Abstract
Background:
In men undergoing definitive radiation for prostate cancer, it is unclear whether early biochemical response can provide additional prognostic value beyond pre-treatment risk stratification.
Methods:
Prostate cancer patients consecutively treated with definitive radiation at our institution by a single provider from 1993 to 2006 and who had an end-of-radiation (EOR) PSA (n=688, median follow-up 11.2 years). We analyzed the association of an EOR PSA level, obtained during the last week of radiation, with survival outcomes. Multivariable-adjusted cox proportional hazards models were constructed to assess associations between a detectable EOR PSA (defined as ⩾0.1 ng ml−1) and biochemical failure-free survival (BFFS), metastasis-free survival (MFS), prostate cancer-specific survival (PCSS) and overall survival (OS). Kaplan–Meier survival curves were constructed, with stratification by EOR PSA.
Results:
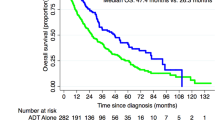
At the end of radiation, the PSA level was undetectable in 30% of patients. Men with a detectable EOR PSA experienced inferior 10-year BFFS (49.7% versus 64.4%, P<0.001), 10-year MFS (84.8% versus 92.0%, P=0.003), 10-year PCSS (94.3% versus 98.2%, P=0.007) and 10-year OS (75.8% versus 82.5%, P=0.01), as compared to men with an undetectable EOR PSA. Among National Comprehensive Care Network (NCCN) intermediate- and high-risk men who were treated with definitive radiation and androgen deprivation therapy (ADT), a detectable EOR PSA was more strongly associated with PCSS than initial NCCN risk level (EOR PSA: HR 5.89, 95% CI 2.37–14.65, P<0.001; NCCN risk level: HR 2.01, 95% CI 0.74–5.42, P=0.168). Main study limitations are retrospective study design and associated biases.
Conclusions:
EOR PSA was significantly associated with survival endpoints in men who received treatment with definitive radiation and ADT. Whether the EOR PSA can be used to modulate treatment intensity merits further investigation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
D’Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. J Am Med Assoc 1998; 280: 969–974.
Mohler JL, Armstrong AJ, Bahnson RR, D’Amico AV, Davis BJ, Eastham JA et al. Prostate cancer, version 1.2016. J Natl Compr Canc Netw 2016; 14: 19–30.
Thompson I, Thrasher JB, Aus G, Burnett AL, Canby-Hagino ED, Cookson MS et al. Guideline for the management of clinically localized prostate cancer: 2007 update. J Urol 2007; 177: 2106–2131.
Heidenreich A, Bellmunt J, Bolla M, Joniau S, Mason M, Matveev V et al. EAU guidelines on prostate cancer. Part 1: screening, diagnosis, and treatment of clinically localised disease. Eur Urol 2011; 59: 61–71.
Cooperberg MR, Pasta DJ, Elkin EP, Litwin MS, Latini DM, Du Chane J et al. The University of California, San Francisco Cancer of the Prostate Risk Assessment score: a straightforward and reliable preoperative predictor of disease recurrence after radical prostatectomy. J Urol 2005; 173: 1938–1942.
Loeb S, Schaeffer EM, Trock BJ, Epstein JI, Humphreys EB, Walsh PC . What are the outcomes of radical prostatectomy for high-risk prostate cancer? Urology 2010; 76: 710–714.
Zelefsky MJ, Lyass O, Fuks Z, Wolfe T, Burman C, Ling CC et al. Predictors of improved outcome for patients with localized prostate cancer treated with neoadjuvant androgen ablation therapy and three-dimensional conformal radiotherapy. J Clin Oncol 1998; 16: 3380–3385.
Sumi M, Ikeda H, Tokuuye K, Kagami Y, Murayama S, Tobisu K et al. The external radiotherapy with three-dimensional conformal boost after the neoadjuvant androgen suppression for patients with locally advanced prostatic carcinoma. Int J Radiat Oncol Biol Phys 2000; 48: 519–528.
Ludgate CM, Bishop DC, Pai H, Eldridge B, Lim J, Berthelet E et al. Neoadjuvant hormone therapy and external-beam radiation for localized high-risk prostate cancer: the importance of PSA nadir before radiation. Int J Radiat Oncol Biol Phys 2005; 62: 1309–1315.
Alexander AS, Mydin A, Jones SO, Christie J, Lim JT, Truong PT et al. Extreme-risk prostate adenocarcinoma presenting with prostate-specific antigen (PSA)> 40 ng/ml: prognostic significance of the preradiation PSA nadir. Int J Radiat Oncol Biol Phys 2011; 81: e713–e719.
Mitchell DM, McAleese J, Park RM, Stewart DP, Stranex S, Eakin RL et al. Failure to achieve a PSA level≤ 1 ng/ml after neoadjuvant LHRHa therapy predicts for lower biochemical control rate and overall survival in localized prostate cancer treated with radiotherapy. Int J Radiat Oncol Biol Phys 2007; 69: 1467–1471.
Foo M, Lavieri M, Pickles T . Impact of neoadjuvant prostate-specific antigen kinetics on biochemical failure and prostate cancer mortality: results from a prospective patient database. Int J Radiat Oncol Biol Phys 2013; 85: 385–392.
McGuire SE, Lee AK, Cerne JZ, Munsell MF, Levy LB, Kudchadker RJ et al. PSA response to neoadjuvant androgen deprivation therapy is a strong independent predictor of survival in high-risk prostate cancer in the dose-escalated radiation therapy era. Int J Radiat Oncol Biol Phys 2013; 85: e39–e46.
Zelefsky MJ, Gomez DR, Polkinghorn WR, Pei X, Kollmeier M . Biochemical response to androgen deprivation therapy before external beam radiation therapy predicts long-term prostate cancer survival outcomes. Int J Radiat Oncol Biol Phys 2013; 86: 529–533.
Zilli T, Jorcano S, Escude L, Linero D, Rouzaud M, Dubouloz A et al. Hypofractionated external beam radiotherapy to boost the prostate with ≥85 Gy/equivalent dose for patients with localised disease at high risk of lymph node involvement: feasibility, tolerance and outcome. Clin Oncol 2014; 26: 316–322.
Crook J, Ludgate C, Malone S, Perry G, Eapen L, Bowen J et al. Final report of multicenter Canadian phase III randomized trial of 3 versus 8 months of neoadjuvant androgen deprivation therapy before conventional-dose radiotherapy for clinically localized prostate cancer. Int J Radiat Oncol Biol Phys 2009; 73: 327–333.
Alexander A, Crook J, Jones S, Malone S, Bowen J, Truong P et al. Is biochemical response more important than duration of neoadjuvant hormone therapy before radiotherapy for clinically localized prostate cancer? An analysis of the 3-versus 8-month randomized trial. Int J Radiat Oncol Biol Phys 2010; 76: 23–30.
Heymann JJ, Benson MC, O’Toole KM, Malyszko B, Brody R, Vecchio D et al. Phase II study of neoadjuvant androgen deprivation followed by external-beam radiotherapy with 9 months of androgen deprivation for intermediate- to high-risk localized prostate cancer. J Clin Oncol 2007; 25: 77–84.
Roach M, Hanks G, Thames H, Schellhammer P, Shipley WU, Sokol GH et al. Defining biochemical failure following radiotherapy with or without hormonal therapy in men with clinically localized prostate cancer: recommendations of the RTOG-ASTRO Phoenix Consensus Conference. Int J Radiat Oncol Biol Phys 2006; 65: 965–974.
Hawkins DM . The problem of overfitting. J Chem Inf Comput Sci 2004; 44: 1–12.
Schwartz CL, Constine LS, Villaluna D, London WB, Hutchison RE, Sposto R et al. A risk-adapted, response-based approach using ABVE-PC for children and adolescents with intermediate- and high-risk Hodgkin lymphoma: the results of P9425. Blood 2009; 114: 2051–2059.
Ryan CJ, Smith A, Lal P, Satagopan J, Reuter V, Scardino P et al. Persistent prostate-specific antigen expression after neoadjuvant androgen depletion: an early predictor of relapse or incomplete androgen suppression. Urology 2006; 68: 834–839.
Spratt DE, Evans MJ, Davis BJ, Doran MG, Lee MX, Shah N et al. Androgen receptor upregulation mediates radioresistance after ionizing radiation. Cancer Res 2015; 75: 4688–4696.
D’Amico AV, Chen M, de Castro M, Loffredo M, Lamb DS, Steigler A et al. Surrogate endpoints for prostate cancer-specific mortality after radiotherapy and androgen suppression therapy in men with localised or locally advanced prostate cancer: an analysis of two randomised trials. Lancet Oncol 2012; 13: 189–195.
Cury FL, Hunt D, Roach M, Shipley W, Gore E, Hsu I et al. Prostate‐specific antigen response after short‐term hormone therapy plus external‐beam radiotherapy and outcome in patients treated on Radiation Therapy Oncology Group study 9413. Cancer 2013; 119: 1999–2004.
De Bono JS, Logothetis CJ, Molina A, Fizazi K, North S, Chu L et al. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med 2011; 364: 1995–2005.
Beer TM, Armstrong AJ, Rathkopf DE, Loriot Y, Sternberg CN, Higano CS et al. Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med 2014; 371: 424–433.
Narang AK, Gergis C, Robertson SP, He P, Ram AN, McNutt TR et al. Very high-risk localized prostate cancer: outcomes following definitive radiation. Int J Radiat Oncol Biol Phys 2016; 94: 254–262.
Morris WJ, Tyldesley S, Pai HH, Halperin R, McKenzie MR, Duncan G et al ASCENDE-RT*: a multicenter, randomized trial of dose-escalated external beam radiation therapy (EBRT-B) versus low-dose-rate brachytherapy (LDR-B) for men with unfavorable-risk localized prostate cancer. In: ASCO Annual Meeting Proceedings, 2015.
Kuban DA, Tucker SL, Dong L, Starkschall G, Huang EH, Cheung MR et al. Long-term results of the MD Anderson randomized dose-escalation trial for prostate cancer. Int J Radiat Oncol Biol Phys 2008; 70: 67–74.
Acknowledgements
PT Tran was supported by the Keeling Family; The Motta Family; The Irene and Bernard L. Schwartz Scholar Award from the Patrick C. Walsh Prostate Cancer Research Fund; A Movember-Prostate Cancer Foundation award; Grant numbers: DoD W81XWH-11-1-0272; Sidney Kimmel Foundation SKF-13-021; ACS 122688-RSG-12-196-01-TBG; NIH/NCI R01CA166348.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Prostate Cancer and Prostatic Diseases website .
Supplementary information
Rights and permissions
About this article
Cite this article
Narang, A., Trieu, J., Radwan, N. et al. End-of-radiation PSA as a novel prognostic factor in patients undergoing definitive radiation and androgen deprivation therapy for prostate cancer. Prostate Cancer Prostatic Dis 20, 203–209 (2017). https://doi.org/10.1038/pcan.2016.67
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pcan.2016.67
This article is cited by
-
18F-DCFPyL PET/CT for primary staging in 160 high-risk prostate cancer patients; metastasis detection rate, influence on clinical management and preliminary results of treatment efficacy
European Journal of Nuclear Medicine and Molecular Imaging (2021)