Abstract
The coordination of pelvic visceral activity with appropriate elimination behaviors is a complex task that requires reciprocal communication between the brain and pelvic organs. Barrington's nucleus, located in the pons, is central to a circuit involved in this function. Barrington's nucleus neurons project to both pelvic visceral motorneurons and cerebral norepinephrine neurons that modulate behavior. This circuit coordinates the descending limb of the micturition reflex with a central limb that initiates arousal and shifts the focus of attention to facilitate elimination behavior. The same circuitry that links the bladder and brain enables pathological processes in one target of the circuit to be expressed in the other. Urological disorders can, therefore, have cognitive and behavioral consequences by affecting components of this circuit; and in the opposing direction, psychosocial stressors can produce voiding dysfunctions and bladder pathology. The stress-related neuropeptide, corticotropin-releasing factor, which is prominent in Barrington's nucleus neurons, is a potential mediator of these effects.
Key Points
-
Barrington's nucleus in the pons coordinates the descending visceral limb of the micturition reflex with a central limb that initiates arousal and facilitates elimination behavior
-
The stress-related neurotransmitter, corticotropin-releasing factor, regulates both limbs of the micturition reflex owing to its actions in the central and spinal projections of Barrington's nucleus neurons
-
Pelvic visceral disorders can affect the major brain norepinephrine nucleus, the locus coeruleus and have neurobehavioral consequences
-
Psychosocial stressors can produce voiding dysfunctions and bladder pathology via spinal projections of Barrington's nucleus neurons
-
Neural circuits linking the brain and bladder provide a basis for the coexistence of bladder and neurobehavioral symptoms
-
Pharmacological modulation of neurotransmitters expressed by Barrington's nucleus neurons—such as corticotropin-releasing factor—offers a novel approach for the treatment of bladder disorders
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Longstreth, G. F. Irritable bowel syndrome and chronic pelvic pain. Obstet. Gynecol. Surv. 49, 505–507 (1994).
Lydiard, R. B., Fossey, M. D., Marsh, W. & Ballenger, J. C. Prevalence of psychiatric disorders in patients with irritable bowel syndrome. Psychosomatics 34, 229–234 (1993).
Lydiard, R. B. et al. Panic disorder and gastrointestinal symptoms: findings from the NIMH epidemiologic catchment area project. Am. J. Psych. 151, 64–70 (1994).
Whorwell, P. J., Lupton, E. W., Erduran, D. & Wilson, K. Bladder smooth muscle dysfunction in patients with irritable bowel syndrome. Gut 27, 1014–1017 (1986).
Whorwell, P. J., McCallum, M., Creed, F. H. & Roberts, C. T. Non-colonic features of irritable bowel syndrome. Gut 27, 37–40 (1987).
Perry, S., McGrother, C. W. & Turner, K. An investigation of the relationship between anxiety and depression and urge incontinence in women: development of a psychological model. Br. J. Health Psychol. 11, 463–482 (2006).
Von Gontard, A. & Hollmann, E. Comorbidity of functional urinary incontinence and encopresis: somatic and behavioral associations. J. Urol. 171, 2644–2647 (2004).
Baeyens, D. et al. Attention deficit/hyperactivity disorder in children with nocturnal enuresis. J. Urol. 171, 2576–2579 (2004).
Desjardins, C., Maruniak, J. A. & Bronson, F. H. Social rank in house mice: differentiation revealed by ultraviolet visualization of urinary marking patterns. Science 182, 939–941 (1973).
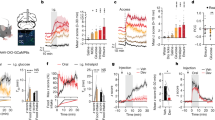
Valentino, R. J., Chen, S., Zhu, Y. & Aston-Jones, G. Evidence for divergent projections of corticotropin-releasing hormone neurons of Barrington's nucleus to the locus coeruleus and spinal cord. Brain Res. 732, 1–15 (1996).
Aston-Jones, G., Shipley, M. T. & Grzanna, R. in The Rat Brain (ed. Paxinos, G.) 183–213 (Academic Press, London, 1995).
Swanson, L. W. & Hartman, B. K. The central adrenergic system. An immunofluorescence study of the location of cell bodies and their efferent connections in the rat using dopamine-beta-hydroxylase as a marker. J. Comp. Neurol. 163, 467–506 (1976).
Aston-Jones, G. & Cohen, J. D. An integrative theory of locus coeruleus-norepinephrine function: adaptive gain and optimal performance. Annu. Rev. Neurosci. 28, 403–450 (2005).
Berridge, C. W. & Waterhouse, B. D. The locus coeruleus-noradrenergic system: modulation of behavioral state and state-dependent cognitive processes. Brain Res. Brain Res. Rev. 42, 33–84 (2003).
Valentino, R. J., Pavcovich, L. A. & Hirata, H. Evidence for corticotropin-releasing hormone projections from Barrington's nucleus to the periaqueductal gray region and dorsal motor nucleus of the vagus in the rat. J. Comp. Neurol. 363, 402–422 (1995).
Sakanaka, M., Shibasaki, T. & Lederes, K. Corticotropin-releasing factor-like immunoreactivity in the rat brain as revealed by a modified cobalt-glucose oxide-diaminobenzidene method. J. Comp. Neurol. 260, 256–298 (1987).
Vincent, S. R. & Satoh, K. Corticotropin-releasing factor (CRF) immunoreactivity in the dorsolateral pontine tegmentum: further studies on the micturition reflex system. Brain Res. 308, 387–391 (1984).
Vale, W., Spiess, J., Rivier, C. & Rivier, J. Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science 213, 1394–1397 (1981).
Owens, M. J. & Nemeroff, C. B. Physiology and pharmacology of corticotropin-releasing factor. Pharmacol. Rev. 43, 425–474 (1991).
Valentino, R. J., Miselis, R. R. & Pavcovich, L. A. Pontine regulation of pelvic viscera: Pharmacological target for pelvic visceral dysfunctions. Trends Pharmacol. Sci. 20, 253–266 (1999).
Noto, H., Roppolo, J. R., Steers, W. D. & De Groat, W. C. Excitatory and inhibitory influences on bladder activity elicited by electrical stimulation in the pontine micturition center in the rat. Brain Res. 492, 99–115 (1989).
Pavcovich, L. A. & Valentino, R. J. Central regulation of micturition in the rat by corticotropin-releasing hormone from Barrington's nucleus. Neurosci. Lett. 196, 185–188 (1995).
Willette, R. N., Morrison, S., Sapru, H. N. & Reis, D. J. Stimulation of opiate receptors in the dorsal pontine tegmentum inhibits reflex contraction of the urinary bladder. J. Pharmacol. Exp. Ther. 244, 403–409 (1988).
Barrington, F. J. T. The effect of lesion of the hind- and mid-brain on micturition in the cat. Quart. J. Exp. Physiol. 15, 81–102 (1925).
Hida, T. & Shimazu, N. The interrelation between the laterdorsal tegmental area and lumbosacral segments of rats as studied by HRP method. Arch. Histol. Jap. 45, 495–504 (1982).
Loewy, A. D., Saper, C. B. & Baker, R. P. Descending projections from the pontine micturition center. Brain Res. 172, 533–538 (1979).
Blok, B. F. M. & Holstege, G. Ultrastructural evidence for a direct pathway from the pontine micturition center to parasympathetic preganglionic motoneurons of the bladder of the cat. Neurosci. Lett. 222, 195–198 (1997).
Rouzade-Dominguez, M. L., Pernar, L., Beck, S. & Valentino, R. J. Convergent responses of Barrington's nucleus neurons to pelvic visceral stimuli: a juxtacellular labeling study. Eur. J. Neurosci. 18, 3325–3334 (2003).
Ding, Y. Q. et al. Direct projections from lumbosacral spinal cord to Barrington's nucleus in the rat: a special reference to micturition reflex. J. Comp. Neurol. 389, 149–160 (1997).
Blok, B. F. M., De Weerd, H. & Holstege, G. Ultrastructural evidence for a paucity of projections from the lumbosacral cord to the pontine micturition center or M-region in the cat: a new concept for the organization of the micturition reflex with the periaqueductal gray as a central relay. J. Comp. Neurol. 359, 300–309 (1995).
Rouzade-Dominguez, M. L., Miselis, R. & Valentino, R. J. Central representation of bladder and colon revealed by dual transsynaptic tracing: substrates for pelvic visceral coordination. Eur. J. Neurosci. 18, 3311–3324 (2003).
Marson, L. Central nervous system neurons identified after injection of pseudorabies virus into the rat clitoris. Neurosci. Lett. 190, 41–44 (1995).
Marson, L. & McKenna, K. E. CNS cell groups involved in the control of the ischiocavernosus and bulbospongiosus muscles; a transneuronal tracing study using pseudorabies virus. J. Comp. Neurol. 374, 161–179 (1996).
Vizzard, M. A., Brisson, M. & de Groat, W. C. Transneuronal labeling of neurons in the adult rat central nervous system following inoculation of pseudorabies virus into the colon. Cell Tissue Res. 299, 9–26 (2000).
Lambert, S. M. & Zderic, S. A. in Textbook of Female Urology and Urogynecology (eds Cardozo, L. & Staskin, D.) 172–184 (Informa Healthcare, UK, 2010).
Guo, Y. J. et al. Lower urinary tract symptoms in women with irritable bowel syndrome. Int. J. Urol. 17, 175–181 (2010).
Grzanna, R. & Molliver, M. E. The locus coeruleus in the rat: an immunohistochemical delineation. Neuroscience 5, 21–40 (1980).
Bouret, S. & Sara, S. J. Network reset: a simplified overarching theory of locus coeruleus noradrenaline function. Trends Neurosci. 28, 574–582 (2005).
Aston-Jones, G. & Bloom, F. E. Norepinephrine-containing locus coeruleus neurons in behaving rats exhibit pronounced responses to non-noxious environmental stimuli. J. Neurosci. 1, 887–900 (1981).
Elam, M., Thoren, T. & Svensson, T. H. Locus coeruleus neurons and sympathetic nerves: activation by visceral afferents. Brain Res. 375, 117–125 (1986).
Foote, S. L., Aston-Jones, G. & Bloom, F. E. Impulse activity of locus coeruleus neurons in awake rats and monkeys is a function of sensory stimulation and arousal. Proc. Natl Acad. Sci. USA 77, 3033–3037 (1980).
Lechner, S., Curtis, A., Brons, R. & Valentino, R. Locus coeruleus activation by colon distention: role of corticotropin-releasing factor and excitatory amino acids. Brain Res. 756, 114–124 (1997).
Page, M. E., Akaoka, H., Aston-Jones, G. & Valentino, R. J. Bladder distention activates locus coeruleus neurons by an excitatory amino acid mechanism. Neuroscience 51, 555–563 (1992).
Bale, T. L. & Vale, W. W. CRF and CRF receptors: role in stress responsivity and other behaviors. Annu. Rev. Pharmacol. Toxicol. 44, 525–557 (2004).
Valentino, R. J. & Van Bockstaele, E. Convergent regulation of locus coeruleus activity as an adaptive response to stress. Eur. J. Pharmacol. 583, 194–203 (2008).
Van Bockstaele, E. J., Colago, E. E. O. & Valentino, R. J. Corticotropin-releasing factor-containing axon terminals synapse onto catecholamine dendrites and may presynaptically modulate other afferents in the rostral pole of the nucleus locus coeruleus in the rat brain. J. Comp. Neurol. 364, 523–534 (1996).
Page, M. E., Berridge, C. W., Foote, S. L. & Valentino, R. J. Corticotropin-releasing factor in the locus coeruleus mediates EEG activation associated with hypotensive stress. Neurosci. Lett. 164, 81–84 (1993).
Curtis, A. L., Bello, N. T., Connally, K. R. & Valentino, R. J. Corticotropin-releasing factor neurons of the central nucleus of the amygdala mediate locus coeruleus activation by cardiovascular stress. J. Neuroendocrinol. 14, 667–682 (2002).
Valentino, R. J., Kosboth, M., Winogren, M. L. & Miselis, R. R. Transneuronal labeling from the rat distal colon: anatomical evidence for regulation of distal colon function by a pontine corticotropin-releasing factor system. J. Comp. Neurol. 417, 399–414 (2000).
Lovejoy, D. A. & Balment, R. J. Evolution and physiology of the corticotropin-releasing factor (CRF) family of neuropeptides in vertebrates. Gen. Comp. Endocrinol. 115, 1–22 (1999).
Denver, R. Structural and functional evolution of vertebrate neuroendocrine stress systems. Ann. NY Acad. Sci. 1163, 1–16 (2009).
Ruggiero, D. A., Underwood, M. D., Rice, P. M., Mann, J. J. & Arango, V. Corticotropin-releasing hormone and serotonin interact in the human brainstem: behavioral implications. Neuroscience 91, 1343–1354 (1999).
Blok, B. F. M., Willemsen, A. T. M. & Holstege, G. A PET study on brain control of micturition in humans. Brain 120, 111–121 (1997).
Rouzade-Dominguez, M. L., Curtis, A. L. & Valentino, R. J. Role of Barrington's nucleus in the activation of rat locus coeruleus neurons by colonic distension. Brain Res. 917, 206–218 (2001).
Pavcovich, L. A., Yang, M., Miselis, R. R. & Valentino, R. J. Novel role for the pontine micturition center, Barrington's nucleus: evidence for coordination of colonic and forebrain activity. Brain Res. 784, 355–361 (1998).
Klausner, A. P. & Steers, W. D. Corticotropin-releasing factor: a mediator of emotional influences on bladder function. J. Urol. 172, 2570–2573 (2004).
Klausner, A. P. et al. The role of corticotropin releasing factor and its antagonist, astressin, on micturition in the rat. Auton. Neurosci. 123, 26–35 (2005).
Kiddoo, D. A. et al. Impact of the state of arousal and stress neuropeptides on urodynamic function in the freely moving rat. Am. J. Physiol. Regul. Integr. Comp. Physiol. 290, R1697–R1706 (2006).
Kawatani, M., Suzuki, T. & de Groat, W. C. Corticotropin-releasing factor-like immunoreactivity in afferent projections to the sacral spinal cord of the cat. J. Auton. Nerv. Sys. 61, 218–226 (1996).
LaBerge, J., Malley, S. E., Girard, B., Corrow, K. & Vizzard, M. A. Postnatal expression of corticotropin releasing factor (CRF) in rat urinary bladder. Auton. Neurosci. 141, 83–93 (2008).
LaBerge, J., Malley, S. E., Zvarova, K. & Vizzard, M. A. Expression of corticotropin-releasing factor and CRF receptors in micturition pathways after cyclophosphamide-induced cystitis. Am. J. Physiol. Regul. Integr. Comp. Physiol. 291, R692–R703 (2006).
Saito, M., Longhurst, P. A., Tammela, T. L., Wein., A. J. & Levin, R. M. Effects of partial outlet obstruction of the rat urinary bladder on micturition characteristics, DNA synthesis and the contractile response to field stimulation and pharmacological agents. J. Urol. 150, 1045–1051 (1993).
Rickenbacher, E. et al. Impact of overactive bladder on the brain: central sequelae of a visceral pathology. Proc. Natl Acad. Sci. USA 105, 10589–10594 (2008).
de Groat, W. C. & Yoshimura, N. Changes in afferent activity after spinal cord injury. Neurourol. Urodyn. 29, 63–76 (2010).
Bland, B. H. & Oddie, S. D. Theta band oscillation and synchrony in the hippocampal formation and associated structures: the case for its role in sensorimotor integration. Behav. Brain Res. 127, 119–136 (2001).
Kirby, R. S. The natural history of benign prostatic hyperplasia: what have we learned in the last decade? Urology 56, 3–6 (2000).
Tubaro, A. Defining overactive bladder: epidemiology and burden of disease. Urology 64, 2–6 (2004).
Mefford, I. N. & Potter, W. Z. A neuroanatomical and biochemical basis for attention deficit disorder with hyperactivity in children: a defect in tonic adrenaline mediated inhibition of locus coeruleus stimulation. Med. Hypotheses 29, 33–42 (1989).
Konrad, K., Gauggel, S. & Schurek, J. Catecholamine functioning in children with traumatic brain injuries and children with attention-deficit/hyperactivity disorder. Brain Res. Cogn. Brain Res. 16, 425–433 (2003).
Pliszka, S. R., McCracken, J. T. & Maas, J. W. Catecholamines in attention-deficit hyperactivity disorder: current perspectives. J. Am. Acad. Child. Adolesc. Psychiatry 35, 264–272 (1996).
Shreeram, S., He, J. P., Kalaydjian, A., Brothers, S. & Merikangas, K. R. Prevalence of enuresis and its association with attention-deficit/hyperactivity disorder among U. S. children: results from a nationally representative study. J. Am. Acad. Child. Adolesc. Psychiatry 48, 35–41 (2009).
Griffiths, D. Clinical studies of cerebral and urinary tract function in elderly people with urinary incontinence. Behav. Brain Res. 92, 151–155 (1998).
Rovner, E. S. & Wein, A. J. Incidence and prevalence of overactive bladder. Curr. Urol. Rep. 3, 434–438 (2002).
Henry, J. P., Meehan, W. P. & Stephens, P. M. Role of subordination in nephritis of socially stressed mice. Contr. Nephrol. 30, 38–42 (1982).
Lumley, L. A., Sipos, M. L., Charles, R. C., Charles, R. F. & Meyerhoff, J. L. Social stress effects on territorial marking and ultrasonic vocalizations in mice. Physiol. Behav. 67, 769–775 (1999).
Hoshaw, B. A., Evans, J. C., Mueller, B., Valentino, R. J. & Lucki, I. Social competition in rats: cell proliferation and behavior. Behav. Brain Res. 175, 343–351 (2006).
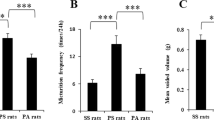
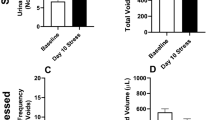
Wood, S. K., Baez, M. A., Bhatnagar, S. & Valentino, R. J. Social stress-induced bladder dysfunction: potential role of corticotropin-releasing factor. Am. J. Physiol. Regul. Integr. Comp. Physiol. 296, R1671–R1678 (2009).
Carrive, P. The periaqueductal gray and defensive behavior: functional representation and neuronal organization. Behav. Brain Res. 58, 27–47 (1993).
Blok, B. F. M. & Holstege, G. Direct projections from the periaqueductal gray to the pontine micturition center (M-region). An anterogreade and retrograde tracing study in the cat. Neurosci. Lett. 166, 93–96 (1994).
Chang, A. et al. Social stress in mice induces voiding dysfunction and bladder wall remodeling. Am. J. Physiol. Renal Physiol. 297, F1101–F1108 (2009).
Reyes, T. M., Walker, J. R., DeCino, C., Hogenesch, J. B. & Sawchenko, P. E. Categorically distinct acute stressors elicit dissimilar transcriptional profiles in the paraventricular nucleus of the hypothalamus. J. Neurosci. 23, 5607–5616 (2003).
Sawchenko, P. E., Li, H. Y. & Ericsson, A. Circuits and mechanisms governing hypothalamic responses to stress: a tale of two paradigms. Prog. Brain Res. 122, 61–78 (2000).
Keay, K. A. & Bandler, R. Parallel circuits mediating distinct emotional coping reactions to different types of stress. Neurosci. Biobehav. Rev. 25, 669–678 (2001).
Abarbanel, J. & Marcus, E. L. Impaired detrusor contractility in community-dwelling elderly presenting with lower urinary tract symptoms. Urology 69, 436–440 (2007).
Varlam, D. E. & Dippell, J. Non-neurogenic bladder and chronic renal insufficiency in childhood. Pediatr. Nephrol. 9, 1–5 (1995).
Fenster, H. & Patterson, B. Urinary retention in sexually abused women. Can. J. Urol. 2, 185–188 (1995).
Bilanakis, N. Psychogenic urinary retention. Gen. Hosp. Psychiatry 28, 259–261 (2006).
Groutz, A. & Blaivas, J. G. Non-neurogenic female voiding dysfunction. Curr. Opin. Urol. 12, 311–316 (2002).
Feldman, A. S. & Bauer, S. B. Diagnosis and management of dysfunctional voiding. Curr. Opin. Pediatr. 18, 139–147 (2006).
Bauer, S. B. Special considerations of the overactive bladder in children. Urology 60, 43–49 (2002).
Ellsworth, P. I., Merguerian, P. A. & Copening, M. E. Sexual abuse: another causative factor in dysfunctional voiding. J. Urol. 153, 773–776 (1995).
Lettgen, B. et al. Urge incontinence and voiding postponement in children: somatic and psychosocial factors. Acta Paediatr. 91, 978–984 (2002).
Lettgen, B. et al. Urge incontinence and voiding postponement in children: somatic and psychosocial factors (discussion). Acta Paediatr. 91, 895–896 (2002).
Davila, G. W., Bernier, F., Franco, J. & Kopka, S. L. Bladder dysfunction in sexual abuse survivors. J. Urol. 170, 476–479 (2003).
Romans, S., Belaise, C., Martin, J., Morris, E. & Raffi, A. Childhood abuse and later medical disorders in women. An epidemiological study. Psychother. Psychosom. 71, 141–150 (2002).
Franzen, K., Johansson, J. E., Andersson, G., Pettersson, N. & Nilsson, K. Urinary incontinence in women is not exclusively a medical problem: a population-based study on urinary incontinence and general living conditions. Scand. J. Urol. Nephrol. 43, 226–232 (2009).
Acknowledgements
The authors acknowledge funding provided by an National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) George O'Brien Center Grant (PO5-DK052620-13) that was essential in facilitating discussions and collaborations between the neuroscientists and urologists involved in this Review.
Author information
Authors and Affiliations
Contributions
R. J. Valentino, S. K. Wood and S. A. Zderic researched data for the article, and R. J. Valentino wrote the article. All authors provided a substantial contribution to discussions of the content, and reviewed and edited the article before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Valentino, R., Wood, S., Wein, A. et al. The bladder–brain connection: putative role of corticotropin-releasing factor. Nat Rev Urol 8, 19–28 (2011). https://doi.org/10.1038/nrurol.2010.203
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2010.203
This article is cited by
-
Functional constipation induces bladder overactivity associated with upregulations of Htr2 and Trpv2 pathways
Scientific Reports (2021)
-
Blebbistatin, a Myosin II Inhibitor, Exerts Antidepressant-Like Activity and Suppresses Detrusor Overactivity in an Animal Model of Depression Coexisting with Overactive Bladder
Neurotoxicity Research (2019)
-
Voluntary urination control by brainstem neurons that relax the urethral sphincter
Nature Neuroscience (2018)
-
A corticopontine circuit for initiation of urination
Nature Neuroscience (2018)
-
Functional urological disorders: a sensitized defence response in the bladder–gut–brain axis
Nature Reviews Urology (2017)