Key Points
-
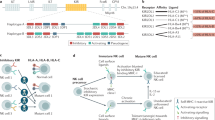
A family of molecules, including CD226, T cell immunoreceptor with immunoglobulin and ITIM domains (TIGIT) and CD96, that bind nectin and nectin-like proteins has recently emerged as important regulators of natural killer (NK) cell functions.
-
This family of molecules regulates NK cell adhesion and cytotoxicity, immune synapse formation, cytokine secretion and crosstalk with dendritic cells.
-
This family of molecules has important roles in the pathophysiological processes of cancer, autoimmunity and viral infection.
-
The signalling properties of CD226, TIGIT and CD96 remain poorly characterized. Future work should discriminate the relative role of TIGIT and CD96 as negative regulators of CD226 activation in human and mouse immune responses.
-
Other well-defined paired receptors, such as the killer immunoglobulin-like receptor family and C-type lectin-like CD94–NKG2 receptor family molecules, control NK cell functions.
-
CD226, TIGIT and CD96 are crucial regulators of lymphocyte-mediated effector functions against tumours and may be promising new therapeutic targets for the treatment of malignancies.
Abstract
Natural killer (NK) cells are innate lymphocytes that are crucial for the control of infections and malignancies. NK cells express a variety of inhibitory and activating receptors that facilitate fine discrimination between damaged and healthy cells. Among them, a family of molecules that bind nectin and nectin-like proteins has recently emerged and has been shown to function as an important regulator of NK cell functions. These molecules include CD226, T cell immunoreceptor with immunoglobulin and ITIM domains (TIGIT), CD96, and cytotoxic and regulatory T cell molecule (CRTAM). In this Review, we focus on the recent advances in our understanding of how these receptors regulate NK cell biology and of their roles in pathologies such as cancer, infection and autoimmunity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Vivier, E., Tomasello, E., Baratin, M., Walzer, T. & Ugolini, S. Functions of natural killer cells. Nature Immunol. 9, 503–510 (2008).
Long, E. O., Kim, H. S., Liu, D., Peterson, M. E. & Rajagopalan, S. Controlling natural killer cell responses: integration of signals for activation and inhibition. Annu. Rev. Immunol. 31, 227–258 (2013). This is an outstanding and comprehensive review on the control of NK cell functions through activating and inhibitory receptors.
Karre, K. Natural killer cell recognition of missing self. Nature Immunol. 9, 477–480 (2008).
Lanier, L. L. Up on the tightrope: natural killer cell activation and inhibition. Nature Immunol. 9, 495–502 (2008).
Chan, C. J. et al. The receptors CD96 and CD226 oppose each other in the regulation of natural killer cell functions. Nature Immunol. 15, 431–438 (2014). This is the first paper showing a crucial role for CD96 in limiting NK cell functions in vivo using Cd96−/− mice.
Stanietsky, N. et al. The interaction of TIGIT with PVR and PVRL2 inhibits human NK cell cytotoxicity. Proc. Natl Acad. Sci. USA 106, 17858–17863 (2009). This study shows a potential role for TIGIT in limiting human NK cell cytotoxicity.
Gilfillan, S. et al. DNAM-1 promotes activation of cytotoxic lymphocytes by nonprofessional antigen-presenting cells and tumors. J. Exp. Med. 205, 2965–2973 (2008).
Shibuya, A. et al. DNAM-1, a novel adhesion molecule involved in the cytolytic function of T lymphocytes. Immunity 4, 573–581 (1996).
Bottino, C. et al. Identification of PVR (CD155) and Nectin-2 (CD112) as cell surface ligands for the human DNAM-1 (CD226) activating molecule. J. Exp. Med. 198, 557–567 (2003). References 8 and 9 report that CD226 engagement by its ligands, CD112 and CD155, on target cells stimulates NK cell cytolytic functions.
Sakisaka, T. & Takai, Y. Biology and pathology of nectins and nectin-like molecules. Curr. Opin. Cell Biol. 16, 513–521 (2004).
Chan, C. J., Smyth, M. J. & Martinet, L. Molecular mechanisms of natural killer cell activation in response to cellular stress. Cell Death Differ. 21, 5–14 (2014).
Bernhardt, G. TACTILE becomes tangible: CD96 discloses its inhibitory peculiarities. Nature Immunol. 15, 406–408 (2014).
Minton, K. Natural killer cells: a TACTILE restraint. Nature Rev. Immunol. 14, 285 (2014).
Yu, X. et al. The surface protein TIGIT suppresses T cell activation by promoting the generation of mature immunoregulatory dendritic cells. Nature Immunol. 10, 48–57 (2009). This is the first identification of TIGIT and its inhibitory functions through its interaction with CD155.
Seth, S. et al. Heterogeneous expression of the adhesion receptor CD226 on murine NK and T cells and its function in NK-mediated killing of immature dendritic cells. J. Leukoc. Biol. 86, 91–101 (2009).
Tahara-Hanaoka, S. et al. Functional characterization of DNAM-1 (CD226) interaction with its ligands PVR (CD155) and nectin-2 (PRR-2/CD112). Int. Immunol. 16, 533–538 (2004).
Liu, J. et al. Crystal structure of cell adhesion molecule nectin-2/CD112 and its binding to immune receptor DNAM-1/CD226. J. Immunol. 188, 5511–5520 (2012).
Hou, S. et al. CD226 protein is involved in immune synapse formation and triggers natural killer (NK) cell activation via its first extracellular domain. J. Biol. Chem. 289, 6969–6977 (2014).
Tahara-Hanaoka, S. et al. Tumor rejection by the poliovirus receptor family ligands of the DNAM-1 (CD226) receptor. Blood 107, 1491–1496 (2006).
Stanietsky, N. et al. Mouse TIGIT inhibits NK-cell cytotoxicity upon interaction with PVR. Eur. J. Immunol. 43, 2138–2150 (2013).
Boles, K. S. et al. A novel molecular interaction for the adhesion of follicular CD4 T cells to follicular DC. Eur. J. Immunol. 39, 695–703 (2009).
Levin, S. D. et al. Vstm3 is a member of the CD28 family and an important modulator of T-cell function. Eur. J. Immunol. 41, 902–915 (2011).
Georgiev, H. et al. To the Editor: TIGIT versus CD226: hegemony or coexistence? Eur. J. Immunol. 44, 307–308 (2014).
Johnston, R. J. et al. The immunoreceptor TIGIT regulates antitumor and antiviral CD8+ T cell effector function. Cancer Cell 26, 923–937 (2014). This study shows that TIGIT is expressed by PD1+ exhausted tumour-infiltrating T cells and that targeting these receptors with monoclonal antibodies represents a promising strategy to restore CD8+ T cell functions in cancer or in chronic infectious disease.
Wang, P. L., O'Farrell, S., Clayberger, C. & Krensky, A. M. Identification and molecular cloning of tactile. A novel human T cell activation antigen that is a member of the Ig gene superfamily. J. Immunol. 148, 2600–2608 (1992).
Meyer, D. et al. CD96 interaction with CD155 via its first Ig-like domain is modulated by alternative splicing or mutations in distal Ig-like domains. J. Biol. Chem. 284, 2235–2244 (2009).
Seth, S. et al. The murine pan T cell marker CD96 is an adhesion receptor for CD155 and nectin-1. Biochem. Biophys. Res. Commun. 364, 959–965 (2007).
de Andrade, L. F., Smyth, M. J. & Martinet, L. DNAM-1 control of natural killer cells functions through nectin and nectin-like proteins. Immunol. Cell Biol. 92, 237–244 (2014).
Fuchs, A., Cella, M., Giurisato, E., Shaw, A. S. & Colonna, M. Cutting edge: CD96 (tactile) promotes NK cell-target cell adhesion by interacting with the poliovirus receptor (CD155). J. Immunol. 172, 3994–3998 (2004).
Stengel, K. F. et al. Structure of TIGIT immunoreceptor bound to poliovirus receptor reveals a cell-cell adhesion and signaling mechanism that requires cis-trans receptor clustering. Proc. Natl Acad. Sci. USA 109, 5399–5404 (2012).
Liu, S. et al. Recruitment of Grb2 and SHIP1 by the ITT-like motif of TIGIT suppresses granule polarization and cytotoxicity of NK cells. Cell Death Differ. 20, 456–464 (2013).
Orange, J. S. Formation and function of the lytic NK-cell immunological synapse. Nature Rev. Immunol. 8, 713–725 (2008).
Ramsbottom, K. M. et al. Cutting edge: DNAX accessory molecule 1-deficient CD8+ T cells display immunological synapse defects that impair antitumor immunity. J. Immunol. 192, 553–557 (2014).
Lagrue, K. et al. The central role of the cytoskeleton in mechanisms and functions of the NK cell immune synapse. Immunol. Rev. 256, 203–221 (2013).
Gross, C. C., Brzostowski, J. A., Liu, D. & Long, E. O. Tethering of intercellular adhesion molecule on target cells is required for LFA-1-dependent NK cell adhesion and granule polarization. J. Immunol. 185, 2918–2926 (2010).
Vyas, Y. M. et al. Spatial organization of signal transduction molecules in the NK cell immune synapses during MHC class I-regulated noncytolytic and cytolytic interactions. J. Immunol. 167, 4358–4367 (2001).
Shibuya, K. et al. CD226 (DNAM-1) is involved in lymphocyte function-associated antigen 1 costimulatory signal for naive T cell differentiation and proliferation. J. Exp. Med. 198, 1829–1839 (2003).
Ralston, K. J. et al. The LFA-1-associated molecule PTA-1 (CD226) on T cells forms a dynamic molecular complex with protein 4.1G and human discs large. J. Biol. Chem. 279, 33816–33828 (2004).
Shibuya, K. et al. Physical and functional association of LFA-1 with DNAM-1 adhesion molecule. Immunity 11, 615–623 (1999).
Bryceson, Y. T., Ljunggren, H. G. & Long, E. O. Minimal requirement for induction of natural cytotoxicity and intersection of activation signals by inhibitory receptors. Blood 114, 2657–2666 (2009).
Lozano, E., Dominguez-Villar, M., Kuchroo, V. & Hafler, D. A. The TIGIT/CD226 axis regulates human T cell function. J. Immunol. 188, 3869–3875 (2012).
Lozano, E., Joller, N., Cao, Y., Kuchroo, V. K. & Hafler, D. A. The CD226/CD155 interaction regulates the proinflammatory (TH1/TH17)/anti-inflammatory (TH2) balance in humans. J. Immunol. 191, 3673–3680 (2013).
Bi, J. et al. TIGIT safeguards liver regeneration through regulating NK cell-hepatocyte crosstalk. Hepatology 60, 1389–1398 (2014).
Li, M. et al. T-cell immunoglobulin and ITIM domain (TIGIT) receptor/poliovirus receptor (PVR) ligand engagement suppresses interferon-γ production of natural killer cells via β-arrestin 2-mediated negative signaling. J. Biol. Chem. 289, 17647–17657 (2014).
Pende, D. et al. Expression of the DNAM-1 ligands, Nectin-2 (CD112) and poliovirus receptor (CD155), on dendritic cells: relevance for natural killer-dendritic cell interaction. Blood 107, 2030–2036 (2006).
Maier, M. K. et al. The adhesion receptor CD155 determines the magnitude of humoral immune responses against orally ingested antigens. Eur. J. Immunol. 37, 2214–2225 (2007).
Kamran, N. et al. Toll-like receptor ligands induce expression of the costimulatory molecule CD155 on antigen-presenting cells. PLoS ONE 8, e54406 (2013).
Oda, T., Ohka, S. & Nomoto, A. Ligand stimulation of CD155alpha inhibits cell adhesion and enhances cell migration in fibroblasts. Biochem. Biophys. Res. Commun. 319, 1253–1264 (2004).
Sato, T. et al. Common signaling pathway is used by the trans-interaction of Necl-5/Tage4/PVR/CD155 and nectin, and of nectin and nectin during the formation of cell-cell adhesion. Cancer Sci. 96, 578–589 (2005).
Walzer, T., Dalod, M., Robbins, S. H., Zitvogel, L. & Vivier, E. Natural-killer cells and dendritic cells: “l'union fait la force”. Blood 106, 2252–2258 (2005).
Morandi, B. et al. Dendritic cell editing by activated natural killer cells results in a more protective cancer-specific immune response. PLoS ONE 7, e39170 (2012).
Zingoni, A., Ardolino, M., Santoni, A. & Cerboni, C. NKG2D and DNAM-1 activating receptors and their ligands in NK-T cell interactions: role in the NK cell-mediated negative regulation of T cell responses. Frontiers Immunol. 3, 408 (2012).
Ardolino, M. et al. DNAM-1 ligand expression on Ag-stimulated T lymphocytes is mediated by ROS-dependent activation of DNA-damage response: relevance for NK-T cell interaction. Blood 117, 4778–4786 (2011).
O'Leary, J. G., Goodarzi, M., Drayton, D. L. & von Andrian, U. H. T cell- and B cell-independent adaptive immunity mediated by natural killer cells. Nature Immunol. 7, 507–516 (2006).
Sun, J. C., Beilke, J. N. & Lanier, L. L. Adaptive immune features of natural killer cells. Nature 457, 557–561 (2009).
Paust, S., Senman, B. & von Andrian, U. H. Adaptive immune responses mediated by natural killer cells. Immunol. Rev. 235, 286–296 (2010).
Foley, B. et al. Cytomegalovirus reactivation after allogeneic transplantation promotes a lasting increase in educated NKG2C+ natural killer cells with potent function. Blood 119, 2665–2674 (2012).
Lopez-Verges, S. et al. Expansion of a unique CD57+NKG2Chi natural killer cell subset during acute human cytomegalovirus infection. Proc. Natl Acad. Sci. USA 108, 14725–14732 (2011).
Nabekura, T. et al. Costimulatory molecule DNAM-1 is essential for optimal differentiation of memory natural killer cells during mouse cytomegalovirus infection. Immunity 40, 225–234 (2014).
Shibuya, A., Lanier, L. L. & Phillips, J. H. Protein kinase C is involved in the regulation of both signaling and adhesion mediated by DNAX accessory molecule-1 receptor. J. Immunol. 161, 1671–1676 (1998).
Bryceson, Y. T., March, M. E., Ljunggren, H. G. & Long, E. O. Synergy among receptors on resting NK cells for the activation of natural cytotoxicity and cytokine secretion. Blood 107, 159–166 (2006).
Kim, H. S., Das, A., Gross, C. C., Bryceson, Y. T. & Long, E. O. Synergistic signals for natural cytotoxicity are required to overcome inhibition by c-Cbl ubiquitin ligase. Immunity 32, 175–186 (2010).
Kim, H. S. & Long, E. O. Complementary phosphorylation sites in the adaptor protein SLP-76 promote synergistic activation of natural killer cells. Sci. Signal. 5, ra49 (2012).
Rozsnyay, Z. Signaling complex formation of CD44 with src-related kinases. Immunol. Lett. 68, 101–108 (1999).
Chambers, C. A. The expanding world of co-stimulation: the two-signal model revisited. Trends Immunol. 22, 217–223 (2001).
Soriani, A. et al. ATM-ATR-dependent up-regulation of DNAM-1 and NKG2D ligands on multiple myeloma cells by therapeutic agents results in enhanced NK-cell susceptibility and is associated with a senescent phenotype. Blood 113, 3503–3511 (2009).
Carlsten, M. et al. Primary human tumor cells expressing CD155 impair tumor targeting by down-regulating DNAM-1 on NK cells. J. Immunol. 183, 4921–4930 (2009).
El-Sherbiny, Y. M. et al. The requirement for DNAM-1, NKG2D, and NKp46 in the natural killer cell-mediated killing of myeloma cells. Cancer Res. 67, 8444–8449 (2007).
Lakshmikanth, T. et al. NCRs and DNAM-1 mediate NK cell recognition and lysis of human and mouse melanoma cell lines in vitro and in vivo. J. Clin. Invest. 119, 1251–1263 (2009).
Sanchez-Correa, B. et al. Decreased expression of DNAM-1 on NK cells from acute myeloid leukemia patients. Immunol. Cell Biol. 90, 109–115 (2012).
Mamessier, E. et al. Human breast cancer cells enhance self tolerance by promoting evasion from NK cell antitumor immunity. J. Clin. Invest. 121, 3609–3622 (2011).
Oshima, T. et al. Nectin-2 is a potential target for antibody therapy of breast and ovarian cancers. Mol. Cancer 12, 60 (2013).
Iguchi-Manaka, A. et al. Accelerated tumor growth in mice deficient in DNAM-1 receptor. J. Exp. Med. 205, 2959–2964 (2008). This paper, together with reference 7, was the first to show the role of CD226 in NK cell- and CD8+ T cell-mediated tumour immunosurveillance using Cd226−/− mice.
Croxford, J. L. et al. ATM-dependent spontaneous regression of early Emu-myc-induced murine B cell leukemia depends on NK and T cells. Blood 121, 2512–2521 (2013).
Chan, C. J. et al. DNAM-1/CD155 interactions promote cytokine and NK cell-mediated suppression of poorly immunogenic melanoma metastases. J. Immunol. 184, 902–911 (2010).
Smyth, M. J., Crowe, N. Y. & Godfrey, D. I. NK cells and NKT cells collaborate in host protection from methylcholanthrene-induced fibrosarcoma. Int. Immunol. 13, 459–463 (2001).
Welch, M. J., Teijaro, J. R., Lewicki, H. A., Colonna, M. & Oldstone, M. B. CD8 T cell defect of TNFα and IL-2 in DNAM-1 deficient mice delays clearance in vivo of a persistent virus infection. Virology 429, 163–170 (2012).
Magri, G. et al. NKp46 and DNAM-1 NK-cell receptors drive the response to human cytomegalovirus-infected myeloid dendritic cells overcoming viral immune evasion strategies. Blood 117, 848–856 (2011).
Prod'homme, V. et al. Human cytomegalovirus UL141 promotes efficient downregulation of the natural killer cell activating ligand CD112. J. Gen. Virol. 91, 2034–2039 (2010).
Tomasec, P. et al. Downregulation of natural killer cell-activating ligand CD155 by human cytomegalovirus UL141. Nature Immunol. 6, 181–188 (2005).
Matusali, G., Potesta, M., Santoni, A., Cerboni, C. & Doria, M. The human immunodeficiency virus type 1 Nef and Vpu proteins downregulate the natural killer cell-activating ligand PVR. J. Virol. 86, 4496–4504 (2012). References 79–81 show that the downregulation of CD112 and CD155 expression is a common evasion strategy developed by viruses to avoid CD226-mediated NK cell recognition.
Todd, J. A. et al. Robust associations of four new chromosome regions from genome-wide analyses of type 1 diabetes. Nature Genet. 39, 857–864 (2007).
Song, G., Bae, S. C., Choi, S., Ji, J. & Lee, Y. Association between the CD226 rs763361 polymorphism and susceptibility to autoimmune diseases: a meta-analysis. Lupus 21, 1522–1530 (2012).
Du, Y. et al. Association of the CD226 single nucleotide polymorphism with systemic lupus erythematosus in the Chinese Han population. Tissue Antigens 77, 65–67 (2011).
Maiti, A. K. et al. Non-synonymous variant (Gly307Ser) in CD226 is associated with susceptibility to multiple autoimmune diseases. Rheumatology 49, 1239–1244 (2010).
Wieczorek, S. et al. Novel association of the CD226 (DNAM-1) Gly307Ser polymorphism in Wegener's granulomatosis and confirmation for multiple sclerosis in German patients. Genes Immun. 10, 591–595 (2009).
Flodstrom-Tullberg, M., Bryceson, Y. T., Shi, F. D., Hoglund, P. & Ljunggren, H. G. Natural killer cells in human autoimmunity. Curr. Opin. Immunol. 21, 634–640 (2009).
Parham, P. MHC class I molecules and KIRs in human history, health and survival. Nature Rev. Immunol. 5, 201–214 (2005).
Thielens, A., Vivier, E. & Romagne, F. NK cell MHC class I specific receptors (KIR): from biology to clinical intervention. Curr. Opin. Immunol. 24, 239–245 (2012).
Raulet, D. H. & Vance, R. E. Self-tolerance of natural killer cells. Nature Rev. Immunol. 6, 520–531 (2006).
Huse, M., Catherine Milanoski, S. & Abeyweera, T. P. Building tolerance by dismantling synapses: inhibitory receptor signaling in natural killer cells. Immunol. Rev. 251, 143–153 (2013).
Stebbins, C. C. et al. Vav1 dephosphorylation by the tyrosine phosphatase SHP-1 as a mechanism for inhibition of cellular cytotoxicity. Mol. Cell. Biol. 23, 6291–6299 (2003).
Abeyweera, T. P., Merino, E. & Huse, M. Inhibitory signaling blocks activating receptor clustering and induces cytoskeletal retraction in natural killer cells. J. Cell Biol. 192, 675–690 (2011).
Peterson, M. E. & Long, E. O. Inhibitory receptor signaling via tyrosine phosphorylation of the adaptor Crk. Immunity 29, 578–588 (2008).
Stewart, C. A. et al. Recognition of peptide-MHC class I complexes by activating killer immunoglobulin-like receptors. Proc. Natl Acad. Sci. USA 102, 13224–13229 (2005).
Sivori, S. et al. Natural killer cells expressing the KIR2DS1-activating receptor efficiently kill T-cell blasts and dendritic cells: implications in haploidentical HSCT. Blood 117, 4284–4292 (2011).
Graef, T. et al. KIR2DS4 is a product of gene conversion with KIR3DL2 that introduced specificity for HLA-A*11 while diminishing avidity for HLA-C. J. Exp. Med. 206, 2557–2572 (2009).
Rajagopalan, S. & Long, E. O. Understanding how combinations of HLA and KIR genes influence disease. J. Exp. Med. 201, 1025–1029 (2005).
Khakoo, S. I. et al. HLA and NK cell inhibitory receptor genes in resolving hepatitis C virus infection. Science 305, 872–874 (2004). This study suggests that inhibitory NK cell interactions are important in determining antiviral immunity and that diminished inhibitory responses confer protection against viral infections.
Alter, G. et al. Differential natural killer cell-mediated inhibition of HIV-1 replication based on distinct KIR/HLA subtypes. J. Exp. Med. 204, 3027–3036 (2007).
Carr, W. H. et al. Cutting edge: KIR3DS1, a gene implicated in resistance to progression to AIDS, encodes a DAP12-associated receptor expressed on NK cells that triggers NK cell activation. J. Immunol. 178, 647–651 (2007).
Malnati, M. S. et al. Peptide specificity in the recognition of MHC class I by natural killer cell clones. Science 267, 1016–1018 (1995).
Zappacosta, F., Borrego, F., Brooks, A. G., Parker, K. C. & Coligan, J. E. Peptides isolated from HLA-Cw*0304 confer different degrees of protection from natural killer cell-mediated lysis. Proc. Natl Acad. Sci. USA 94, 6313–6318 (1997).
Fadda, L. et al. Peptide antagonism as a mechanism for NK cell activation. Proc. Natl Acad. Sci. USA 107, 10160–10165 (2010).
Rajagopalan, S. & Long, E. O. Antagonizing inhibition gets NK cells going. Proc. Natl Acad. Sci. USA 107, 10333–10334 (2010).
Alter, G. & Altfeld, M. NK cells in HIV-1 infection: evidence for their role in the control of HIV-1 infection. J. Internal Med. 265, 29–42 (2009).
Katz, G. et al. MHC class I-independent recognition of NK-activating receptor KIR2DS4. J. Immunol. 173, 1819–1825 (2004).
Brooks, A. G., Posch, P. E., Scorzelli, C. J., Borrego, F. & Coligan, J. E. NKG2A complexed with CD94 defines a novel inhibitory natural killer cell receptor. J. Exp. Med. 185, 795–800 (1997).
Borrego, F., Ulbrecht, M., Weiss, E. H., Coligan, J. E. & Brooks, A. G. Recognition of human histocompatibility leukocyte antigen (HLA)-E complexed with HLA class I signal sequence-derived peptides by CD94/NKG2 confers protection from natural killer cell-mediated lysis. J. Exp. Med. 187, 813–818 (1998).
Carretero, M. et al. Specific engagement of the CD94/NKG2-A killer inhibitory receptor by the HLA-E class Ib molecule induces SHP-1 phosphatase recruitment to tyrosine-phosphorylated NKG2-A: evidence for receptor function in heterologous transfectants. Eur. J. Immunol. 28, 1280–1291 (1998).
Lanier, L. L., Corliss, B., Wu, J. & Phillips, J. H. Association of DAP12 with activating CD94/NKG2C NK cell receptors. Immunity 8, 693–701 (1998).
Braud, V. M. et al. HLA-E binds to natural killer cell receptors CD94/NKG2A, B and C. Nature 391, 795–799 (1998).
Tomasec, P. et al. Surface expression of HLA-E, an inhibitor of natural killer cells, enhanced by human cytomegalovirus gpUL40. Science 287, 1031 (2000).
Bossard, C. et al. HLA-E/beta2 microglobulin overexpression in colorectal cancer is associated with recruitment of inhibitory immune cells and tumor progression. Int. J. Cancer 131, 855–863 (2012).
Kraemer, T., Blasczyk, R. & Bade-Doeding, C. HLA-E: a novel player for histocompatibility. J. Immunol. Res. 2014, 352160 (2014).
Vance, R. E., Kraft, J. R., Altman, J. D., Jensen, P. E. & Raulet, D. H. Mouse CD94/NKG2A is a natural killer cell receptor for the nonclassical major histocompatibility complex (MHC) class I molecule Qa-1(b). J. Exp. Med. 188, 1841–1848 (1998).
Orr, M. T. et al. Development and function of CD94-deficient natural killer cells. PLoS ONE 5, e15184 (2010).
Fang, M. et al. CD94 is essential for NK cell-mediated resistance to a lethal viral disease. Immunity 34, 579–589 (2011). This study using CD94-deficient mice shows that the activating receptor formed by CD94 and NKG2E is essential for the resistance of C57BL/6 mice to mousepox.
Guma, M. et al. Imprint of human cytomegalovirus infection on the NK cell receptor repertoire. Blood 104, 3664–3671 (2004).
Beziat, V. et al. CMV drives clonal expansion of NKG2C+ NK cells expressing self-specific KIRs in chronic hepatitis patients. Eur. J. Immunol. 42, 447–457 (2012).
Guma, M. et al. Expansion of CD94/NKG2C+ NK cells in response to human cytomegalovirus-infected fibroblasts. Blood 107, 3624–3631 (2006).
Lopez-Botet, M., Muntasell, A. & Vilches, C. The CD94/NKG2C+ NK-cell subset on the edge of innate and adaptive immunity to human cytomegalovirus infection. Semin. Immunol. 26, 145–151 (2014).
Kim, S. et al. Licensing of natural killer cells by host major histocompatibility complex class I molecules. Nature 436, 709–713 (2005).
Anfossi, N. et al. Human NK cell education by inhibitory receptors for MHC class I. Immunity 25, 331–342 (2006).
Viant, C. et al. SHP-1-mediated inhibitory signals promote responsiveness and anti-tumour functions of natural killer cells. Nature Commun. 5, 5108 (2014).
Elliott, J. M. & Yokoyama, W. M. Unifying concepts of MHC-dependent natural killer cell education. Trends Immunol. 32, 364–372 (2011).
Tripathy, S. K. et al. Continuous engagement of a self-specific activation receptor induces NK cell tolerance. J. Exp. Med. 205, 1829–1841 (2008).
Pradeu, T., Jaeger, S. & Vivier, E. The speed of change: towards a discontinuity theory of immunity? Nature Rev. Immunol. 13, 764–769 (2013). This is an outstanding review on the formulation of a new immune paradigm 'the discontinuity theory'.
Seth, S. et al. Intranodal interaction with dendritic cells dynamically regulates surface expression of the co-stimulatory receptor CD226 protein on murine T cells. J. Biol. Chem. 286, 39153–39163 (2011).
Callahan, M. K. & Wolchok, J. D. At the Bedside: CTLA-4- and PD-1-blocking antibodies in cancer immunotherapy. J. Leukoc. Biol. 94, 41–53 (2013).
Koyama, M. et al. Promoting regulation via the inhibition of DNAM-1 after transplantation. Blood 121, 3511–3520 (2013).
Takai, Y., Miyoshi, J., Ikeda, W. & Ogita, H. Nectins and nectin-like molecules: roles in contact inhibition of cell movement and proliferation. Nature Rev. Mol. Cell Biol. 9, 603–615 (2008).
Erickson, B. M., Thompson, N. L. & Hixson, D. C. Tightly regulated induction of the adhesion molecule necl-5/CD155 during rat liver regeneration and acute liver injury. Hepatology 43, 325–334 (2006).
Hirota, T., Irie, K., Okamoto, R., Ikeda, W. & Takai, Y. Transcriptional activation of the mouse Necl-5/Tage4/PVR/CD155 gene by fibroblast growth factor or oncogenic Ras through the Raf–MEK–ERK–AP-1 pathway. Oncogene 24, 2229–2235 (2005).
Sloan, K. E. et al. CD155/PVR plays a key role in cell motility during tumor cell invasion and migration. BMC Cancer 4, 73 (2004).
Tane, S. et al. The role of Necl-5 in the invasive activity of lung adenocarcinoma. Exp. Mol. Pathol. 94, 330–335 (2013).
Nakai, R. et al. Overexpression of Necl-5 correlates with unfavorable prognosis in patients with lung adenocarcinoma. Cancer Sci. 101, 1326–1330 (2010).
Morimoto, K. et al. Interaction of cancer cells with platelets mediated by Necl-5/poliovirus receptor enhances cancer cell metastasis to the lungs. Oncogene 27, 264–273 (2008).
Vassena, L., Giuliani, E., Matusali, G., Cohen, E. & Doria, M. The human immunodeficiency virus type 1 Vpr protein upregulates PVR via activation of the ATR-mediated DNA damage response pathway. J. General Virol. 94, 2664–2669 (2013).
Cerboni, C. et al. The DNA damage response: a common pathway in the regulation of NKG2D and DNAM-1 ligand expression in normal, infected, and cancer cells. Front. Immunol. 4, 508 (2014).
Kennedy, J. et al. A molecular analysis of NKT cells: identification of a class-I restricted T cell-associated molecule (CRTAM). J. Leukoc. Biol. 67, 725–734 (2000).
Arase, N. et al. Heterotypic interaction of CRTAM with Necl2 induces cell adhesion on activated NK cells and CD8+ T cells. Int. Immunol. 17, 1227–1237 (2005).
Boles, K. S., Barchet, W., Diacovo, T., Cella, M. & Colonna, M. The tumor suppressor TSLC1/NECL-2 triggers NK-cell and CD8+ T-cell responses through the cell-surface receptor CRTAM. Blood 106, 779–786 (2005).
Takeuchi, A. et al. CRTAM confers late-stage activation of CD8+ T cells to regulate retention within lymph node. J. Immunol. 183, 4220–4228 (2009).
Yeh, J. H., Sidhu, S. S. & Chan, A. C. Regulation of a late phase of T cell polarity and effector functions by Crtam. Cell 132, 846–859 (2008). This article provides an in-depth analysis of the role and signalling of CRTAM in CD4+ T cells using CRTAM-deficient mice.
Ikeda, W. et al. Nectin-like molecule-5/Tage4 enhances cell migration in an integrin-dependent, Nectin-3-independent manner. J. Biol. Chem. 279, 18015–18025 (2004).
Freistadt, M. S. & Eberle, K. E. Physical association between CD155 and CD44 in human monocytes. Mol. Immunol. 34, 1247–1257 (1997).
Mueller, S. & Wimmer, E. Recruitment of nectin-3 to cell-cell junctions through trans-heterophilic interaction with CD155, a vitronectin and poliovirus receptor that localizes to αvβ3 integrin-containing membrane microdomains. J. Biol. Chem. 278, 31251–31260 (2003).
Acknowledgements
The authors thank members of their laboratory for contributions in this area. L.M. and M.J.S. are supported by a National Health and Medical Research Council of Australia Fellowship and Project Grant. L.M. is supported by the Association pour la Recherche Contre le Cancer.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
M.J.S. holds a provisional patent “Immunoreceptor modulation for treating cancer and viral infections”. L.M. declares no competing interests.
Rights and permissions
About this article
Cite this article
Martinet, L., Smyth, M. Balancing natural killer cell activation through paired receptors. Nat Rev Immunol 15, 243–254 (2015). https://doi.org/10.1038/nri3799
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3799
This article is cited by
-
The role of exhausted natural killer cells in the immunopathogenesis and treatment of leukemia
Cell Communication and Signaling (2024)
-
NK cells as powerful therapeutic tool in cancer immunotherapy
Cellular Oncology (2024)
-
ADSC secretome constrains NK cell activity by attenuating IL-2-mediated JAK-STAT and AKT signaling pathway via upregulation of CIS and DUSP4
Stem Cell Research & Therapy (2023)
-
The Role of NK Cells and Their Exosomes in Graft Versus Host Disease and Graft Versus Leukemia
Stem Cell Reviews and Reports (2023)
-
Genomic view of the origins of cell-mediated immunity
Immunogenetics (2023)