Abstract
Background:
Partner of SLD5 1 (PSF1) is an evolutionarily conserved DNA replication factor. Previous studies have suggested that transcriptional activity of the PSF1 gene correlated with malignancy of cancer cells. The objective of the current study was to evaluate the relationship between PSF1 expression and the clinical features of prostate cancer.
Methods:
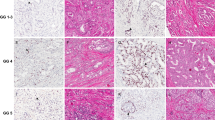
We determined the expression of PSF1 in 120 needle biopsy samples of prostate cancer by immunohistochemistry. We divided patients into PSF1-positive or -negative groups and analyzed the relationships between the expression of PSF1, the Gleason score, PSA level, TNM classification and prognosis.
Results:
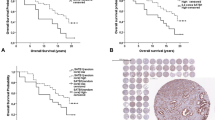
Our results showed that the PSF1 expression correlated significantly with PSA values at diagnosis (P=0.0028), with tumor grade (P<0.0001), and with clinical stage (P=0.0005). Moreover, the PSF1 expression correlated significantly with overall survival (hazard ratio (HR) 5.5; 95% confidence interval (CI) 2.17–15.8; P=0.003) and progression-free survival in 99 consecutive patients with prostate cancer. Noteworthy, the prognosis of PSF1-positive cases was also worse in patients with a Gleason score of 8–10 (HR 3.7; 95% CI 1.28–13.43; P=0.0143). Limitations include that this study had a retrospective design, that patients in the study were heterogeneous and included those with early and advanced cancer, and that small tumor fragments may not be representative of the entire carcinoma.
Conclusions:
PSF1 is expressed in high-grade prostate cancer and may be a useful biomarker to identify patients with a poor prognosis at the time of diagnosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Baade PD, Youlden DR, Cramb SM, Dunn J, Gardiner RA . Epidemiology of prostate cancer in the Asia-Pacific region. Prostate Int 2013; 1: 47–58.
Williams S, Chiong E, Lojanapiwat B, Umbas R, Akaza H Asian Oncology Summit 2013. Management of prostate cancer in Asia: resource-stratified guidelines from the Asian Oncology Summit 2013. Lancet Oncol 2013; 14: e524–e534.
Hankey BF, Feuer EJ, Clegg LX, Hayes RB, Legler JM, Prorok PC et al. Cancer surveillance series: interpreting trends in prostate cancer—part I: Evidence of the effects of screening in recent prostate cancer incidence, mortality, and survival rates. J Natl Cancer Inst 1999; 91: 1017–1024.
Nelson WG, De Marzo AM, Isaacs WB . Prostate cancer. N Engl J Med 2003; 349: 366–381.
Gulley J, Figg WD, Dahut WL . Treatment options for androgen-independent prostate cancer. Clin Adv Hematol Oncol 2003; 1: 49–57.
Epstein JI, Allsbrook WC Jr ., Amin MB, Egevad LL ISUP Grading Committee. The 2005 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma. Am J Surg Pathol 2005; 29: 1228–1242.
Akimoto S, Ichikawa T, Ito H . Relationship of prostate-specific antigen levels to prostate volume and age in mass screening subjects. Urol Int 1998; 60: 216–219.
Fradet Y . Biomarkers in prostate cancer diagnosis and prognosis: beyond prostate-specific antigen. Curr Opin Urol 2009; 19: 243–246.
Bickers B, Aukim-Hastie C . New molecular biomarkers for the prognosis and management of prostate cancer—the post PSA era. Anticancer Res 2009; 29: 3289–3298.
Fall K, Garmo H, Andren O, Bill-Axelson A, Adolfsson J, Adami HO et al. Prostate-specific antigen levels as a predictor of lethal prostate cancer. J Natl Cancer Inst 2007; 99: 526–532.
Epstein JI, Allsbrook WC Jr ., Amin MB, Egevad LL . Update on the Gleason grading system for prostate cancer: results of an international consensus conference of urologic pathologists. Adv Anat Pathol 2006; 13: 57–59.
Takayama Y, Kamimura Y, Okawa M, Muramatsu S, Sugino A, Araki H . GINS, a novel multiprotein complex required for chromosomal DNA replication in budding yeast. Genes Dev 2003; 17: 1153–1165.
Bauerschmidt C, Pollok S, Kremmer E, Nasheuer HP, Grosse F . Interactions of human Cdc45 with the Mcm2-7 complex, the GINS complex, and DNA polymerases delta and epsilon during S phase. Genes Cells 2007; 12: 745–758.
Gambus A, Jones RC, Sanchez-Diaz A, Kanemaki M, van Deursen F, Edmondson RD et al. GINS maintains association of Cdc45 with MCM in replisome progression complexes at eukaryotic DNA replication forks. Nat Cell Biol 2006; 8: 358–366.
Kanemaki M, Sanchez-Diaz A, Gambus A, Labib K . Functional proteomic identification of DNA replication proteins by induced proteolysis in vivo. Nature 2003; 423: 720–724.
Moyer SE, Lewis PW, Botchan MR . Isolation of the Cdc45/Mcm2-7/GINS (CMG) complex, a candidate for the eukaryotic DNA replication fork helicase. Proc Natl Acad Sci USA 2006; 103: 10236–10241.
Pacek M, Tutter AV, Kubota Y, Takisawa H, Walter JC . Localization of MCM2-7, Cdc45, and GINS to the site of DNA unwinding during eukaryotic DNA replication. Mol Cell 2006; 21: 581–587.
Ueno M, Itoh M, Kong L, Sugihara K, Asano M, Takakura N . PSF1 is essential for early embryogenesis in mice. Mol Cell Biol 2005; 25: 10528–10532.
Ueno M, Itoh M, Sugihara K, Asano M, Takakura N . Both alleles of PSF1 are required for maintenance of pool size of immature hematopoietic cells and acute bone marrow regeneration. Blood 2009; 113: 555–562.
Nagahama Y, Ueno M, Miyamoto S, Morii E, Minami T, Mochizuki N et al. PSF1, a DNA replication factor expressed widely in stem and progenitor cells, drives tumorigenic and metastatic properties. Cancer Res 2010; 70: 1215–1224.
Matsui T, Kinugasa Y, Tahara H, Kanakura Y, Takakura N . Possible role of mural cell-covered mature blood vessels in inducing drug resistance in cancer-initiating cells. Am J Pathol 2013; 182: 1790–1799.
Kinugasa Y, Matsui T, Takakura N . CD44 expressed on cancer-associated fibroblasts is a functional molecule supporting the stemness and drug resistance of malignant cancer cells in the tumor microenvironment. Stem Cells 2014; 32: 145–156.
Miller DC, Hafez KS, Stewart A, Montie JE, Wei JT . Prostate carcinoma presentation, diagnosis, and staging: an update form the National Cancer Data Base. Cancer 2003; 98: 1169–1178.
Naito H, Takara K, Wakabayashi T, Kawahara H, Kidoya H, Takakura N . Changes in blood vessel maturation in the fibrous cap of the tumor rim. Cancer Sci 2012; 103: 433–438.
Kidoya H, Ueno M, Yamada Y, Mochizuki N, Nakata M, Yano T et al. Spatial and temporal role of the apelin/APJ system in the caliber size regulation of blood vessels during angiogenesis. EMBO J 2008; 27: 522–534.
Naito H, Kidoya H, Sakimoto S, Wakabayashi T, Takakura N . Identification and characterization of a resident vascular stem/progenitor cell population in preexisting blood vessels. EMBO J 2012; 31: 842–855.
D'Amico AV, Halabi S, Vollmer R, Loffredo M, McMahon E, Sanford B et al. p53 protein expression status and recurrence in men treated with radiation and androgen suppression therapy for higher-risk prostate cancer: a prospective phase II Cancer and Leukemia Group B Study (CALGB 9682). Urology 2008; 71: 933–937.
Vergis R, Corbishley CM, Thomas K, Horwich A, Huddart R, Khoo V et al. Expression of Bcl-2, p53, and MDM2 in localized prostate cancer with respect to the outcome of radical radiotherapy dose escalation. Int J Radiat Oncol Biol Phys 2010; 78: 35–41.
Kristiansen G . Diagnostic and prognostic molecular biomarkers for prostate cancer. Histopathology 2012; 60: 125–141.
Fisher G, Yang ZH, Kudahetti S, Moller H, Scardino P, Cuzick J et al. Prognostic value of Ki-67 for prostate cancer death in a conservatively managed cohort. Br J Cancer 2013; 108: 271–277.
Bubendorf L, Sauter G, Moch H, Schmid HP, Gasser TC, Jordan P et al. Ki67 labelling index: an independent predictor of progression in prostate cancer treated by radical prostatectomy. J Pathol 1996; 178: 437–441.
Matsuura H, Hayashi N, Kawamura J, Shiraishi T, Yatani R . Prognostic significance of Ki-67 expression in advanced prostate cancers in relation to disease progression after androgen ablation. Eur Urol 2000; 37: 212–217.
Acknowledgements
We thank Ms K Fukuhara, Ms N Fujimoto, Ms C Takeshita and Ms Y Morioka for technical assistance. This work was partly supported by the Japanese Ministry of Education, Culture, Sports, Science and Technology (NT) and Grants-in-Aid for Scientific Research (KAKENHI 25830080) from Japan Society for the Promotion of Science (JSPS) (HN).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Prostate Cancer and Prostatic Diseases website
Supplementary information
Rights and permissions
About this article
Cite this article
Tahara, H., Naito, H., Kise, K. et al. Evaluation of PSF1 as a prognostic biomarker for prostate cancer. Prostate Cancer Prostatic Dis 18, 56–62 (2015). https://doi.org/10.1038/pcan.2014.46
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pcan.2014.46
This article is cited by
-
Androgen receptor binding sites enabling genetic prediction of mortality due to prostate cancer in cancer-free subjects
Nature Communications (2023)
-
Identification of GINS1 as a therapeutic target in the cancer patients infected with COVID-19: a bioinformatics and system biology approach
Hereditas (2022)
-
Anlotinib inhibits synovial sarcoma by targeting GINS1: a novel downstream target oncogene in progression of synovial sarcoma
Clinical and Translational Oncology (2019)