxygen-resistant phosphorescent emitters.10 However, because of the commonly low compatibility between organic polymers and inorganic components, it has often been difficult to realize homogeneous mixing at the nanolevel without specific modifications such as the introduction of polar substituents into polymers and/or hydrophobic moieties to inorganic components.11, 12 Thus, there are still difficulties in the preparation of hybrids with highly functional modern polymers.
POSS has been regarded as a molecular hybrid because of its structural feature, and a large number of smart materials have been obtained based on POSS.13, 14, 15, 16, 17, 18, 19, 20, 21 If the POSS derivative can be mixed into polymer matrices, a similar situation to that in a hybrid material, where the inorganic components are dispersed at the nanometer or molecular scale, would be obtained in conventional polymer hybrids (Figure 2). Therefore, it can be said that POSS is the element-block for easily fabricating a hybrid without sol–gel methods. Moreover, by loading functional groups on POSS and by controlling the morphology of POSS in the materials, the addition of further properties can be expected, according to the preprogrammed design. Thus, POSS can be regarded as an element-block for producing ‘designable hybrids’. Indeed, in previous reports, the POSS-based designable hybrid materials were prepared just by mixing POSS derivatives and polymers in solution, and significant improvements of thermal and mechanical properties were accomplished.22, 23 Furthermore, by modifying POSS, the trade-off relationships were maintained between multiple competing functions in the materials.24, 25, 26, 27, 28 In this part, the preparation of ‘designable hybrids’ by utilizing modified POSS and the material properties are described.
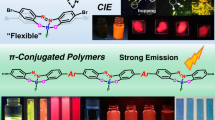
Initially, POSS element-blocks were applied to enhance the material properties of conjugated polymers, which are the key component in modern opto-electronic devices.2, 3 As we mention above, the enhancement of polymer components in organic devices is strongly required. However, it has been challenging to find a molecular filler that can reinforce thermal stability without critical losses of the opto-electronic properties of the functional polymers used in these devices. To examine the basic affinity of POSS, hybrid films using conventional conjugated polymers such as polyfluorene and poly(3-hexylthiophene) (P3HT) as matrices were prepared (Figure 3).29 Interestingly, POSS fillers showed high miscibility in both matrices. All POSS fillers were able to be loaded to over 40 wt%, and it was revealed that the thermal stability of the films was significantly improved. This fact means that POSS can work as an element-block for realizing hybrid materials with commodity conjugated polymers. In addition, the influence of a series of alkyl- and phenyl-substituted POSS on the optical properties was evaluated with respect to loading. Accordingly, homogeneity in the hybrid materials was efficiently improved. In the polyfluorene hybrids, sharp emission bands were obtained in the blue region by suppressing main-chain aggregation. Moreover, in the poly(3-hexylthiophene) films, it was proposed that the extension of the main-chain conjugation was facilitated by the POSS fillers. It was summarized that the POSS fillers can have a significant role in the improvement of homogeneity in the polymer films without losses of thermal stability because of the fillers’ high affinity to the conjugated systems.
Alkyl and aryl-substituted POSS derivatives usually showed good solubility in common organic solvents. However, because of the highly symmetric structures of a silica cube, aggregation and crystallization often proceeded in the polymer matrices, leading to a critical phase separation in the materials. Thus, the next challenge for obtaining larger filler effects was improvement of the miscibility of POSS fillers with commodity polymers. The modified POSS derivatives were synthesized via in situ sol–gel reactions with mixtures containing two types of alkyl-silanes (Figure 4).30 As a result, POSS derivatives having different types of alkyl substituents were obtained. The products had molecular distributions originating from the formation of cubic and pentagonal prism structures and the variable introduction ratios of alkyl substituents. Because of the reduction in symmetry, the synthesized POSS fillers presented high miscibility in PMMA. As an application, it was demonstrated that POSS fillers with dual types of substituents can lower the refractive index of the PMMA matrix and enhance thermal and mechanical properties.
Chemical structures of POSS fillers having different types of alkyl substituents and changes in refractive indices (Δn), the thermal decomposition temperature with 20 wt% weight loss (ΔTd20) and rigidity of poly(methyl methacrylate) (ΔE′) by filler addition. (iC4+CP), (iC4+C18) and (CP+C18) indicates the modified POSS having different types of alkyl chains in the single core, and iC4&CPmix, iC4&C18mix and CP&C18mix were prepared by simply mixing with each octa-substituted POSS in the matrix (iC4: isobutyl, CP: cyclopentyl, C18: octadecyl groups). n.d.=not detectable because of low film-formability.
It is clearly indicated from the above research that POSS element-blocks can be used as a filler for modulating polymer properties. The next topic is on the POSS filler for enhancing thermal properties with the new matrices that had never been used as a hybrid before. Ionic liquids (ILs), which are defined as an ion salt having a melting temperature below 100 °C, have attracted much attention because of their versatility in a wide variety of research fields and industrial applications, such as an electrolyte in lithium batteries and fuel cells.31 Therefore, to extend the lifetime of these devices, the enhancement of the thermal stability of these media is required. However, it remains difficult to invent novel ILs with improved thermal stability and no significant changes in the other functions of the ILs simply by altering the chemical structures of the components because the thermal properties of ILs are drastically changed upon altering the chemical structure. One simple procedure to maintain both properties of thermal stability and intrinsic functions could be the use of fillers, as we mention above. Therefore, we sought to develop molecular fillers to readily obtain thermally stable ILs with other characteristics.
It has been reported that the enhancement of thermal stability and minimizing changes to the melting temperature of an ion pair can be simultaneously obtained by tethering to the POSS core via covalent bonds.32, 33, 34 As a result, room-temperature ILs, which are defined as ionic salts with a melting temperature below 25 °C, were produced based on POSS. Based on these characteristics of POSS ILs, we next sought to transmit the POSS effect on thermal properties to the IL matrices via non-covalent bonds (Figure 5). By hydrogen bond formation between the acid functional groups in the POSS and the ionic salts, we presumed that crystallinity should be significantly reduced, and a reduction in the melting temperature of the ionic salts could then be observed. Furthermore, thermal stability should be enhanced by the POSS core. To evaluate the validity of this idea, we designed and synthesized the carboxylic and sulfonic acid-presenting POSS derivatives.35, 36 In particular, the superior effects of the POSS filler with sulfonic acid (POSS-[SO3H]8) is explained.
The desired POSS was prepared in good yields, and good dispersions in various kinds of conventional ionic compounds were obtained.36 From thermal measurements, it was found that POSS-[SO3H]8 had significant effects on both decreasing the melting temperature and enhancing the thermal stability of a wide variety of ionic compounds, as we proposed. The calculation of the thermodynamic parameters during the melting process revealed lowering effects on transition enthalpy and entropy by the introduction of the POSS filler, indicating that the rigid cubic and symmetric structure of POSS played significant roles in improving the thermal properties. In addition, these effects were lost upon adding alkyl- and ammonium-POSS and the sulfonic acid without POSS. These results suggested that the formation of hydrogen bonds with ionic salts and the POSS core should be necessary for expressing the enhancement in thermal properties. Finally, the melting temperature of 1,2,3-Φethylimidazolium methanesulfonate (TriMIM-MeSO4) was lowered from 122 to 94 °C by POSS-[SO3H]8. These data suggest that POSS-[SO3H]8 can work as a filler for transforming an ion salt, as a representative IL. From these results, the idea that hybrid formation can be extended to other matrices apart from polymers by employing the POSS filler is revealed, indicating the versatility of POSS element-blocks.
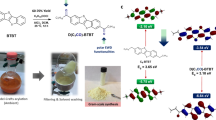
To demonstrate further applicability of the idea on ‘POSS-based designable hybrids’, bio-related materials containing POSS and their characteristics are reviewed. It was shown that water-soluble POSS materials can encapsulate hydrophobic molecules inside these materials by adsorption onto the POSS surface via van der Waals interactions that originated from the extensive low polarity of the cubic silica core.37, 38, 39, 40, 41, 42, 43 Based on this character, a detection system with 19F nuclear magnetic resonance (NMR) probes was constructed for bio-active amine compounds at sub-micromolar concentrations (Figure 6).44 The application of 19F NMR probes for the monitoring of biological events and reactions has been well known as a valid strategy for obtaining detailed information at deep spots inside vital bodies.45, 46, 47, 48, 49, 50, 51 To date, although various types of molecular probes and detection systems have been manufactured for evaluating biological events and alteration of environmental factors, the monitoring of the bioreactions with 19F NMR probes commonly suffers from an intrinsically low sensitivity in 19F NMR measurements. Therefore, it was difficult to monitor slight amounts of bio-active molecules. Water-soluble fluorinated POSS derivatives were prepared, and the paramagnetic NiPor (Ni-porphyrin) was adsorbed with F-POSS in the buffer.44 By adding a series of amine derivatives to the homogeneous dispersion containing the POSS-NiPor complexes, the changes in the signal intensity were evaluated. It was clear that the signal intensity of 19F NMR decreased especially in the presence of ethanediamine and hexylamine. From mechanical studies, it was suggested that the assembly of the F-POSS complexes should be induced by the amine compounds, and the signal reduction of 19F NMR from the POSS-NiPor complexes could be induced via the strong paramagnetic relaxation enhancement from NiPor in the assembly. We were able to detect the amine derivatives, which show critical effects as a signal transducer or a stimulant drug, at concentrations under 1 μM, similar in sensitivity to conventional fluorescence probes.52, 53 It was expected that this system should be valid to monitor the distribution of bio-active molecules at cellular concentrations.
The superior adsorption ability of the cubic silica in the POSS element-block was utilized for recognizing the shape of nanomaterials. It was shown that the encapsulation ability can be enhanced in the POSS network polymers.54, 55, 56, 57 Based on this fact, POSS networks were designed and synthesized with octaammonium POSS by connecting with bithiophene dicarboxylic acid (Figure 7).58 By modulating the feed ratio of the bithiophene linker, a series of the POSS networks were obtained with various cross-linking ratios among the POSS units. The resulting networks showed good water solubility, and a strong luminescence originated from the bithiophene linkers in water. The POSS networks with relatively low cross-linking ratios showed a higher affinity to silica particles. Interestingly, it was shown that the emission properties were altered simply by adding the silica particles into the sample. It should be noted that the redshifted emission of the POSS network was induced in the presence of the nanoparticles dispersed in the sample, whereas the blueshifted emission was observed from the samples containing the microparticles. These peak shifts depended on the size of coexisting particles and should originate from polarity changes around the bithiophene cross-linkers induced by the adsorption of the POSS networks onto the particle surface. These emission characteristics are applicable for not only detecting water pollution by particles but also discriminating the size of the nanomaterials.
POSS-containing networks were used as a scaffold for molecular recognition with structural isomers of significant compounds.54, 55, 56, 57 A class of trans-fatty acids has been recognized as a harmful byproduct in food processing according to recent medical reports.59, 60 Therefore, the development of facile detection methods for trans-fatty acid in foods is urgently needed. However, it is still challenging to construct molecular probes for trans-fatty acids because the olefin moiety generally only slightly interacts with the probe molecules. Thus, we sought to establish a molecular recognition system via van der Waals interactions with POSS to perform trans-fatty acid discrimination using fluorescence spectroscopy (Figure 8).61 The water-soluble POSS network polymers connected with triphenylamine derivatives (were prepared, and their sensing ability for discriminating the geometric isomers of fatty acids was examined. In the photoluminescence spectra, different time-courses of the intensity and peak wavelength of the emission band were observed from the triphenylamine derivatives-containing solution in the presence of cis- or trans-fatty acids during incubation. In particular, variable time-dependent changes were obtained by changing coexisting ratios between two geometric isomers. From the mechanistic studies, it was implied that these changes could be derived from the difference in the degree of interaction between the POSS network and each fatty acid.
We demonstrate here recent examples on the development of ‘designable hybrids’ based on POSS element-blocks. By loading expected functional groups on the POSS unit, polymeric materials including dendritic molecules and networks were constructed. Finally, it was revealed that these ‘element-block polymers’ provided various useful functions for not only solving problems in conventional materials but also creating new materials. Thus, we believe that ‘designable hybrids’ still have huge potential as a platform for realizing superior functions.
Carborane element-blocks for solid-state luminescent materials having stimuli-responsivity
As POSS is composed of silica, significant electronic properties were hardly obtained. Next, we explain the cluster-type element-block possessing optical and electronic properties originating from a steric conjugation system. o-Carborane is an icosahedral cluster composed of two carbon and ten boron atoms and has attracted attention as a versatile component for constructing luminescent materials (Figure 1).62 All atoms are linked via the three-center-two-electron bonds, and 3D-electronic conjugation is developed through a whole molecule. Therefore, o-carborane has high stability. In contrast, o-carborane shows characteristic behavior by excitation. The o-carborane unit can play a role as a strong electron acceptor in the excited state of conjugated systems originating from the electron-deficient nature of the σ*–π* conjugation on the C–C bond of the o-carborane unit. Therefore, by combination with various aromatic rings, the intramolecular charge transfer (ICT) state could be readily developed in the excited state. Then, bright emission from the ICT state was obtained.63 Furthermore, similar to other donor-acceptor systems, it was possible to tune the luminescent color by modulating the electron donation at the conjugated aromatic moieties. Thus, o-carborane has attracted significant attention as a luminescence-inducible element-block.
There are three significant characteristics of o-carborane in luminescent molecules (Figure 9). First, the o-carborane unit often contributed to avoiding aggregation-caused quenching (ACQ).64 Commodity organic luminescent dyes usually present intense emission in dilute solutions, whereas most of luminescent properties are critically spoiled in the condensed state because of the ACQ effect. Because of the steric hindrance originating from the spherical shape of o-carborane, intermolecular interactions, which is one of major processes to the ACQ, should be disturbed. As a result, the intense ICT emission could be preserved even in the condensed and crystalline states.64 Second, the luminescent properties of o-carborane-containing conjugated systems are sensitive toward molecular vibrations. As a typical example, the optical properties of a series of phenyl-substituted o-carborane derivatives are presented.63 In the absence of any structural restriction around the o-carborane unit, a slight emission was observed in dilute solutions. In contrast, a bright emission was detected in the aggregation, as energy-consumable molecular motions should be prohibited. Electronic conjugation was constructed involving the σ*–π* conjugation on the C–C bond in the o-carborane unit. In the solution state, molecular vibrations at the C–C bond in the excited state occurred, and an excitation deactivation was critically induced. On the other hand, molecular vibration should be suppressed in the condensed state. This aggregation-induced emission (AIE) behavior is useful for designing environment-responsive optical materials.65 Third, it was suggested that the electron-accepting ability of o-carborane should be dependent on the dihedral angle between the direction of the C–C bond and the plane of the π-aromatic rings.66, 67 When the C–C bond was placed in a parallel conformation, a donor-acceptor interaction was hardly obtained;66 whereas the strongest electron-accepting ability was observed at the perpendicular conformation because the orbital overlap of the σ*–π* conjugation on the C–C bond in the o-carborane unit and π-conjugation on the aromatic ring should be maximized. This fact means that the electronic and optical properties of aromatic-modified o-carboranes can be drastically changed by the rotation at the o-carborane unit (or the aromatic moiety). Based on these fundamental issues, further luminescent properties were found from o-carborane-containing element-block materials.
In some donor-acceptor-type organic compounds, ICT proceeds with conformational changes such as twisting at the bridge between electron-donating and electron-accepting units in the excited state. Sometimes, emission could be observed from this twisted intramolecular charge transfer (TICT) state.68 Because of the environment-sensitive character of ICT emission and the physical motion in the formation of the TICT state, luminescent molecules with TICT emission are used as a versatile platform for constructing bioprobes and environmental sensors. Moreover, by regulating the mobility of the molecules, dual-emission peaks derived from locally excited (LE) and TICT states were simultaneously observed in the single spectrum.68 These behaviors are applicable to construct quantitative detection systems, especially for biological events. However, very few molecules present TICT emission in the solid state because of two critical factors known as concentration quenching and structural restriction. Therefore, applications of luminescent dyes with TICT emission for opto-electronic materials remained a challenge.
The TICT emission from the crystalline state of an anthracene-o-carborane dyad was eventually discovered (Figure 10a).69 The dual-emissive property from LE and TICT states was observed in a dilute solution containing the dyad. In a conventional TICT system, the LE emission usually appears only under a frozen or structurally restricted environmental condition, whereas the dyad presented emission from the LE state of the o-carborane dyad. It was implied that the energy barriers could be small in the rotation of the anthracene moiety. Actually, from variable-temperature photoluminescence measurements, it was shown that the energy barrier in the transition from LE to ICT was allowed at room temperature in the excited state. Next, the solid-state luminescence property of the dyad was investigated. Surprisingly, it was determined that the dyad had both aggregation-induced emission enhancement and crystallization-induced emission enhancement properties attributable to the TICT emission band. From X-ray single-crystal analysis and theoretical calculations, it was suggested that the rotation at the o-carborane unit occurred even in the crystalline state. As a result, a TICT-based crystallization-induced emission enhancement was detected. It was implied that the spherical shape of o-carborane could also have a positive role in the rotation in the excited state.
Highly efficient solid-state emission was obtained from the o-carborane dyads.70 A pyrene-substituted o-carborane dyad was synthesized, and similar to the anthracene-substituted dyad, a dual emission attributable to LE and TICT emission bands in the solution and aggregation-induced emission enhancement and crystallization-induced emission enhancement properties (absolute emission quantum yield: ΦPL=0.40 and 0.80, respectively) attributable to the TICT emission from the solid sample were observed (Figure 10b). To achieve efficient solid-state luminescence, a bulky substituent was introduced to the adjacent carbon atom in the o-carborane unit to freeze the molecular conformation in the perpendicular position and was followed by suppressed energy consumption during rotation in the excited state. Methyl and trimethylsilyl-substituted carborane dyads were prepared, and extremely bright luminescent compounds with solid-state emission properties (ΦPL>0.99) were detected. The color tuning of a highly efficient solid-state emission was also accomplished in the recent result.71 These results clearly indicated that o-carborane should be an element-block for realizing solid-state luminescent materials.
Multi-chromic luminescent behaviors were observed from a di-o-carborane-substituted anthracene triad (Figure 11).72 Recrystallization from CH2Cl2, C6H6 and CHCl3 afforded solvent-incorporated crystals, and each crystal exhibited different emission colors (yellowish-orange, orange and red, respectively). Furthermore, these solvent molecules were able to be removed by heating and adding mechanical stresses, followed by luminescent color changes. These data represent that the triad has solid-state luminescence with vapo-, thermo- and mechanochromic characteristics. From the single-crystal X-ray analyses, it was revealed that the distorted central anthracene moiety formed a π-dimer in the crystal, and the stacking area was significantly changed by the type of incorporated solvents. Therefore, an alteration of the electronic properties was induced.
In this part, the series of luminescent properties of o-carborane derivatives are illustrated. Particularly, it was revealed that o-carborane is a solid-state luminescent element-block. By combining electron-donating groups and stimuli-responsive units, stimuli-responsive luminescent chromism was readily designed. These advantageous characteristics are promised to be a key function of the next generation of opto-electronic devices based on element-block materials.
Conjugated polymeric materials based on group 13 element-blocks
The next topic is concerned with environment-responsive solid-state luminescent materials based on group 13 element-blocks (Figure 1). As we mentioned above, although organoboron complex-containing conjugated polymers have been studied as a versatile substance for fabricating advanced organic opto-electronic devices because of their superior optical properties and carrier-transport abilities,73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84 ACQ problems remain as critical limitations to the practical use of these materials. In 2001, an AIE-active molecule was first reported.85 We also discovered AIE-active boron complexes and built conjugated polymers possessing AIE properties.86, 87 Furthermore, various applications have been accomplished based on the series of organoboron complexes.88, 89, 90, 91, 92 Therefore, we regarded these organoboron complexes as ‘AIE-inducible element-blocks’ to obtain AIE-active materials. In this part, we present recent works on the development of environment-sensitive solid-state emission from these materials.
In previous reports, it was shown that ACQ-presenting common boron complexes could be transformed to the AIE-active molecules.86 By replacing one or both oxygen atoms with nitrogen in boron diketonates, which can show strong luminescence only in the dilute state, the AIE behaviors were induced. Thereby, by employing the resulting molecular structures of boron ketoiminate and diiminate, various types of polymeric materials including polymers, gels and dendritic molecules were generated. It should be mentioned that the AIE properties of these boron complexes could be tuned by introducing functional groups and connecting with other conjugation units.87 This means that the luminescent chromic properties could be designable. The series of triads composed of two boron ketoiminates and a central bithiophene unit with various functional groups at both ends were synthesized (Figure 12).93 As we expected, these triads provided solid-state emission. Interestingly, the emission colors were clearly changed upon grinding the crystalline sample powders to the point the X-ray diffraction peaks disappeared. These behaviors are typical of mechanochromic luminescence, and from X-ray crystal analysis, it was shown that these optical changes should be induced by a crystal-amorphous transition. From the comparison of peak shifts in the luminescence spectra, unique properties were revealed in the mechanochromic luminescent behaviors of the triads. The hypsochromic and bathochromic shifts of the emission bands were controllable by selecting the functional groups at the end of the triads. By introducing relatively small end groups such as hydrogen, the AIE colors were red in the initial crystalline state. Hypsochromic changes occurred after grinding. In contrast, the complexes with larger end groups such as iodo and trimethylsilyl groups gave a yellow AIE before grinding. After adding the same treatment, bathochromic shifts in the emission spectra were induced. From the X-ray single-crystal analysis with the initial crystalline samples, it was suggested that the triads with small substituents formed relatively densely packed structures because of small steric hindrances; meanwhile, weak π–π intermolecular interactions relative to those in the crystal state should be induced in the amorphous state. As a result, the emission band could be shifted to shorter wavelengths after the transition. In contrast, less-dense packing structures were obtained by the large substituents in the crystalline state. With external mechanical stress, the transition to a random configuration would occur, followed by the formation of stronger π–π intermolecular interactions in the amorphous state. Finally, bathochromic shifts of emission bands were induced. Because of the environment-sensitive electronic properties and solid-state luminescent properties, precise control was available.
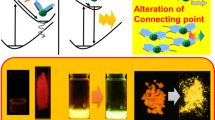
It was demonstrated that a slight alteration of the molecular distribution in the crystalline packing manifested as drastic luminescent color changes with group 13 element-blocks. Boron diiminates showed not only AIE but also crystallization-induced emission properties.94 Corresponding to the transitions between crystalline and amorphous states, the emission intensity reversibly changed. In addition, by introducing chemical modifications, the luminescent color was altered. These properties were maintained in the conjugated polymers. The methyl sulfide group was introduced into the copolymer with boron diiminate and the fluorene comonomer, and it was observed that the emission intensity of the polymer film was enhanced by oxidation at the sulfide group (Figure 13a).95 The electron-donating property of the methyl sulfide group was conversely shifted to electron accepting upon oxidation. Thereby, the electronic structure of conjugated polymers should be crucially perturbed, followed by an emission enhancement from the polymer film. As another example, the dimethylamine group was introduced into boron diiminate in the conjugated polymer (Figure 13b).96 By exposing the polymer film to acidic vapor, the emission color turned from red to yellow. Moreover, the pristine luminescent color was recovered by a treatment with amine vapor. These data suggest the applicability of AIE-active element-block polymers as film-type sensors.
Far more precise recognition for volatile organic compounds (VOCs) was accomplished with another group 13 element-block. It was discovered that replacing boron with gallium enhanced the solid-state emissive property with the AIE-active diiminate complex (Figure 14).97 It was proposed that the larger size of gallium could contribute to the suppression of ACQ efficiently, even in the crystalline state. Then, a larger emission intensity was obtained from the crystalline sample of the gallium complex. Furthermore, it was observed that gallium could create channels in the crystal packing. As a result, room for encapsulating VOCs was produced. The optical properties of gallium diiminate were monitored by fuming 17 types of VOCs. Interestingly, it was indicated that eight types of VOCs were able to turn the luminescent color of the gallium diiminate crystal from blue to green. In particular, it was found that the degree of luminescent chromism was dependent not on the chemical components of the captured VOCs but on their radius of gyration. Finally, it was also demonstrated that precise discrimination of the molecular size of VOCs by 0.1 Å was accomplished by the vapochromic luminescent property of gallium diiminate. From the structural analysis before and after fuming VOCs, it was revealed that a crystal-crystal structural transition was induced, followed by luminescent chromism. This result indicates that molecular recognition with high accuracy was accomplished by the gallium element-block. Because of the solid-state emission with the environmental sensitivity of gallium diiminate, VOC recognition was detected as luminescent chromism.
(a) Chemical structures of gallium diiminate and VOCs used in this study. (b) Quantum yields and degree of peak shifts of the emission bands in the crystalline state before and after fuming. (c) Correlations of the numbers of encapsulated molecules after fuming and the degree of peak shifts in the photoluminescence spectra to the radius of gyration of VOCs. Reproduced from Ref. 97 with permission from the Royal Society of Chemistry.
Except for this example, a variety of properties of gallium element-blocks and polymers have been explored in the recent research.98, 99, 100, 101, 102, 103, 104 We have reported on the synthesis of air- and moisture-stable gallafluorenes by employing a protection group to form the tetracoordinate gallium atom (Figure 15).98 Because of the high durability, a series of optical properties can be monitored. As a result, a bright phosphorescence was observed from the solution containing gallafluorene. In addition, a series of stable conjugated polymers with tetracoordinate gallium atoms was also prepared, and emission color tuning was accomplished.99 AIE-active conjugated polymers with gallium and boron were also obtained.105 From the comparison study between the electronic roles of boron and gallium in the AIE-active conjugated polymers, it was revealed that gallium was responsible for narrowing the energy band gaps of the polymers. From the mechanical studies, including theoretical approaches, it was proposed that the stabilization of the energy levels of molecular orbitals should be induced by gallium. These data suggest that gallium element-blocks are a potential platform for obtaining narrow-band gap polymers.
The final topic on group 13 element-blocks is on the logical design of AIE-active molecules with theoretical calculations based on ‘flexible’ boron complexes.106 From the first report on the AIE-active silole derivative in 2001, significant efforts have been dedicated to developing functional luminescent materials based on AIE-active molecules. Moreover, new molecules possessing AIE and/or other types of enhanced emission properties in the solid state have been continuously discovered. However, it is still challenging to theoretically predict an AIE-active molecular skeleton. As we mention above, we have investigated the photochemistry of AIE-active boron complexes. From these data, we developed a hypothesis for designing an AIE-active boron complex from scratch. In this part, the fundamental concept and process of developing a novel AIE-active boron complex is explained.
We demonstrated the transformation of the ACQ-presenting boron complex to the AIE-active dye by replacing one of the coordinated atoms in the ligand moiety.86 To understand the origin of this drastic change in the optical properties, we performed theoretical calculations to estimate the optimized structures.106 Significantly, by comparing the structure in the ground state obtained from density-functional theory with the result from the density-functional theory organic comp calculation, which can provide the structural information on the excited state, it was suggested that a larger degree of structural relaxation should proceed in the AIE-active boron complex. It is reasonable that excitation energy should be effectively dissipated by vigorous molecular motions in the solution state, followed by emission annihilation. On the other hand, the molecular motions would be highly restricted in the condensed state. Thereby, emission could be recovered. As a result, the typical AIE behavior could be observed. From these facts, we presumed that ‘flexible’ boron complexes, which show large structural relaxation, could be a potential AIE-active molecule (Figure 16). In particular, these structural relaxations should be predictable by theoretical calculations estimating the optimized structures of both ground and excited states. To prove the validity of this hypothesis, we designed the boron complexes and evaluated their optical properties.
Plausible emission mechanisms of BPI and FBPI in the solution state. Reproduced from Ref. 107 with permission from the Royal Society of Chemistry.
The boron complex BPI was designed and synthesized (Figure 17).107 Comparing the optimized structures in the ground and excited states via the same protocol, it was proposed that a large structural relaxation should be allowed. To fix the molecular structure, the fused boron complex FBPI was also prepared. Initially, UV-vis absorption and photoluminescence spectra were measured in tetrahydrofuran. It was clear that BPI showed little emission, meanwhile FBPI was a luminescent molecule. The larger width of the Stokes shift obtained from BPI meant that structural relaxation could occur in the excited state. In the frozen state at 77 K, a significant emission was observed even in BPI. This fact suggests that excitation deactivation should be induced by molecular motions. Next, the solid-state luminescent properties were evaluated with crystalline powders of both complexes. A significant strong emission was observed only from BPI. In contrast, FBPI showed a critical ACQ. From the emission efficiencies, it was shown that BPI should be an AIE- and crystallization-induced emission-active molecule. From the X-ray single-crystal analysis, it was proposed that both phenyl groups could play a role in the suppression of ACQ in the crystalline state. Recently, another molecule was designed, and the validity of the strategy based on a ‘flexible’ boron complex was proved.108 From these data, we conclude that our strategy based on ‘flexible’ boron complexes should be valid for designing brilliant new AIE-active molecules. We are continuously exploring another example with this idea.
(a) Photographs of BPI and FBPI under UV irradiation with variable water contents in acetonitrile (1.0 × 10−4 M). (b) Changes in emission-intensity ratios of BPI and FBPI with increasing water content in the samples. Reproduced from Ref. 107 with permission from the Royal Society of Chemistry.
P-stereogenic cyclic phosphines for chiral element-blocks
When two mirror-image molecules are non-superimposable, like right and left hands, we call those molecules chiral. Chirality exists in various molecules, including amino acids and sugars, which make up the natural world, and this property is essential to understanding the intrinsic properties of substances, as well as being effective in creating functional materials. In this section, we reveal that functional units based on chiral molecules are called ‘chiral element-blocks’ and combinations of the element-blocks served as the origin for versatile functional materials. As an example of the chiral element-blocks, chiral phosphorus atoms were selected. Chirality based on the trivalent phosphorus atom is used as a stable chiral center because the inversion energy of the unshared electron pair is high, and the inversion is restricted at room temperature.109 Moreover, phosphorus atoms have a high affinity toward transition metals, and their substituents affect coordination ability.110 Therefore, their complexes are widely used as chiral catalysts.111, 112, 113 There are several optically active polymers whose structures are highly controlled by repeating asymmetric reactions; however, optically active polymers including chiral heteroatoms in the main chain are limited. In our group, we have successfully reported oligomers,114, 115, 116, 117 polymers,118, 119, 120, 121, 122, 123 dendrimers124 and macrocycles,125, 126, 127, 128, 129, 130, 131, 132, 133 including chiral phosphorus atoms in the main chain, and we have revealed their properties and potential applications.
Recently, we have focused on diphosphacrowns as a kind of P-stereogenic cyclic phosphine and synthesized various types of optically active diphosphacrowns with P-stereogenic bisphosphine-borane complexes. The phosphacrown is a class of crown ethers134 in which some oxygen atoms have been replaced with phosphorus atoms. The precursor of the diphosphacrown (S,S)-3-BH3 was synthesized from (S,S)-1-BH3, as shown in Scheme 1.128 (S,S)-1-BH3 was first developed by the groups of Evans135 and Imamoto,136 and it was modified by our group to afford (S,S)-1-BH3 with >99%ee (enantiomeric excess).125 The methyl group of (S,S)-1-BH3 was lithiated by alkyllithium reagents such as sec-BuLi, and this enabled us to prepare various P-stereogenic phosphines having different functional groups. (S,S)-3-BH3 was readily converted into the corresponding P-stereogenic diphosphacrown, (S,S)-4-BH3. The enantiomer (R,R)-4-BH3 was also synthesized from the corresponding P-stereogenic dialcohol (R,R)-3-BH3. However, the original synthesis was challenging because the precursor (R,R)-1-BH3 had to be prepared with (+)-sparteine.137 The problem was solved by the development of another reaction route. Scheme 2 shows the new synthetic route to obtain both (S,S)-3-BH3 and (R,R)-3-BH3 from 5-BH3 with the only (−)-sparteine. The key point to obtaining (R,R)-3-BH3 was that one of the methyl groups of 5-BH3 was enantioselectively masked with (−)-sparteine, which enabled the subsequent oxidation coupling reaction of the other methyl groups.
As the phosphorus atom has a high affinity to various transition metals, we tried preparing complexes of P-stereogenic diphosphacrowns (S,S)-4-BH3 and palladium(II) with PdCl2 (cod) (cod=1,5-cyclooctadiene) as a precursor. Scheme 3 shows the synthesis of (S,S)-4 and the corresponding diphosphacrown-palladium complex 7a with PdCl2(cod) in CHCl3. The coordinated boranes were readily removed by treatment with a strong organic base such as 1,4-diazabicyclo[2.2.2]octane. The obtained diphosphacrowns were used for the subsequent complexation without further purification. To investigate the catalytic activity of 7a, we selected asymmetric 1,4-additions.138 This reaction with arylboronic acids to 2-cyclopentenone was investigated with several palladium/P-stereogenic bisphosphine/AgSbF6 catalytic systems.128 The use of 7a markedly increased the catalytic activity to afford the corresponding (R)-3-phenylcyclopentanone with an expected absolute configuration and 90% isolated yield, 85%ee. When the larger ring-size diphosphacrown-palladium complex 7b was used as the catalyst, the reactivity was similar, whereas the selectivity decreased (81% isolated yield, 30%ee). It was found that ring size is significant to the selectivity. The application of these materials to transition metal-catalyzed asymmetric reactions are ongoing.
The P-stereogenic diphosphacrowns present various intriguing functions; however, the poor isolated yields (~20%) of the enantiopure diphosphacrowns would restrict the widespread application of these compounds. We developed a new synthetic route that afforded enantiopure diphosphacrowns in substantially better isolated yields than the previous method.129 Scheme 4 describes the synthetic route to the target (R,R)-benzodiphosphacrowns (R,R)-11-BH3 from the P-stereogenic bisphosphine-borane complex (R)-12-BH3. The key point for improving the yield was to use (S,S)-10-BH3 as a precursor. As a result, the efficiency of the cyclization reaction was successfully increased. (S,S)-10-BH3 was synthesized from (R,R)-13-BH3 by a Ru-catalysed oxidation with K2S2O8/KOH and decarboxylation in an 82% isolated yield. The deprotonation of (S,S)-10-BH3 by n-BuLi and the subsequent reaction with an electrophile afforded (R,R)-benzodiphosphacrowns (R,R)-11-BH3 in a 62% isolated yield. The cyclization efficiency was high, and we successfully developed effective tools for approaching various derivatives of cyclic compounds based on chiral P-stereogenic phosphorus atoms. The optically active phosphacrowns that were recently reported by our group are listed in Figure 18. For example, P-stereogenic tetraphosphacrowns, containing four chiral phosphorus atoms in the main ring scaffold, was synthesized by using this new synthetic approach.130
Next, we were interested in the affinity of diphosphacrowns towards alkali metal ions in crown ethers. The crown ether (dibenzo-18-crown-6) was reported by Pedersen in 1967, and it represents a host molecule that can capture cationic guests such as alkali metal ions or ammonium ions.134 The guest selectivity of crown ethers can be controlled by the ring size, skeleton and donor atoms. To investigate the guest selectivity of diphosphacrowns, we newly prepared enantiopure P-stereogenic 12-, 15-, 18- and 21-membered aliphatic diphosphacrowns.132 The deboronation of (R,R)-11-BH3 was carried out, and the resulting (R,R)-11 was readily used for complexation (Scheme 5). The deboronated products of the aliphatic diphosphacrowns (R,R)-17–20 were obtained from the same procedure as that of (R,R)-17–20-BH3. Their complexation behavior with alkali metal ions, Li+, Na+, K+, Rb+ and Cs+, was examined and compared with benzo-18-diphosphacrown-6 (R,R)-11 and benzo-18-crown-6. The results are summarized in Table 1. The 15-, 18-, and 21-membered diphosphacrowns encapsulated alkali metal ions to form 1:1 metal complexes. Interestingly, diphosphacrowns captured smaller alkali metal ions than common crown ethers, for example, 18-membered diphosphacrowns interacted more strongly with Na+ than K+. The difference in guest selectivity between diphosphacrowns and common crown ethers was speculated to result from the cavity size because of the large phosphorus atom, as well as steric hindrance around the phosphine moiety. The guest-exchange behaviors of (R,R)-11 were monitored by using 31P NMR spectra, as shown in Figure 19. The addition of three equivalents of Na+ to the (R,R)-11-K+ complex solution led to complete guest exchange because of the large K value of the complexation with Na+ [logK(Na+)=3.99] compared with that of K+ [logK(K+)=2.37] (entry 1 in Table 1). The signal at −11.1 ppm disappeared, and a new signal was observed at −22.5 ppm. On the other hand, when four equivalents of free benzo-18-crown-6 moieties were added to the (R,R)-11-K+ complex solution, K+ was moved to benzo-18-crown-6, which had a larger K value [logK(K+)=3.96, entry 6 in Table 1] than (R,R)-11 [logK(K+)=2.37, entry 1 in Table 1]. Thus, the signal at −11.1 ppm was shifted to −0.4 ppm, which was the identical chemical shift of the free bisphosphine in (R,R)-11.
Scheme of guest exchanging of (R,R)-11 and 31P NMR spectra in each state. State A: free (R,R)-11. State B: addition of K+ to (R,R)-11=(R,R)-11-K+ complex. State C: addition of Na+ to (R,R)-11-K+ complex=(R,R)-11-Na+ complex. State D: addition of benzo-18-crown-6 to (R,R)-11-K+ complex=State A. All procedures were performed under an Ar atmosphere.
As clearly observed in the above examples, optically active P-stereogenic bisphosphine created various types of cyclic compounds, and they showed unique functionality. Recently, another functionality was preliminary observed in the macrocycles. For instance, it was found that pyridine-containing P-stereogenic diphosphacrown 26 (deboronated compound of 26-BH3) was capable of capturing guest molecules and especially discriminating (S)- and (R)-type chiral ammonium ions or d- and l-type chiral carboxylic acids.131 These results also prove that the diphosphacrown has the capacity to capture the guest molecules, and P-stereogenic bisphosphine has the potential to induce chirality into the macrocycles. Following these results, we constructed macrocycles including two chiral P-stereogenic bisphosphines and phenylene–ethynylene moieties for detecting chirality as circular dichroism and CPL (circularly polarized luminescence) signals.133 The chiroptical data obtained from circular dichroism and CPL spectra measurements were useful for understanding the structural information in the ground state and in the excited state, respectively. As a result, the macrocycles 27-BH3 and 28-BH3 showed circular dichroism properties, especially, 28-BH3 exhibited good CPL performance (glum=2.0 × 10−3, ΦPL=0.76, glum=ΔI/I (ΔI=Ileft−Iright), where Ileft and Iright indicate the luminescence intensities of left- and right-handed CPL, respectively), which indicated that chiral P-stereogenic bisphosphine induced chirality to the phenylene–ethynylene units both in the ground state and in the excited state.
The P-stereogenic bisphosphine has versatile applications including catalysis, the selective encapsulation of metal ions and guest molecules, and the induction of chirality into macrocyclic structures. Therefore, P-stereogenic bisphosphine is a representative element-blocks for creating optically active materials for applications derived from the intrinsic properties based on the elements.
Conclusions
We briefly introduced several recent studies on the development of element-block materials. Apart from the examples demonstrated here, numerous element-blocks and highly functional materials have been developed. In these studies, the fundamental characteristics of heteroatoms were more deeply investigated, and various useful properties were discovered. Moreover, it should be emphasized that advanced concepts such as ‘designable hybrids’ and ‘flexible’ boron complexes were conceived. Each idea is expected to not only be the driving force for breakthroughs in material research but also be the trigger to exploit new research fields. We believe that there is still considerable potential in the development of element-block materials.

Syntheses of bisphosphine (S,S)-3-BH3 and diphosphacrown (S,S)-4-BH3.

Syntheses of bisphosphines (S,S)-3-BH3 and (R,R)-3-BH3.

Syntheses of palladium complex 7a and asymmetric 1,4-addition of boronic acids to 2-cyclopentenone.

Synthetic routes of (a) (S,S)-11-BH3 and (b) (R,R)-11-BH3 with (−)-sparteine.

Deboranation of (R,R)-11-BH3. Ionic radius of alkali metal ions and chemical structure of host compounds.
References
Chujo, Y. & Tanaka, K. New polymeric materials based on element-blocks. Bull. Chem. Soc. Jpn 88, 633–643 (2015).
Guo, X., Baumgarten, M. & Müllen, K. Designing π-conjugated polymers for organic electronics. Prog. Polym. Sci. 38, 1832–1908 (2013).
De Gans, B.-J., Duineveld, P. C. & Schubert, U. S. Inkjet printing of polymers: State of the art and future developments. Adv. Mater. 16, 203–213 (2004).
Gon, M., Tanaka, K. & Chujo, Y. Creative synthesis of organic-inorganic molecular hybrid materials. Bull. Chem. Soc. Jpn 90, 463–474 (2017).
Kajiwara, Y., Nagai, A., Tanaka, K. & Chujo, Y. Efficient simultaneous emission from RGB-emitting organoboron dyes incorporated into organic-inorganic hybrids and preparation of white light-emitting materials. J. Mater. Chem. C 1, 4437–4444 (2013).
Okada, H., Tanaka, K. & Chujo, Y. Preparation of environmentally resistant conductive silica-based polymer hybrids containing tetrathiafulvalen-tetracyanoquinodimethane charge-transfer complexes. Polym. J. 46, 800–805 (2014).
Okada, H., Kajiwara, Y., Tanaka, K. & Chujo, Y. Rapid heat generation under microwave irradiation by imidazolium-presenting silica nanoparticles. Colloids Surf. A 428, 65–69 (2013).
Okada, H., Tanaka, K. & Chujo, Y. Microwave-driven enzyme deactivation using imidazolium salt-presenting silica nanoparticles. Bioorg. Med. Chem. Lett. 24, 4622–4625 (2014).
Okada, H., Tanaka, K., Ohashi, W. & Chujo, Y. Photo-triggered molecular release based on auto-degradable polymer-containing organic-inorganic hybrids. Bioorg. Med. Chem. 22, 3435–3440 (2014).
Okada, H., Tanaka, K. & Chujo, Y. Regulation of responsiveness of phosphorescence toward dissolved oxygen concentration by modulating polymer contents in organic-inorganic hybrid materials. Bioorg. Med. Chem. 22, 3141–3145 (2014).
Morisaki, Y., Tsuji, Y. & Chujo, Y. Versatile hybridization of conjugated polymers with silica. J. Mater. Chem. 21, 14402–14405 (2011).
Kajiwara, Y., Tanaka, K. & Chujo, Y. Enhancement of dye dispersibility in silica hybrids through local heating induced by the imidazolium group under microwave irradiation. Polym. J. 46, 195–199 (2014).
Cordes, D. B., Lickiss, P. D. & Rataboul, F. Recent developments in the chemistry of cubic polyhedral oligosilsesquioxanes. Chem. Rev. 110, 2081–2173 (2010).
Roll, M. F., Kampf, J. W., Kim, Y., Yi, E. & Laine, R. M. Nano building blocks via iodination of [PhSiO1.5]n, forming [p-I-C6H4SiO1.5]n n=8, 10, 12), and a new route to high-surface-area, thermally stable, microporous materials via thermal elimination of I2 . J. Am. Chem. Soc. 132, 10171–10183 (2010).
Tanaka, K. & Chujo, Y. Chemicals-inspired biomaterials; developing biomaterials inspired by material science based on POSS. Bull. Chem. Soc. Jpn 86, 1231–1239 (2013).
Tanaka, K. & Chujo, Y. Advanced functional materials based on polyhedral oligomeric silsesquioxane (POSS). J. Mater. Chem. 22, 1733–1746 (2012).
Pielichowski, K., Njuguna, J., Janowski, B. & Pielichowski, J. Polyhedral oligomeric silsesquioxanes (POSS)-containing nanohybrid polymers. Adv. Polym. Sci. 201, 225–296 (2006).
Lickiss, P. D. & Rataboul, F. Fully condensed polyhedral oligosilsesquioxanes (POSS): from synthesis to application. Adv. Organomet. Chem 57, 1–116 (2008).
Joshi, M. & Butola, B. S. Polymeric nanocomposites—polyhedral oligomeric silsesquioxanes (POSS) as hybrid nanofiller. J. Macromol. Sci., Polym. Rev. C44, 389–410 (2004).
Zhang, W. & Müller, A. H. E. Architecture, self-assembly and properties of well-defined hybrid polymers based on polyhedral oligomericsilsequioxane (POSS). Prog. Polym. Sci. 38, 1121–1162 (2013).
Ghanbari, H., Cousins, B. G. & Seifalian, A. M. A nanocage for nanomedicine: polyhedral oligomeric silsesquioxane (POSS). Macromol. Rapid Commun. 32, 1032–1046 (2011).
Tanaka, K., Adachi, S. & Chujo, Y. Structure-property relationship of octa-substituted POSS in thermal and mechanical reinforcements of conventional polymers. J. Polym. Sci. Part A: Polym. Chem. 47, 5690–5697 (2009).
Tanaka, K., Kozuka, H., Ueda, K., Jeon, J.-H. & Chujo, Y. POSS-based molecular fillers for simultaneously enhancing thermal and viscoelasticity of poly(methyl methacrylate) films. Mater. Lett. 203, 62–67 (2017).
Tanaka, K., Adachi, S. & Chujo, Y. Side-chain effect of octa-substituted POSS fillers on refraction in polymer composites. J. Polym. Sci. Part A: Polym. Chem. 48, 5712–5717 (2010).
Jeon, J.-H., Tanaka, K. & Chujo, Y. Rational design of POSS fillers for simultaneous improvements of thermomechanical properties and lowering refractive indices of polymer films. J. Polym. Sci. Part A: Polym. Chem. 51, 3583–3589 (2013).
Tanaka, K., Yamane, H., Mitamura, K., Watase, S., Matsukawa, K. & Chujo, Y. Transformation of sulfur to organic-inorganic hybrids employed by POSS networks and their application for the modulation of refractive indices. J. Polym. Sci. Part A: Polym. Chem. 52, 2588–2595 (2014).
Suenaga, K., Tanaka, K. & Chujo, Y. Heat-resistant mechanoluminescent chromism of the hybrid molecule based on boron ketoiminate-modified octa-substituted polyhedral oligomeric silsesquioxane. Chem. Eur. J. 23, 1409–1414 (2017).
Gon, M., Sato, K., Tanaka, K. & Chujo, Y. Controllable intramolecular interaction of 3D arranged π-conjugated luminophores based on a POSS scaffold, leading to highly thermostable and emissive materials. RSC Adv. 6, 78652–78660 (2016).
Ueda, K., Tanaka, K. & Chujo, Y. Remarkably high miscibility of octa-substituted POSS with commodity conjugated polymers and molecular fillers for the improvement of homogeneities of polymer matrices. Polym. J. 48, 1133–1139 (2016).
Ueda, K., Tanaka, K. & Chujo, Y. Synthesis of POSS derivatives having dual types of alkyl substituents via in situ sol-gel reactions and their application as a molecular filler for low-refractive and highly-durable materials. Bull. Chem. Soc. Jpn 90, 205–209 (2017).
Watanabe, M., Thomas, M. L., Zhang, S., Ueno, K., Yasuda, T. & Dokko, K. Application of ionic liquids to energy storage and conversion materials and devices. Chem. Rev. 10, 7190–7239 (2017).
Tanaka, K., Ishiguro, F. & Chujo, Y. POSS ionic liquid. J. Am. Chem. Soc. 132, 17649–17651 (2010).
Tanaka, K., Ishiguro, F. & Chujo, Y. Thermodynamic study of POSS-based ionic liquids with various numbers of ion pairs. Polym. J. 43, 708–713 (2011).
Tanaka, K., Ishiguro, F., Jeon, J.-H., Hiraoka, T. & Chujo, Y. POSS ionic liquid crystals. NPG Asia Mater. 7, e174 (2015).
Jeon, J.-H., Tanaka, K. & Chujo, Y. POSS fillers for modulating thermal properties of ionic liquids. RSC Adv. 3, 2422–2427 (2013).
Jeon, J.-H., Tanaka, K. & Chujo, Y. Synthesis of sulfonic acid-containing POSS and its filler effects for enhancing thermal stabilities and lowering melting temperatures of ionic liquids. J. Mater. Chem. A 2, 624–630 (2014).
Kota, A. K., Kwon, G., Choi, W., Mabry, J. M. & Tuteja, A. Hygro-responsive membranes for effective oil-water separation. Nat. Commun. 3, 1025 (2012).
Nishizawa, N., Fuji, S., Kakuta, T., Tanaka, K., Yusa, S., Chujo, Y. & Nakamura, Y. Polystyrene-polyhedral oligomeric silsesquioxane core-shell element-block polymer particles fabricated via heterocoagulation method. Chem. Lett. 45, 1168–1170 (2016).
Tanaka, K., Okada, H., Jeon, J.-H., Inafuku, K., Ohashi, W. & Chujo, Y. Hypoxic conditions-selective upconversion via triplet-triplet annihilation based on POSS-core dendrimer complexes. Bioorg. Med. Chem. 21, 2678–2681 (2013).
Tanaka, K., Jeon, J.-H., Inafuku, K. & Chujo, Y. Enhancements of optical properties of dyes for bioprobes by freezing effect of molecular motion using POSS-core dendrimers. Bioorg. Med. Chem. 20, 915–919 (2012).
Tanaka, K., Murakami, M., Jeon, J.-H. & Chujo, Y. Enhancement of affinity in molecular recognition via hydrogen bonds by POSS-core dendrimer and its application for selective complex formation between guanosine triphosphate and 1,8-naphthyridine derivatives. Org. Biomol. Chem. 10, 90–95 (2012).
Tanaka, K., Inafuku, K. & Chujo, Y. Environment-responsive upconversion based on dendrimer-supported efficient triplet-triplet annihilation in aqueous media. Chem. Commun. 46, 4378–4380 (2010).
Tanaka, K., Inafuku, K., Naka, K. & Chujo, Y. Enhancement of entrapping ability of dendrimers by a cubic silsesquioxane core. Org. Biomol. Chem. 6, 3899–3901 (2008).
Kakuta, T., Jeon, J.-H., Narikiyo, H., Tanaka, K. & Chujo, Y. Development of highly-sensitive detection system in 19F NMR for bioactive compounds based on the assembly of paramagnetic complexes with fluorinated cubic silsesquioxanes. Bioorg. Med. Chem. 25, 1389–1393 (2017).
Tirotta, I., Dichiarante, V., Pigliacelli, C., Cavallo, G., Terraneo, G., Bombelli, F. B., Metrangolo, P. & Resnati, G. (19)F magnetic resonance imaging (MRI): from design of materials to clinical applications. Chem. Rev. 115, 1106–1129 (2015).
Kitamura, N., Hiraoka, T., Tanaka, K. & Chujo, Y. Reduced glutathione-resisting 19F NMR sensors for detecting HNO. Bioorg. Med. Chem. 20, 4668–4674 (2012).
Tanaka, K., Kitamura, N. & Chujo, Y. Heavy metal-free 19F NMR probes for quantitative measurements of glutathione reductase activity using silica nanoparticles as a signal quencher. Bioorg. Med. Chem. 20, 96–100 (2012).
Tanaka, K., Kitamura, N. & Chujo, Y. Bi-modal quantitative monitoring for enzymatic activity with simultaneous signal increases in 19F NMR and fluorescence using silica nanoparticle-based molecular probes. Bioconjugate Chem. 22, 1484–1490 (2011).
Tanaka, K., Kitamura, N., Takahashi, Y. & Chujo, Y. Reversible signal regulation system of 19F NMR by redox reactions using a metal complex as a switching module. Bioorg. Med. Chem. 17, 3818–3823 (2009).
Tanaka, K., Inafuku, K. & Chujo, Y. Ratiometric multimodal chemosensors based on cubic silsesquioxanes for monitoring solvent polarity. Bioorg. Med. Chem. 16, 10029–10033 (2008).
Tanaka, K., Kitamura, N., Naka, K. & Chujo, Y. Multi-modal 19F NMR probe using perfluorinated cubic silsesquioxane-coated silica nanoparticles for monitoring enzymatic activity. Chem. Commun. 6176–6178 (2008).
Nakamura, M., Sanji, T. & Tanaka, M. Fluorometric sensing of biogenic amines with aggregation-induced emission-active tetraphenylethenes. Chem. Eur. J 17, 5344–5349 (2011).
Carvalho, M., Carmo, H., Costa, V. M., Capela, J. P., Pontes, H., Remião, F., Carvalho, F. & Bastos, M. d. L. Toxicity of amphetamines: an update. Arch. Toxicol. 86, 1167–1231 (2012).
Jeon, J.-H., Kakuta, T., Tanaka, K. & Chujo, Y. Facile design of organic-inorganic hybrid gels for molecular recognition of nucleoside triphosphates. Bioorg. Med. Chem. Lett. 25, 2050–2055 (2015).
Jeon, J.-H., Tanaka, K. & Chujo, Y. Light-driven artificial enzymes for selective oxidation of guanosine triphosphate using water-soluble POSS network polymers. Org. Biomol. Chem. 12, 6500–6506 (2014).
Tanaka, K., Ohashi, W., Kitamura, N. & Chujo, Y. Reductive glutathione-responsive molecular release using water-soluble POSS network polymers. Bull. Chem. Soc. Jpn 84, 612–616 (2011).
Tanaka, K., Inafuku, K., Adachi, S. & Chujo, Y. Tuning of properties of POSS-condensed water-soluble network polymers by modulating the crosslinking ratio between POSS. Macromolecules 42, 3489–3492 (2009).
Kakuta, T., Tanaka, K. & Chujo, Y. Synthesis of emissive water-soluble network polymers based on polyhedral oligomeric silsesquioxane and their application as an optical sensor for discriminating the particle size. J. Mater. Chem. C 3, 12539–12545 (2015).
Li, C., Zhang, Y., Li, S., Wang, G., Xu, C., Deng, Y. & Wang, S. Mechanism of formation of trans fatty acids under heating conditions in triolein. J. Agric. Food Chem. 61, 10392–10397 (2013).
Gebauer, S. K., Chardigny, J.-M., Jakobsen, M. U., Lamarche, B., Lock, A. L., Proctor, S. D. & Baer, D. J. Effects of ruminant trans fatty acids on cardiovascular disease and cancer: A comprehensive review of epidemiological, clinical, and mechanistic studies. Adv. Nutr. 2, 332–354 (2011).
Narikiyo, H., Kakuta, T., Matsuyama, H., Gon, M., Tanaka, K. & Chujo, Y. Development of optical sensor for discriminating isomers of fatty acids based on emissive network polymers composed of polyhedral oligomeric silsesquioxane. Bioorg. Med. Chem. 25, 3431–3436 (2017).
Núñez, R., Tarrés, M., Ferrer-Ugalde, A., de Biani, F. F. & Teixidor, F. Electrochemistry and photoluminescence of icosahedral carboranes, boranes, metallacarboranes, and their derivatives. Chem. Rev. 116, 14307–14378 (2016).
Kokado, K. & Chujo, Y. Multicolor tuning of aggregation-induced emission through substituent variation of diphenyl-o-carborane. J. Org. Chem. 76, 316–319 (2010).
Kokado, K. & Chujo, Y. Emission via aggregation of alternating polymers with o-carborane and p-phenylene-ethynylene sequences. Macromolecules 42, 1418–1420 (2009).
Mei, J., Leung, N. L. C., Kwok, R. T. K., Lam, J. W. Y. & Tang, B. Z. Aggregation-induced emission: together we shine, united we soar!. Chem. Rev. 115, 11718–11940 (2015).
Nishino, K., Hashimoto, K., Tanaka, K., Morisaki, Y. & Chujo, Y. Synthesis and properties of highly-rigid conjugation system based on bi(benzo[b]thiophene)-fused o-carborane. Tetrahedron Lett. 57, 2025–2028 (2016).
Weber, L., Kahlert, J., Brockhinke, R., Böhling, L., Brockhinke, A., Stammler, H.-G., Neumann, B., Harder, R. A. & Fox, M. A. Luminescence properties of o-diazaborolyl-ortho-carboranes as donor-acceptor systems. Chem. Eur. J. 18, 8347–8357 (2012).
Wu, J., Liu, W., Ge, J., Zhang, H. & Wang, P. New sensing mechanisms for design of fluorescent chemosensors emerging in recent years. Chem. Soc. Rev. 40, 3483–3495 (2011).
Naito, H., Nishino, K., Morisaki, Y., Tanaka, K. & Chujo, Y. Solid-state emission of the anthracene-o-carborane dyad via twisted-intramolecular charge transfer in the crystalline state. Angew. Chem. Int. Ed. 56, 254–259 (2017).
Nishino, K., Yamamoto, H., Tanaka, K. & Chujo, Y. Development of solid-state emissive materials based on multi-functional o-carborane-pyrene dyads. Org. Lett. 18, 4064–4067 (2016).
Naito, H., Nishino, K., Morisaki, Y., Tanaka, K. & Chujo, Y. Luminescence color tuning of stable luminescent solid materials from blue to NIR based on bis-o-carborane-substituted oligoacenes. Chem. Asian J. 12 (2017).
Naito, H., Morisaki, Y. & Chujo, Y. o-Carborane-based anthracene: a variety of emission behaviors. Angew. Chem. Int. Ed. 54, 5084–5087 (2015).
Tanaka, K. & Chujo, Y. Recent progress of optical functional nanomaterials based on organoboron complexes with β-diketonate, ketoiminate and diiminate. NPG Asia Mater. 7, e223 (2015).
Tanaka, K. & Chujo, Y. Advanced luminescent materials based on organoboron polymers. Macromol. Rapid Commun. 33, 1235–1255 (2012).
Yeo, H., Tanaka, K. & Chujo, Y. Tunable optical property between pure red luminescence and dual-emission depended on the length of light-harvesting antennae in the dyads containing the cardo structure of BODIPY and oligofluorene. Macromolecules 49, 8899–8904 (2016).
Yamane, H., Ito, S., Tanaka, K. & Chujo, Y. Preservation of main-chain conjugation through BODIPY-containing alternating polymers from electronic interactions with side-chain substituents by cardo boron structures. Polym. Chem. 7, 2799–2807 (2016).
Tanaka, K., Yanagida, T., Yamane, H., Hirose, A., Yoshii, R. & Chujo, Y. Liquid scintillators with near infrared emission based on organoboron conjugated polymers. Bioorg. Med. Chem. Lett. 25, 5331–5334 (2015).
Yoshii, R., Yamane, H., Tanaka, K. & Chujo, Y. Synthetic strategy for low-band gap oligomers and homopolymers using characteristics of thiophene-fused boron dipyrromethene. Macromolecules 47, 3755–3760 (2014).
Yoshii, R., Yamane, H., Nagai, A., Tanaka, K., Taka, H., Kita, H. & Chujo, Y. π-Conjugated polymers composed of BODIPY or aza-BODIPY derivatives exhibiting high electron mobility and low threshold voltage in electron-only devices. Macromolecules 47, 2316–2323 (2014).
Tanaka, K., Tamashima, K., Nagai, A., Okawa, T. & Chujo, Y. Facile modulation of optical properties of diketonate-containing polymers by regulating complexation ratios with boron. Macromolecules 46, 2969–2975 (2013).
Yeo, H., Tanaka, K. & Chujo, Y. Effective light-harvesting antennae based on BODIPY-tethered cardo polyfluorenes via rapid energy transferring and low concentration quenching. Macromolecules 46, 2599–2605 (2013).
Yoshii, R., Nagai, A., Tanaka, K. & Chujo, Y. Highly NIR emissive boron di(iso)indomethene (BODIN)-based polymer: drastic change from deep-red to NIR emission via quantitative polymer reaction. J. Polym. Sci. Part A: Polym. Chem. 51, 1726–1733 (2013).
Yeo, H., Tanaka, K. & Chujo, Y. Synthesis and energy transfer through heterogeneous dyes-substituted fluorene-containing alternating copolymers and their dual-emission properties. J. Polym. Sci. A: Polym. Chem. 53, 2026–2035 (2015).
Yeo, H., Tanaka, K. & Chujo, Y. Synthesis of dual-emissive polymers based on ineffective energy transfer through cardo fluorene-containing conjugated polymers. Polymer 60, 228–233 (2015).
Luo, J., Xie, Z., Lam, Y., W. J., Cheng, L., Chen, H., Qiu, C., Kwok, H. S., Zhan, X., Liu, Y., Zhu, D. & Tang, B. Z. Aggregation-induced emission of 1-methyl-1,2,3,4,5-pentaphenylsilole. Chem. Commun. 1740–1741 (2001).
Yoshii, R., Nagai, A., Tanaka, K. & Chujo, Y. Highly emissive boron ketoiminate derivatives as new class of aggregation-induced emission fluorophores. Chem. Eur. J. 19, 4506–4512 (2013).
Yoshii, R., Tanaka, K. & Chujo, Y. Conjugated polymers based on tautomeric units: regulation of main-chain conjugation and expression of aggregation induced emission property via boron-complexation. Macromolecules 47, 2268–2278 (2014).
Suenaga, K., Tanaka, K. & Chujo, Y. Design and luminescent chromism of fused boron complexes having constant emission efficiencies in solution, amorphous and crystalline states. Eur. J. Org. Chem. 2017, doi:10.1002/ejoc.201700704 (2017).
Yamaguchi, M., Ito, S., Hirose, A., Tanaka, K. & Chujo, Y. Modulation of sensitivity to mechanical stimulus in mechanofluorochromic properties by altering substituent positions in solid-state emissive diiodo boron diiminates. J. Mater. Chem. C 3, 5314–5319 (2016).
Suenaga, K., Yoshii, R., Tanaka, K. & Chujo, Y. Sponge-type emissive chemosensors for the protein detection based on boron ketoiminate-modifying hydrogels with aggregation-induced blue shift emission property. Macromol. Chem. Phys. 217, 414–417 (2016).
Tanaka, K., Yanagida, T., Hirose, A., Yamane, H., Yoshii, R. & Chujo, Y. Synthesis and color tuning of boron diiminate conjugated polymers with aggregation-induced scintillation properties. RSC Adv. 5, 96653–96659 (2015).
Yoshii, R., Nagai, A., Tanaka, K. & Chujo, Y. Boron ketoiminate-based polymers: fine-tuning of the emission color and expression of strong emission both in the solution and film state. Macromol. Rapid Commun. 35, 1315–1319 (2014).
Yoshii, R., Suenaga, K., Tanaka, K. & Chujo, Y. Mechanofluorochromic materials based on aggregation-induced emission-active boron ketoiminates: regulation of the direction of the emission color changes. Chem. Eur. J. 21, 7231–7237 (2015).
Yoshii, R., Hirose, A., Tanaka, K. & Chujo, Y. Boron diiminate with aggregation-induced emission and crystallization-induced emission enhancement characteristics. Chem. Eur. J. 20, 8320–8324 (2014).
Hirose, A., Tanaka, K., Yoshii, R. & Chujo, Y. Film-type chemosensors based on boron diminate polymers having oxidation-induced emission properties. Polym. Chem. 6, 5590–5595 (2015).
Yoshii, R., Hirose, A., Tanaka, K. & Chujo, Y. Functionalization of boron diiminates with unique optical properties: multicolor tuning of crystallization-induced emission and introduction into the main-chain of conjugated polymers. J. Am. Chem. Soc. 136, 18131–18139 (2014).
Ito, S., Hirose, A., Yamaguchi, M., Tanaka, K. & Chujo, Y. Size-discrimination for volatile organic compounds utilizing gallium diiminate by luminescent chromism of crystallization-induced emission via encapsulation-triggered crystal-crystal transition. J. Mater. Chem. C 3, 5564–5571 (2016).
Matsumoto, T., Tanaka, K. & Chujo, Y. Synthesis and optical properties of stable gallafluorene derivatives: investigation of their emission via triplet states. J. Am. Chem. Soc. 135, 4211–4214 (2013).
Matsumoto, T., Tanaka, K. & Chujo, Y. Synthesis and characterization of gallafluorene-containing conjugated polymers: control of emission colors and electronic effects of gallafluorene units on π-conjugation system. Macromolecules 48, 1343–1351 (2015).
Matsumoto, T., Takamine, H., Tanaka, K. & Chujo, Y. Synthesis and characterization of heterofluorenes with five-coordinated group 13 elements. Chem. Lett. 44, 1658–1660 (2015).
Matsumoto, T., Tanaka, K. & Chujo, Y. High HOMO levels and narrow energy band gaps of dithienogalloles. RSC Adv. 5, 55406–55410 (2015).
Matsumoto, T., Tanaka, K., Tanaka, K. & Chujo, Y. Synthesis and characterization of heterofluorenes containing four-coordinated group 13 elements: theoretical and experimental analyses and comparison of structures, optical properties and electronic states. Dalton Trans. 44, 8697–8707 (2015).
Matsumoto, T., Takamine, H., Tanaka, K. & Chujo, Y. Synthesis of air- and moisture-stable dibenzogallepins: control of planarity of seven-membered rings in solid states by coordination to gallium atoms. Org. Lett. 17, 1593–1596 (2015).
Matsumoto, T., Onishi, Y., Tanaka, K., Fueno, H., Tanaka, K. & Chujo, Y. Synthesis of conjugated polymers containing gallium atoms and evaluation of conjugation though four-coordinate gallium atoms. Chem. Commun. 50, 15740–15743 (2014).
Ito, S., Hirose, A., Yamaguchi, M., Tanaka, K. & Chujo, Y. Synthesis of aggregation-induced emission-active conjugated polymers composed of group 13 diiminate complexes with tunable energy levels via alteration of central element. Polymers 9, 68–78 (2017).
Tanaka, K., Nishino, K., Ito, S., Yamane, H., Suenaga, K., Hashimoto, K. & Chujo, Y. Development of the solid-state emissive o-carborane and theoretical investigation for mechanism of aggregation-induced emission behaviors of organoboron ‘element-blocks’. Faraday Discuss. 196, 31–42 (2017).
Yamaguchi, M., Ito, S., Hirose, A., Tanaka, K. & Chujo, Y. Control of aggregation-induced emission versus fluorescence aggregation-caused quenching by the bond existence at the single site in boron pyridinoiminate complexes. Mater. Chem. Front. 1 (2017).
Ohtani, S., Gon, M., Tanaka, K. & Chujo, Y. Flexible fused azomethine-boron complex: thermally-induced switching of crystalline-state luminescent property and thermosalient behaviors based on phase transition between polymorphs. Chem. Eur. J. 23 (2017).
Baechler, R. D. & Mislow, K. Effect of structure on the rate of pyramidal inversion of acyclic phosphines. J. Am. Chem. Soc. 92, 3090–3093 (1970).
Tolman, C. A. Steric effects of phosphorus ligands in organometallic chemistry and homogeneous catalysis. Chem. Rev. 77, 313–348 (1977).
Grabulosa, A. P-stereogenic ligands in enantioselective catalysis, (Royal Society of Chemistry, Cambridge, UK, 2011).
Crépy, K. V. L. & Imamoto, T. Recent developments in catalytic asymmetric hydrogenation employing P-chirogenic diphosphine ligands. Adv. Synth. Catal. 345, 79–101 (2003).
Imamoto, T. P-chiral phosphine ligands for transition-metal-catalyzed asymmetric reactions. J Synth. Org. Chem. Jpn 65, 1060–1069 (2007).
Morisaki, Y., Ouchi, Y., Fukui, T., Naka, K. & Chujo, Y. Synthesis of oligomers including eight P-chiral centers and the construction of the 12-phosphacrown-4 skeleton. Tetrahedron Lett. 46, 7011–7014 (2005).
Morisaki, Y., Ouchi, Y., Naka, K. & Chujo, Y. Synthesis of optically active oligomers consisting of chiral phosphorus atoms: capture of an intermediate between a polymer and a small molecule. Tetrahedron Lett. 48, 1451–1455 (2007).
Morisaki, Y., Ouchi, Y., Naka, K. & Chujo, Y. Synthesis of optically active P-chiral and optically inactive oligophosphines. Chem. Asian J. 2, 1166–1173 (2007).
Imoto, H., Kato, R., Morisaki, Y. & Chujo, Y. Synthesis of unsymmetrical P-stereogenic oligophosphines and chemoselective cleavage of phosphine-borane coordinate bonds. Polym J. 44, 579–585 (2012).
Morisaki, Y., Ouchi, Y., Tsurui, K. & Chujo, Y. Synthesis of optically active polymers containing chiral phosphorus atoms in the main chain. J. Polym. Sci. A Polym. Chem. 45, 866–872 (2007).
Morisaki, Y., Ouchi, Y., Tsurui, K. & Chujo, Y. Synthesis of the optically active polymer consisting of chiral phosphorus atoms and p-phenylene-ethynylene units. Polymer Bull. 58, 665–671 (2007).
Ouchi, Y., Morisaki, Y., Ogoshi, T. & Chujo, Y. Synthesis of a stimuli-responsive P-chiral polymer with chiral phosphorus atoms and azobenzene moieties in the main chain. Chem. Asian J. 2, 397–402 (2007).
Morisaki, Y., Saito, K., Imoto, H., Suzuki, K., Ouchi, Y. & Chujo, Y. Synthesis of optically active polymers using P-chiral bisphosphines as anionic initiators. Polym. Sci. Series A 51, 1218–1228 (2009).
Imoto, H., Morisaki, Y. & Chujo, Y. Synthesis and coordination behaviors of P-stereogenic polymers. Chem. Commun. 46, 7542–7544 (2010).
Morisaki, Y., Suzuki, K., Imoto, H. & Chujo, Y. P-stereogenic optically active polymer and the complexation behavior. Macromol. Chem. Phys. 212, 2603–2611 (2011).
Ouchi, Y., Morisaki, Y. & Chujo, Y. Synthesis of optically active dendrimers having chiral bisphosphine as a core. Polym. Bull. 59, 339–350 (2007).
Morisaki, Y., Imoto, H., Ouchi, Y., Nagata, Y. & Chujo, Y. Stereospecific construction of a trans-1,4-diphosphacyclohexane skeleton. Org. Lett. 10, 1489–1492 (2008).
Morisaki, Y., Imoto, H., Kato, R., Ouchi, Y. & Chujo, Y. Stereospecific synthesis of trans-1,4-diphosphacyclohexanes. Heterocycles 85, 2543–2550 (2012).
Morisaki, Y., Imoto, H., Tsurui, K. & Chujo, Y. Practical synthesis of P-stereogenic diphosphacrowns. Org. Lett. 11, 2241–2244 (2009).
Morisaki, Y., Imoto, H., Hirano, K., Hayashi, T. & Chujo, Y. Synthesis of enantiomerically pure P-stereogenic diphosphacrowns and their palladium complexes. J. Org. Chem. 76, 1795–1803 (2011).
Morisaki, Y., Kato, R. & Chujo, Y. Synthesis of enantiopure P-stereogenic diphosphacrowns using P-stereogenic secondary phosphines. J. Org. Chem. 78, 2769–2774 (2013).
Kato, R., Morisaki, Y. & Chujo, Y. Synthesis of P-stereogenic tetraphosphacrowns. Asian J. Org. Chem. 4, 1410–1416 (2015).
Kato, R., Watanabe, H., Morisaki, Y. & Chujo, Y. P-stereogenic diphosphacrowns: facile incorporation of aromatic rings. Heterocycles 91, 2295–2306 (2015).
Morisaki, Y., Kato, R. & Chujo, Y. Synthesis and alkali-metal-ion complexation of P-stereogenic diphosphacrowns. ChemistryOpen 5, 325–330 (2016).
Kato, R., Fukuyama, M., Morisaki, Y. & Chujo, Y. Synthesis of P-stereogenic macrocycles. Heteroatom. Chem. 28, e21354 (2017).
Pedersen, C. J. Cyclic polyethers and their complexes with metal salts. J. Am. Chem. Soc. 89, 7017–7036 (1967).
Muci, A. R., Campos, K. R. & Evans, D. A. Enantioselective deprotonation as a vehicle for the asymmetric synthesis of C2-symmetric P-chiral diphosphines. J. Am. Chem. Soc. 117, 9075–9076 (1995).
Imamoto, T., Watanabe, J., Wada, Y., Masuda, H., Yamada, H., Tsuruta, H., Matsukawa, S. & Yamaguchi, K. P-Chiral bis(trialkylphosphine) ligands and their use in highly enantioselective hydrogenation reactions. J. Am. Chem. Soc. 120, 1635–1636 (1998).
Smith, B. T., Wendt, J. A. & Aubé, J. First asymmetric total synthesis of (+)-sparteine. Org. Lett. 4, 2577–2579 (2002).
Christoffers, J., Koripelly, G., Rosiak, A. & Rössle, M. Recent advances in metal-catalyzed asymmetric conjugate additions. Synthesis 2007, 1279–1300 (2007).
Acknowledgements
This work was supported by a Grant-in-Aid for Scientific Research on Innovative Areas ‘New Polymeric Materials Based on Element-Blocks (No.2401)’ (JSPS KAKENHI Grant Number JP24102013).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Gon, M., Tanaka, K. & Chujo, Y. Recent progress in the development of advanced element-block materials. Polym J 50, 109–126 (2018). https://doi.org/10.1038/pj.2017.56
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pj.2017.56
This article is cited by
-
Development of fluorescence sensors for quantifying anions based on polyhedral oligomeric silsesquioxane that contains flexible side chains with urea structures
Polymer Journal (2024)
-
Tripod-shaped POSS compounds as single-component silsesquioxane hybrids
Polymer Journal (2024)
-
Creation of photofunctional materials through supramolecular complex formation and host–guest chemistry
Journal of Inclusion Phenomena and Macrocyclic Chemistry (2024)
-
Preparation and properties of PDMS elastomer cross-linked with hydrolyzate of tetraethoxysilane, hexaethoxydisiloxane, and octaethoxytrisiloxane: influence of cross-linker structure
Journal of Sol-Gel Science and Technology (2024)
-
Synthesis of main-chain-type triphenylarsine polymers
Polymer Journal (2023)