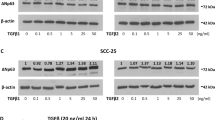
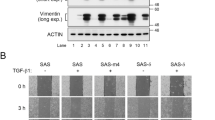
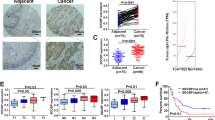
Abstract
The progression and negative outcome of a variety of human carcinomas are intimately associated with aberrant activity of the c-Met oncogene. The underlying cause of this dysregulation, however, remains a subject of discussion, as the majority of cancer patients do not present with activating mutations in c-Met receptor itself. In this study, we show that the oncogenic protease matriptase is ubiquitously co-expressed with the c-Met in human squamous cell carcinomas and amplifies migratory and proliferative responses of primary epithelial cells to the cognate ligand for c-Met, pro-hepatocyte growth factor/scatter factor (proHGF/SF), through c-Met and Gab1 signaling. Furthermore, the selective genetic ablation of c-Met from matriptase-expressing keratinocytes completely negates the oncogenic potential of matriptase. In addition, matriptase-dependent carcinoma formation could be blocked by the pharmacological inhibition of the Akt–mammalian target of Rapamycin (mTor) pathway. Our data identify matriptase as an initiator of c-Met-Akt–mTor-dependent signaling axis in tumors and reveal mTor activation as an essential component of matriptase/c-Met-induced carcinogenesis. The study provides a specific example of how epithelial transformation can be promoted by epigenetic acquisition of the capacity to convert a widely available paracrine growth factor precursor to its signaling competent state.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Amornphimoltham P, Patel V, Sodhi A, Nikitakis NG, Sauk JJ, Sausville EA et al. (2005). Mammalian target of rapamycin, a molecular target in squamous cell carcinomas of the head and neck. Cancer Res 65: 9953–9961.
Birchmeier C, Birchmeier W, Gherardi E, Vande Woude GF . (2003). Met, metastasis, motility and more. Nat Rev Mol Cell Biol 4: 915–925.
Bladt F, Riethmacher D, Isenmann S, Aguzzi A, Birchmeier C . (1995). Essential role for the c-met receptor in the migration of myogenic precursor cells into the limb bud. Nature 376: 768–771.
Bugge TH, Flick MJ, Daugherty CC, Degen JL . (1995). Plasminogen deficiency causes severe thrombosis but is compatible with development and reproduction. Genes Dev 9: 794–807.
Camerer E, Barker A, Duong DN, Ganesan R, Kataoka H, Cornelissen I et al. (2010). Local protease signaling contributes to neural tube closure in the mouse embryo. Dev Cell 18: 25–38.
Carmeliet P, Schoonjans L, Kieckens L, Ream B, Degen J, Bronson R et al. (1994). Physiological consequences of loss of plasminogen activator gene function in mice. Nature 368: 419–424.
Chmielowiec J, Borowiak M, Morkel M, Stradal T, Munz B, Werner S et al. (2007). c-Met is essential for wound healing in the skin. J Cell Biol 177: 151–162.
Dai C, Saleem MA, Holzman LB, Mathieson P, Liu Y . (2010). Hepatocyte growth factor signaling ameliorates podocyte injury and proteinuria. Kidney Int 77: 962–973.
Gailani D, Lasky NM, Broze Jr GJ . (1997). A murine model of factor XI deficiency. Blood Coagul Fibrinolysis 8: 134–144.
Gherardi E, Sandin S, Petoukhov MV, Finch J, Youles ME, Ofverstedt LG et al. (2006). Structural basis of hepatocyte growth factor/scatter factor and MET signalling. Proc Natl Acad Sci USA 103: 4046–4051.
Hanahan D, Weinberg RA . (2000). The hallmarks of cancer. Cell 100: 57–70.
Hartmann G, Prospero T, Brinkmann V, Ozcelik C, Winter G, Hepple J et al. (1998). Engineered mutants of HGF/SF with reduced binding to heparan sulphate proteoglycans, decreased clearance and enhanced activity in vivo. Curr Biol 8: 125–134.
Herter S, Piper DE, Aaron W, Gabriele T, Cutler G, Cao P et al. (2005). Hepatocyte growth factor is a preferred in vitro substrate for human Hepsin, a membrane-anchored serine protease implicated in prostate and ovarian cancers. Biochem J 390: 125–136.
Huh CG, Factor VM, Sanchez A, Uchida K, Conner EA, Thorgeirsson SS . (2004). Hepatocyte growth factor/c-met signaling pathway is required for efficient liver regeneration and repair. Proc Natl Acad Sci USA 101: 4477–4482.
Itoh H, Naganuma S, Takeda N, Miyata S, Uchinokura S, Fukushima T et al. (2004). Regeneration of injured intestinal mucosa is impaired in hepatocyte growth factor activator-deficient mice. Gastroenterology 127: 1423–1435.
Jin X, Yagi M, Akiyama N, Hirosaki T, Higashi S, Lin CY et al. (2006). Matriptase activates stromelysin (MMP-3) and promotes tumor growth and angiogenesis. Cancer Sci 97: 1327–1334.
Kang JY, Dolled-Filhart M, Ocal IT, Singh B, Lin CY, Dickson RB et al. (2003). Tissue microarray analysis of hepatocyte growth factor/Met pathway components reveals a role for Met, matriptase, and hepatocyte growth factor activator inhibitor 1 in the progression of node-negative breast cancer. Cancer Res 63: 1101–1105.
Kirchhofer D, Lipari MT, Santell L, Billeci KL, Maun HR, Sandoval WN et al. (2007). Utilizing the activation mechanism of serine proteases to engineer hepatocyte growth factor into a Met antagonist. Proc Natl Acad Sci USA 104: 5306–5311.
Knudsen BS, Vande Woude G . (2008). Showering c-MET-dependent cancers with drugs. Curr Opin Genet Dev 18: 87–96.
Lee SL, Dickson RB, Lin CY . (2000). Activation of hepatocyte growth factor and urokinase/plasminogen activator by matriptase, an epithelial membrane serine protease. J Biol Chem 275: 36720–36725.
Lichti U, Weinberg WC, Goodman L, Ledbetter S, Dooley T, Morgan D et al. (1993). in vivo regulation of murine hair growth: insights from grafting defined cell populations onto nude mice. J Invest Dermatol 101: 124S–129S.
List K, Bugge TH, Szabo R . (2006a). Matriptase: potent proteolysis on the cell surface. Mol Med 12: 1–7.
List K, Haudenschild CC, Szabo R, Chen W, Wahl SM, Swaim W et al. (2002). Matriptase/MT-SP1 is required for postnatal survival, epidermal barrier function, hair follicle development, and thymic homeostasis. Oncogene 21: 3765–3779.
List K, Szabo R, Molinolo A, Nielsen BS, Bugge TH . (2006b). Delineation of matriptase protein expression by enzymatic gene trapping suggests diverging roles in barrier function, hair formation, and squamous cell carcinogenesis. Am J Pathol 168: 1513–1525.
List K, Szabo R, Molinolo A, Sriuranpong V, Redeye V, Murdock T et al. (2005). Deregulated matriptase causes ras-independent multistage carcinogenesis and promotes ras-mediated malignant transformation. Genes Dev 19: 1934–1950.
Lopez-Otin C, Hunter T . (2010). The regulatory crosstalk between kinases and proteases in cancer. Nat Rev Cancer 10: 278–292.
Ma H, Saenko M, Opuko A, Togawa A, Soda K, Marlier A et al. (2009). Deletion of the Met receptor in the collecting duct decreases renal repair following ureteral obstruction. Kidney Int 76: 868–876.
McCawley LJ, Crawford HC, King Jr LE, Mudgett J, Matrisian LM . (2004). A protective role for matrix metalloproteinase-3 in squamous cell carcinoma. Cancer Res 64: 6965–6972.
Molinolo AA, Amornphimoltham P, Squarize CH, Castilho RM, Patel V, Gutkind JS . (2009). Dysregulated molecular networks in head and neck carcinogenesis. Oral Oncol 45: 324–334.
Molinolo AA, Hewitt SM, Amornphimoltham P, Keelawat S, Rangdaeng S, Meneses Garcia A et al. (2007). Dissecting the Akt/mammalian target of rapamycin signaling network: emerging results from the head and neck cancer tissue array initiative. Clin Cancer Res 13: 4964–4973.
Naldini L, Tamagnone L, Vigna E, Sachs M, Hartmann G, Birchmeier W et al. (1992). Extracellular proteolytic cleavage by urokinase is required for activation of hepatocyte growth factor/scatter factor. EMBO J 11: 4825–4833.
Owen KA, Qiu D, Alves J, Schumacher AM, Kilpatrick LM, Li J et al. (2010). Pericellular activation of hepatocyte growth factor by the transmembrane serine proteases matriptase and hepsin, but not by the membrane-associated protease uPA. Biochem J 426: 219–228.
Park M, Dean M, Cooper CS, Schmidt M, O'Brien SJ, Blair DG et al. (1986). Mechanism of met oncogene activation. Cell 45: 895–904.
Pauer HU, Renne T, Hemmerlein B, Legler T, Fritzlar S, Adham I et al. (2004). Targeted deletion of murine coagulation factor XII gene-a model for contact phase activation in vivo. Thromb Haemost 92: 503–508.
Peek M, Moran P, Mendoza N, Wickramasinghe D, Kirchhofer D . (2002). Unusual proteolytic activation of pro-hepatocyte growth factor by plasma kallikrein and coagulation factor XIa. J Biol Chem 277: 47804–47809.
Raimondi AR, Molinolo A, Gutkind JS . (2009). Rapamycin prevents early onset of tumorigenesis in an oral-specific K-ras and p53 two-hit carcinogenesis model. Cancer Res 69: 4159–4166.
Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D et al. (2004). ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 6: 1–6.
Schmidt C, Bladt F, Goedecke S, Brinkmann V, Zschiesche W, Sharpe M et al. (1995). Scatter factor/hepatocyte growth factor is essential for liver development. Nature 373: 699–702.
Seiwert TY, Jagadeeswaran R, Faoro L, Janamanchi V, Nallasura V, El Dinali M et al. (2009). The MET receptor tyrosine kinase is a potential novel therapeutic target for head and neck squamous cell carcinoma. Cancer Res 69: 3021–3031.
Shanmukhappa K, Matte U, Degen JL, Bezerra JA . (2009). Plasmin-mediated proteolysis is required for hepatocyte growth factor activation during liver repair. J Biol Chem 284: 12917–12923.
Shimomura T, Miyazawa K, Komiyama Y, Hiraoka H, Naka D, Morimoto Y et al. (1995). Activation of hepatocyte growth factor by two homologous proteases, blood-coagulation factor XIIa and hepatocyte growth factor activator. Eur J Biochem 229: 257–261.
Squarize CH, Castilho RM, Gutkind JS . (2008). Chemoprevention and treatment of experimental Cowden′s disease by mTOR inhibition with rapamycin. Cancer Res 68: 7066–7072.
Szabo R, Bugge TH . (2008). Type II transmembrane serine proteases in development and disease. Int J Biochem Cell Biol 40: 1297–1316.
Takeuchi T, Harris JL, Huang W, Yan KW, Coughlin SR, Craik CS . (2000). Cellular localization of membrane-type serine protease 1 and identification of protease-activated receptor-2 and single-chain urokinase-type plasminogen activator as substrates. J Biol Chem 275: 26333–26342.
Uehara Y, Minowa O, Mori C, Shiota K, Kuno J, Noda T et al. (1995). Placental defect and embryonic lethality in mice lacking hepatocyte growth factor/scatter factor. Nature 373: 702–705.
Uhland K . (2006). Matriptase and its putative role in cancer. Cell Mol Life Sci 63: 2968–2978.
Vivanco I, Sawyers CL . (2002). The phosphatidylinositol 3-Kinase AKT pathway in human cancer. Nat Rev Cancer 2: 489–501.
Williams-Simons L, Westphal H . (1999). EIIaCre—utility of a general deleter strain. Transgenic Res 8: 53–54.
Wu Q, Yu D, Post J, Halks-Miller M, Sadler JE, Morser J . (1998). Generation and characterization of mice deficient in hepsin, a hepatic transmembrane serine protease. J Clin Invest 101: 321–326.
Zhou HM, Nichols A, Meda P, Vassalli JD . (2000). Urokinase-type plasminogen activator and its receptor synergize to promote pathogenic proteolysis. Embo J 19: 4817–4826.
Acknowledgements
We thank Dr Snorri Thorgeirsson for floxed Hgfr mice. Supported by NIDCR Intramural Research Program. We thank Dr Mary Jo Danton for critically reviewing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Szabo, R., Rasmussen, A., Moyer, A. et al. c-Met-induced epithelial carcinogenesis is initiated by the serine protease matriptase. Oncogene 30, 2003–2016 (2011). https://doi.org/10.1038/onc.2010.586
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.586
Keywords
This article is cited by
-
TMPRSS13 promotes cell survival, invasion, and resistance to drug-induced apoptosis in colorectal cancer
Scientific Reports (2020)
-
Loss of SPINT2 expression frequently occurs in glioma, leading to increased growth and invasion via MMP2
Cellular Oncology (2020)
-
Inhibition of HGF/MET signaling decreases overall tumor burden and blocks malignant conversion in Tpl2-related skin cancer
Oncogenesis (2019)
-
SPINT2 is hypermethylated in both IDH1 mutated and wild-type glioblastomas, and exerts tumor suppression via reduction of c-Met activation
Journal of Neuro-Oncology (2019)
-
Cell surface–anchored serine proteases in cancer progression and metastasis
Cancer and Metastasis Reviews (2019)