Abstract
Background:
Expanding interest in and use of active surveillance for early state prostate cancer (PC) has increased need for prognostic biomarkers. Using a multi-institutional tissue microarray resource including over 1000 radical prostatectomy samples, we sought to correlate Ki67 expression captured by an automated image analysis system with clinicopathological features and validate its utility as a clinical grade test in predicting cancer-specific outcomes.
Methods:
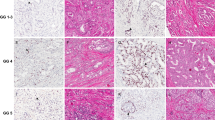
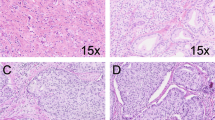
After immunostaining, the Ki67 proliferation index (PI) of tumor areas of each core (three cancer cores/case) was analyzed using a nuclear quantification algorithm (Aperio). We assessed whether Ki67 PI was associated with clinicopathological factors and recurrence-free survival (RFS) including biochemical recurrence, metastasis or PC death (7-year median follow-up).
Results:
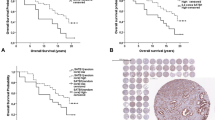
In 1004 PCs (∼4000 tissue cores) Ki67 PI showed significantly higher inter-tumor (0.68) than intra-tumor variation (0.39). Ki67 PI was associated with stage (P<0.0001), seminal vesicle invasion (SVI, P=0.02), extracapsular extension (ECE, P<0.0001) and Gleason score (GS, P<0.0001). Ki67 PI as a continuous variable significantly correlated with recurrence-free, overall and disease-specific survival by multivariable Cox proportional hazard model (hazards ratio (HR)=1.04-1.1, P=0.02–0.0008). High Ki67 score (defined as ⩾5%) was significantly associated with worse RFS (HR=1.47, P=0.0007) and worse overall survival (HR=2.03, P=0.03).
Conclusions:
In localized PC treated by radical prostatectomy, higher Ki67 PI assessed using a clinical grade automated algorithm is strongly associated with a higher GS, stage, SVI and ECE and greater probability of recurrence.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Fisher G, Yang ZH, Kudahetti S, Moller H, Scardino P, Cuzick J et al. Prognostic value of ki-67 for prostate cancer death in a conservatively managed cohort. Br J Cancer 2013; 108: 271–277.
Etzioni R, Penson DF, Legler JM, di Tommaso D, Boer R, Gann PH et al. Overdiagnosis due to prostate-specific antigen screening: lessons from U.S. prostate cancer incidence trends. J Natl Cancer Inst 2002; 94: 981–990.
Berney DM, Gopalan A, Kudahetti S, Fisher G, Ambroisine L, Foster CS et al. Ki-67 and outcome in clinically localised prostate cancer: analysis of conservatively treated prostate cancer patients from the trans-atlantic prostate group study. Br J Cancer 2009; 100: 888–893.
Hawley S, Fazli L, McKenney JK, Simko J, Troyer D, Nicolas M et al. A model for the design and construction of a resource for the validation of prognostic prostate cancer biomarkers: The canary prostate cancer tissue microarray. Adv Anat Pathol 2013; 20: 39–44.
Kachroo N, Gnanapragasam VJ . The role of treatment modality on the utility of predictive tissue biomarkers in clinical prostate cancer: a systematic review. J Cancer Res Clin Oncol 2013; 139: 1–24.
Sebo TJ, Cheville JC, Riehle DL, Lohse CM, Pankratz VS, Myers RP et al. Perineural invasion and MIB-1 positivity in addition to Gleason score are significant preoperative predictors of progression after radical retropubic prostatectomy for prostate cancer. Am J Surg Pathol 2002; 26: 431–439.
Gunia S, Albrecht K, Koch S, Herrmann T, Ecke T, Loy V et al. Ki67 staining index and neuroendocrine differentiation aggravate adverse prognostic parameters in prostate cancer and are characterized by negligible inter-observer variability. World J Urol 2008; 26: 243–250.
Van der Kwast TH . Prognostic prostate tissue biomarkers of potential clinical use. Virchows Arch 2014; 464: 293–300.
Zhao L, Yu N, Guo T, Hou Y, Zeng Z, Yang X et al. Tissue biomarkers for prognosis of prostate cancer: a systematic review and meta-analysis. Cancer Epidemiol Biomarkers Prev 2014; 23: 1047–1054.
Dowsett M, Nielsen TO, A'Hern R, Bartlett J, Coombes RC, Cuzick J et al. Assessment of Ki67 in breast cancer: Recommendations from the International Ki67 in Breast Cancer Working Group. J Natl Cancer Inst 2011; 103: 1656–1664.
Voros A, Csorgo E, Nyari T, Cserni G . An intra- and interobserver reproducibility analysis of the ki-67 proliferation marker assessment on core biopsies of breast cancer patients and its potential clinical implications. Pathobiology 2013; 80: 111–118.
Bosman FT, Carneiro F, Hruban RH, Theise ND . WHO Classification of Tumours of the Digestive System, 4th edn. WHO Press: Geneva, Switzerland, 2010.
Edge S, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A . AJCC cancer staging manual, 7th edn. Springer: New York, NY, 2009.
Luporsi E, Andre F, Spyratos F, Martin PM, Jacquemier J, Penault-Llorca F et al. Ki-67: level of evidence and methodological considerations for its role in the clinical management of breast cancer: analytical and critical review. Breast Cancer Res Treat 2012; 132: 895–915.
Chen L, Yang B . Immunohistochemical analysis of p16, p53, and ki-67 expression in uterine smooth muscle tumors. Int J Gynecol Pathol 2008; 27: 326–332.
Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H et al editors. HO classification of tumours of haematopoietic and lymphoid tissues, 4th edn. Geneva, Switzeland: WHO Press, 2008.
Rekhtman N, Bishop JA . Quick Reference Handbook For Surgical Pathologists, 1st edn. Springer: Berlin, Heidelberg, 2011.
Tretiakova M, Turkyilmaz M, Grushko T, Kocherginsky M, Rubin C, Teh B et al. Topoisomerase IIalpha in wilms' tumour: gene alterations and immunoexpression. J Clin Pathol 2006; 59: 1272–1277.
Tang LH, Gonen M, Hedvat C, Modlin IM, Klimstra DS . Objective quantification of the Ki67 proliferative index in neuroendocrine tumors of the gastroenteropancreatic system: a comparison of digital image analysis with manual methods. Am J Surg Pathol 2012; 36: 1761–1770.
Halvorsen OJ, Haukaas S, Hoisaeter PA, Akslen LA . Maximum ki-67 staining in prostate cancer provides independent prognostic information after radical prostatectomy. Anticancer Res 2001; 21: 4071–4076.
Gravdal K, Halvorsen OJ, Haukaas SA, Akslen LA . Proliferation of immature tumor vessels is a novel marker of clinical progression in prostate cancer. Cancer Res 2009; 69: 4708–4715.
May M, Siegsmund M, Hammermann F, Loy V, Gunia S . Prognostic significance of proliferation activity and neuroendocrine differentiation to predict treatment failure after radical prostatectomy. Scand J Urol Nephrol 2007; 41: 375–381.
Rubio J, Ramos D, Lopez-Guerrero JA, Iborra I, Collado A, Solsona E et al. Immunohistochemical expression of ki-67 antigen, cox-2 and Bax/Bcl-2 in prostate cancer; prognostic value in biopsies and radical prostatectomy specimens. Eur Urol 2005; 48: 745–751.
Miyake H, Muramaki M, Kurahashi T, Takenaka A, Fujisawa M . Expression of potential molecular markers in prostate cancer: correlation with clinicopathological outcomes in patients undergoing radical prostatectomy. Urol Oncol 2010; 28: 145–151.
Deutsch EW, Ball CA, Berman JJ, Bova GS, Brazma A, Bumgarner RE et al. Minimum information specification for in situ hybridization and immunohistochemistry experiments (MISFISHIE). Nat Biotechnol 2008; 26: 305–312.
Altman DG, McShane LM, Sauerbrei W, Taube SE . Reporting recommendations for tumor marker prognostic studies (REMARK): explanation and elaboration. PLoS Med 2012; 9: e1001216.
McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM et al. Reporting recommendations for tumor marker prognostic studies (REMARK). J Natl Cancer Inst 2005; 97: 1180–1184.
Kristiansen G . Diagnostic and prognostic molecular biomarkers for prostate cancer. Histopathology 2012; 60: 125–141.
Shimizu Y, Segawa T, Inoue T, Shiraishi T, Yoshida T, Toda Y et al. Increased akt and phosphorylated akt expression are associated with malignant biological features of prostate cancer in japanese men. BJU Int 2007; 100: 685–690.
Verhoven B, Yan Y, Ritter M, Khor LY, Hammond E, Jones C et al. Ki-67 is an independent predictor of metastasis and cause-specific mortality for prostate cancer patients treated on radiation therapy oncology group (RTOG) 94-08. Int J Radiat Oncol Biol Phys 2013; 86: 317–323.
Royston P, Altman DG, Sauerbrei W . Dichotomizing continuous predictors in multiple regression: a bad idea. Stat Med 2006; 25: 127–141.
Newcomb LF, Thompson IM Jr, Boyer HD, Brooks JD, Carroll PR, Cooperberg MR et al. Outcomes of active surveillance for clinically localized prostate cancer in the prospective, multi-institutional Canary PASS Cohort. J Urol 2016; 195: 313–320.
Acknowledgements
We cordially thank a large and dedicated Canary Foundation team of coordinating center staff, local coordinators, laboratory staff and physicians who have made this study possible. The study was supported by The Canary Foundation; the Department of Defense W81XWH-11-1-0380; the NCI Early Detection Research Network U01 CA086402, CA152737, CA08636815; P30 CA054174; and the Pacific Northwest Prostate Cancer SPORE P50 CA097186.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Prostate Cancer and Prostatic Diseases website
Rights and permissions
About this article
Cite this article
Tretiakova, M., Wei, W., Boyer, H. et al. Prognostic value of Ki67 in localized prostate carcinoma: a multi-institutional study of >1000 prostatectomies. Prostate Cancer Prostatic Dis 19, 264–270 (2016). https://doi.org/10.1038/pcan.2016.12
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pcan.2016.12
This article is cited by
-
Inter- and intra-tumor heterogeneity of metastatic prostate cancer determined by digital spatial gene expression profiling
Nature Communications (2021)
-
Ki-67 is an independent predictor of prostate cancer death in routine needle biopsy samples: proving utility for routine assessments
Modern Pathology (2019)
-
Development and validation of a novel automated Gleason grade and molecular profile that define a highly predictive prostate cancer progression algorithm-based test
Prostate Cancer and Prostatic Diseases (2018)