Abstract
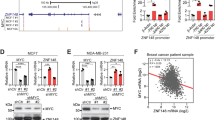
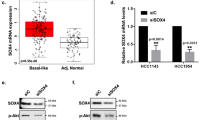
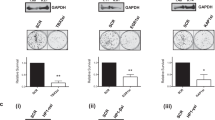
T-box 2 (TBX2) is a transcription factor involved in mammary development and is known to be overexpressed in a subset of aggressive breast cancers. TBX2 has previously been shown to repress growth control genes such as p14ARF and p21WAF1/cip1. In this study we show that TBX2 drives proliferation in breast cancer cells and this is abrogated after TBX2 small interfering RNA (siRNA) knockdown or after the expression of a dominant-negative TBX2 protein. Using microarray analysis we identified a large cohort of novel TBX2-repressed target genes including the breast tumour suppressor NDRG1 (N-myc downregulated gene 1). We show that TBX2 targets NDRG1 through a previously undescribed mechanism involving the recruitment of early growth response 1 (EGR1). We show EGR1 is required for the ability of TBX2 to repress NDRG1 and drive cell proliferation. We show that TBX2 interacts with EGR1 and that TBX2 requires EGR1 to target the NDRG1 proximal promoter. Abrogation of either TBX2 or EGR1 expression is accompanied by the upregulation of cell senescence and apoptotic markers. NDRG1 can recapitulate these effects when transfected into TBX2-expressing cells. Together, these data identify a novel mechanism for TBX2-driven oncogenesis and highlight the importance of NDRG1 as a growth control gene in breast tissue.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Boogerd KJ, Wong LY, Christoffels VM, Klarenbeek M, Ruijter JM, Moorman AF et al. (2008). Msx1 and Msx2 are functional interacting partners of T-box factors in the regulation of Connexin43. Cardiovasc Res 78: 485–493.
Buckley NE, Hosey AM, Gorski JJ, Purcell JW, Mulligan JM, Harkin DP et al. (2007). BRCA1 regulates IFN-gamma signaling through a mechanism involving the type I IFNs. Mol Cancer Res 5: 261–270.
Carlson H, Ota S, Song Y, Chen Y, Hurlin PJ . (2002). Tbx3 impinges on the p53 pathway to suppress apoptosis, facilitate cell transformation and block myogenic differentiation. Oncogene 21: 3827–3835.
Chapman DL, Garvey N, Hancock S, Alexiou M, Agulnik SI, Gibson-Brown JJ et al. (1996). Expression of the T-box family genes, Tbx1-Tbx5, during early mouse development. Dev Dyn 206: 379–390.
de Belle I, Huang RP, Fan Y, Liu C, Mercola D, Adamson ED . (1999). p53 and Egr-1 additively suppress transformed growth in HT1080 cells but Egr-1 counteracts p53-dependent apoptosis. Oncogene 18: 3633–3642.
Habets PE, Moorman AF, Clout DE, van Roon MA, Lingbeek M, van Lohuizen M et al. (2002). Cooperative action of Tbx2 and Nkx2.5 inhibits ANF expression in the atrioventricular canal: implications for cardiac chamber formation. Genes Dev 16: 1234–1246.
Hosey AM, Gorski JJ, Murray MM, Quinn JE, Chung WY, Stewart GE et al. (2007). Molecular basis for estrogen receptor alpha deficiency in BRCA1-linked breast cancer. J Natl Cancer Inst 99: 1683–1694.
Ismail A, Bateman A . (2009). Expression of TBX2 promotes anchorage-independent growth and survival in the p53-negative SW13 adrenocortical carcinoma. Cancer Lett 278: 230–240.
Jacobs JJ, Keblusek P, Robanus-Maandag E, Kristel P, Lingbeek M, Nederlof PM et al. (2000). Senescence bypass screen identifies TBX2, which represses Cdkn2a (p19(ARF)) and is amplified in a subset of human breast cancers. Nat Genet 26: 291–299.
Kennedy RD, Gorski JJ, Quinn JE, Stewart GE, James CR, Moore S et al. (2005). BRCA1 and c-Myc associate to transcriptionally repress psoriasin, a DNA damage-inducible gene. Cancer Res 65: 10265–10272.
Krones-Herzig A, Adamson E, Mercola D . (2003). Early growth response 1 protein, an upstream gatekeeper of the p53 tumor suppressor, controls replicative senescence. Proc Natl Acad Sci USA 100: 3233–3238.
Le NT, Richardson DR . (2004). Iron chelators with high antiproliferative activity up-regulate the expression of a growth inhibitory and metastasis suppressor gene: a link between iron metabolism and proliferation. Blood 104: 2967–2975.
Lingbeek ME, Jacobs JJ, van Lohuizen M . (2002). The T-box repressors TBX2 and TBX3 specifically regulate the tumor suppressor gene p14ARF via a variant T-site in the initiator. J Biol Chem 277: 26120–26127.
Lu S, Becker KA, Hagen MJ, Yan H, Roberts AL, Mathews LA et al. (2008). Transcriptional responses to estrogen and progesterone in mammary gland identify networks regulating p53 activity. Endocrinology 149: 4809–4820.
Mikkelsen TS, Ku M, Jaffe DB, Issac B, Lieberman E, Giannoukos G et al. (2007). Genome-wide maps of chromatin state in pluripotent and lineage-committed cells. Nature 448: 553–560.
Mullan PB, Quinn JE, Gilmore PM, McWilliams S, Andrews H, Gervin C et al. (2001). BRCA1 and GADD45 mediated G2/M cell cycle arrest in response to antimicrotubule agents. Oncogene 20: 6123–6131.
Prince S, Carreira S, Vance KW, Abrahams A, Goding CR . (2004). Tbx2 directly represses the expression of the p21(WAF1) cyclin-dependent kinase inhibitor. Cancer Res 64: 1669–1674.
Qian Y, Zhang J, Yan B, Chen X . (2008). DEC1, a basic helix-loop-helix transcription factor and a novel target gene of the p53 family, mediates p53-dependent premature senescence. J Biol Chem 283: 2896–2905.
Ronski K, Sanders M, Burleson JA, Moyo V, Benn P, Fang M . (2005). Early growth response gene 1 (EGR1) is deleted in estrogen receptor-negative human breast carcinoma. Cancer 104: 925–930.
Roylance R, Gorman P, Harris W, Liebmann R, Barnes D, Hanby A et al. (1999). Comparative genomic hybridization of breast tumors stratified by histological grade reveals new insights into the biological progression of breast cancer. Cancer Res 59: 1433–1436.
Serrano M, Lin AW, McCurrach ME, Beach D, Lowe SW . (1997). Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 88: 593–602.
Sinclair CS, Adem C, Naderi A, Soderberg CL, Johnson M, Wu K et al. (2002). TBX2 is preferentially amplified in BRCA1- and BRCA2-related breast tumors. Cancer Res 62: 3587–3591.
Stein S, Thomas EK, Herzog B, Westfall MD, Rocheleau JV, Jackson II RS et al. (2004). NDRG1 is necessary for p53-dependent apoptosis. J Biol Chem 279: 48930–48940.
Tirkkonen M, Johannsson O, Agnarsson BA, Olsson H, Ingvarsson S, Karhu R et al. (1997). Distinct somatic genetic changes associated with tumor progression in carriers of BRCA1 and BRCA2 germ-line mutations. Cancer Res 57: 1222–1227.
Vance KW, Carreira S, Brosch G, Goding CR . (2005). Tbx2 is overexpressed and plays an important role in maintaining proliferation and suppression of senescence in melanomas. Cancer Res 65: 2260–2268.
Virolle T, Adamson ED, Baron V, Birle D, Mercola D, Mustelin T et al. (2001). The Egr-1 transcription factor directly activates PTEN during irradiation-induced signalling. Nat Cell Biol 3: 1124–1128.
Yu J, Baron V, Mercola D, Mustelin T, Adamson ED . (2007). A network of p73, p53 and Egr1 is required for efficient apoptosis in tumor cells. Cell Death Differ 14: 436–446.
Zagurovskaya M, Shareef MM, Das A, Reeves A, Gupta S, Sudol M et al. (2009). EGR-1 forms a complex with YAP-1 and upregulates Bax expression in irradiated prostate carcinoma cells. Oncogene 28: 1121–1131.
Zhang J, Chen S, Zhang W, Liu X, Shi H, Che H et al. (2008). Human differentiation-related gene NDRG1 is a Myc downstream-regulated gene that is repressed by Myc on the core promoter region. Gene 417: 5–12.
Zhang P, Tchou-Wong KM, Costa M . (2007). Egr-1 mediates hypoxia-inducible transcription of the NDRG1 gene through an overlapping Egr-1/Sp1 binding site in the promoter. Cancer Res 67: 9125–9133.
Acknowledgements
Special thanks to Dr James Murray (CCRCB, QUB) for gift of sheep anti-human NDRG1 antibody, Professor Carlos Caldas (University of Cambridge) for TBX2-expressing breast cancer cell lines and Professor Max Costa (University of New York) for NDRG1 promoter luciferase constructs. We also thank Professor Neil Perkins (University of Bristol) for reading this manuscript. This study was supported by the Department of Employment and Learning Northern Ireland (KLR), Breast Cancer Campaign (NTC, PBM), Action Cancer (HF, PBM), OSF (ZCD’C), Cancer Research UK (GJO’B, PGJ, DPH) and Research and Development Northern Ireland (NEB).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Redmond, K., Crawford, N., Farmer, H. et al. T-box 2 represses NDRG1 through an EGR1-dependent mechanism to drive the proliferation of breast cancer cells. Oncogene 29, 3252–3262 (2010). https://doi.org/10.1038/onc.2010.84
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.84
Keywords
This article is cited by
-
Bortezomib induces methylation changes in neuroblastoma cells that appear to play a significant role in resistance development to this compound
Scientific Reports (2021)
-
Functional and regulatory aspects of oxidative stress response in X monosomy
In Vitro Cellular & Developmental Biology - Animal (2021)
-
TBX2 interacts with heterochromatin protein 1 to recruit a novel repression complex to EGR1-targeted promoters to drive the proliferation of breast cancer cells
Oncogene (2019)
-
NDRG1 inhibition sensitizes osteosarcoma cells to combretastatin A-4 through targeting autophagy
Cell Death & Disease (2017)
-
TBX2 represses PTEN in rhabdomyosarcoma and skeletal muscle
Oncogene (2016)