Abstract
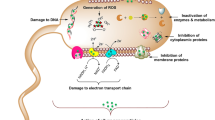
Silver nanoparticles have antibacterial properties, but their use has been a cause for concern because they persist in the environment. Here, we show that lignin nanoparticles infused with silver ions and coated with a cationic polyelectrolyte layer form a biodegradable and green alternative to silver nanoparticles. The polyelectrolyte layer promotes the adhesion of the particles to bacterial cell membranes and, together with silver ions, can kill a broad spectrum of bacteria, including Escherichia coli, Pseudomonas aeruginosa and quaternary-amine-resistant Ralstonia sp. Ion depletion studies have shown that the bioactivity of these nanoparticles is time-limited because of the desorption of silver ions. High-throughput bioactivity screening did not reveal increased toxicity of the particles when compared to an equivalent mass of metallic silver nanoparticles or silver nitrate solution. Our results demonstrate that the application of green chemistry principles may allow the synthesis of nanoparticles with biodegradable cores that have higher antimicrobial activity and smaller environmental impact than metallic silver nanoparticles.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Panáček, A. et al. Antifungal activity of silver nanoparticles against Candida spp. Biomaterials 30, 6333–6340 (2009).
Lara, H., Garza-Trevino, E., Ixtepan-Turrent, L. & Singh, D. Silver nanoparticles are broad-spectrum bactericidal and virucidal compounds. J. Nanobiotechnol. 9, 30 (2011).
Sondi, I. & Salopek-Sondi, B. Silver nanoparticles as antimicrobial agent: a case study on E. coli as a model for Gram-negative bacteria. J. Colloid Interface Sci. 275, 177–182 (2004).
Poole, K., Krebes, K., McNally, C. & Neshat, S. Multiple antibiotic resistance in Pseudomonas aeruginosa: evidence for involvement of an efflux operon. J. Bacteriol. 175, 7363–7372 (1993).
Panáček, A. et al. Silver colloid nanoparticles: synthesis, characterization, and their antibacterial activity. J. Phys. Chem. B 110, 16248–16253 (2006).
Morones, J. R. et al. The bactericidal effect of silver nanoparticles. Nanotechnology 16, 2346–2353 (2005).
Rai, M., Yadav, A. & Gade, A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 27, 76–83 (2009).
Walser, T. et al. Persistence of engineered nanoparticles in a municipal solid-waste incineration plant. Nature Nanotech. 7, 520–524 (2012).
Jeong, E. et al. Different susceptibilities of bacterial community to silver nanoparticles in wastewater treatment systems. J. Environ. Sci. Health Part A 49, 685–693 (2014).
Levard, C., Hotze, E. M., Lowry, G. V. & Brown, G. E. Environmental transformations of silver nanoparticles: impact on stability and toxicity. Environ. Sci. Technol. 46, 6900–6914 (2012).
Chinnapongse, S. L., MacCuspie, R. I. & Hackley, V. A. Persistence of singly dispersed silver nanoparticles in natural freshwaters, synthetic seawater, and simulated estuarine waters. Sci. Total Environ. 409, 2443–2450 (2011).
Sharma, V. K., Siskova, K. M., Zboril, R. & Gardea-Torresdey, J. L. Organic-coated silver nanoparticles in biological and environmental conditions: fate, stability and toxicity. Adv. Colloid Interface Sci. 204, 15–34 (2014).
Dobias, J. & Bernier-Latmani, R. Silver release from silver nanoparticles in natural waters. Environ. Sci. Technol. 47, 4140–4146 (2013).
Stern, S. T. & McNeil, S. E. Nanotechnology safety concerns revisited. Toxicol. Sci. 101, 4–21 (2008).
Ahamed, M., AlSalhi, M. S. & Siddiqui, M. K. J. Silver nanoparticle applications and human health. Clin. Chim. Acta 411, 1841–1848 (2010).
Fabrega, J., Luoma, S. N., Tyler, C. R., Galloway, T. S. & Lead, J. R. Silver nanoparticles: behaviour and effects in the aquatic environment. Environ. Int. 37, 517–531 (2011).
Justo-Hanani, R. & Dayan, T. The role of the state in regulatory policy for nanomaterials risk: analyzing the expansion of state-centric rulemaking in EU and US chemicals policies. Res. Policy 43, 169–178 (2014).
Marchant, G., Sylvester, D. & Abbott, K. Risk management principles for nanotechnology. Nanoethics 2, 43–60 (2008).
Anastas, P. & Eghbali, N. Green chemistry: principles and practice. Chem. Soc. Rev. 39, 301–312 (2010).
Sharma, V. K., Yngard, R. A. & Lin, Y. Silver nanoparticles: green synthesis and their antimicrobial activities. Adv. Colloid Interface Sci. 145, 83–96 (2009).
Kumar, A., Vemula, P. K., Ajayan, P. M. & John, G. Silver-nanoparticle-embedded antimicrobial paints based on vegetable oil. Nature Mater. 7, 236–241 (2008).
Raveendran, P., Fu, J. & Wallen, S. L. Completely ‘green’ synthesis and stabilization of metal nanoparticles. J. Am. Chem. Soc. 125, 13940–13941 (2003).
Xiu, Z., Zhang, Q., Puppala, H. L., Colvin, V. L. & Alvarez, P. J. J. Negligible particle-specific antibacterial activity of silver nanoparticles. Nano Lett. 12, 4271–4275 (2012).
Nel, A. E. et al. Understanding biophysicochemical interactions at the nano–bio interface. Nature Mater. 8, 543–557 (2009).
Ge, C. et al. Towards understanding of nanoparticle–protein corona. Arch. Toxicol. 89, 519–539 (2015).
Lundqvist, M. et al. Nanoparticle size and surface properties determine the protein corona with possible implications for biological impacts. Proc. Natl Acad. Sci. USA 105, 14265–14270 (2008).
Verma, A. & Stellacci, F. Effect of surface properties on nanoparticle–cell interactions. Small 6, 12–21 (2010).
Monopoli, M. P., Aberg, C., Salvati, A. & Dawson, K. A. Biomolecular coronas provide the biological identity of nanosized materials. Nature Nanotech. 7, 779–786 (2012).
El Badawy, A. M. et al. Surface charge-dependent toxicity of silver nanoparticles. Environ. Sci. Technol. 45, 283–287 (2010).
Sotiriou, G. A. & Pratsinis, S. E. Antibacterial activity of nanosilver ions and particles. Environ. Sci. Technol. 44, 5649–5654 (2010).
Feng, Q. L. et al. A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus. J. Biomed. Mater. Res. 52, 662–668 (2000).
Matsumura, Y., Yoshikata, K., Kunisaki, S. & Tsuchido, T. Mode of bactericidal action of silver zeolite and its comparison with that of silver nitrate. Appl. Environ. Microbiol. 69, 4278–4281 (2003).
Norgren, M. & Edlund, H. Lignin: recent advances and emerging applications. Curr. Opin. Colloid Interface Sci. 19, 409–416 (2014).
Duval, A. & Lawoko, M. A review on lignin-based polymeric, micro- and nano-structured materials. React. Funct. Polym. 85, 78–96 (2014).
Guo, X., Zhang, S. & Shan, X.-Q. Adsorption of metal ions on lignin. J. Hazard. Mater. 151, 134–142 (2008).
Harmita, H., Karthikeyan, K. G. & Pan, X. Copper and cadmium sorption onto kraft and organosolv lignins. Bioresour. Technol. 100, 6183–6191 (2009).
Wege, H. A., Kim, S., Paunov, V. N., Zhong, Q. & Velev, O. D. Long-term stabilization of foams and emulsions with in-situ formed microparticles from hydrophobic cellulose. Langmuir 24, 9245–9253 (2008).
Frangville, C. et al. Fabrication of environmentally biodegradable lignin nanoparticles. ChemPhysChem 13, 4235–4243 (2012).
Petridis, L. et al. Self-similar multiscale structure of lignin revealed by neutron scattering and molecular dynamics simulation. Phys. Rev. E 83, 061911 (2011).
Kim, S., Barraza, H. & Velev, O. D. Intense and selective coloration of foams stabilized with functionalized particles. J. Mater. Chem. 19, 7043–7049 (2009).
Langsrud, S., Sundheim, G. & Borgmann-Strahsen, R. Intrinsic and acquired resistance to quaternary ammonium compounds in food-related Pseudomonas spp. J. Appl. Microbiol. 95, 874–882 (2003).
Silva, T. et al. Particle size, surface charge and concentration dependent ecotoxicity of three organo-coated silver nanoparticles: comparison between general linear model-predicted and observed toxicity. Sci. Total Environ. 468–469, 968–976 (2014).
Tan, S., Erol, M., Attygalle, A., Du, H. & Sukhishvili, S. Synthesis of positively charged silver nanoparticles via photoreduction of AgNO3 in branched polyethyleneimine/HEPES solutions. Langmuir 23, 9836–9843 (2007).
Kavlock, R. et al. Update on EPA's ToxCast program: providing high throughput decision support tools for chemical risk management. Chem. Res. Toxicol. 25, 1287–1302 (2012).
Choi, O. et al. Role of sulfide and ligand strength in controlling nanosilver toxicity. Water Res. 43, 1879–1886 (2009).
Wandrey, C., Hernández-Barajas, J. & Hunkeler, D. in Radical polymerisation polyelectrolytes Vol. 145 Advances in Polymer Science (eds Capek, I. et al.) Ch. 3, 123–183 (Springer, 1999).
Gélinas, P. & Goulet, J. Neutralization of the activity of eight disinfectants by organic matter. J. Appl. Bacteriol. 54, 243–247 (1983).
Lundquist, K., Kirk, T. K. & Connors, W. Fungal degradation of kraft lignin and lignin sulfonates prepared from synthetic 14C-lignins. Arch. Microbiol. 112, 291–296 (1977).
Bugg, T. D. H., Ahmad, M., Hardiman, E. M. & Rahmanpour, R. Pathways for degradation of lignin in bacteria and fungi. Nat. Prod. Rep. 28, 1883–1896 (2011).
Klein, C. L. et al. NM-series of Representative Manufactured Nanomaterials NM-300 Silver Characterisation, Stability, Homogeneity (JRC Scientific and Technical Reports, Joint Research Centre, European Commission, 2011).
Acknowledgements
The authors acknowledge funding from the United States Environmental Protection Agency (US EPA) via a Pathfinder Innovation Projects grant, the National Science Foundation Tringle Center for Programmable Soft Matter (DMR-1121107) and North Carolina State University. The authors thank D. Plemmons and A.K. Sarkar for assistance with initial studies, and H. Armstrong for assistance with Ag+ ion desorption studies. Thanks go to M. Moore for the analysis and characterization of PDAC-resistant Ralstonia sp. bacteria, R. Garcia for transmission electron microscopy analysis and K. Hutchinson and the Analytical Spectroscopy Service Laboratory at North Carolina State University for silver content analysis. S.D.S. acknowledges financial support from European Cooperation in Science and Technology Actions MP1305 and MP1106, as well European Union Project FP7-REGPOT-2011-1, ‘Beyond Everest’. The views expressed in this paper are those of the authors and do not necessarily represent the views or policies of the United States Environmental Protection Agency.
Author information
Authors and Affiliations
Contributions
A.P.R. planned and performed the key experiments and analysed the results. J.S.B. synthesized EbNPs and tested their antimicrobial efficiency. B.B. contributed to discussions and confocal microscopy imaging. A.W., S.G., K.H., and E.A.H. carried out the EPA toxicity evaluation, and analysed and plotted the ToxCast data. A.P.R., A.W. and O.D.V. analysed the results, and all authors discussed them and commented on the manuscript. O.D.V., V.N.P. and S.D.S. conceived the project and contributed with ideas and analysis. O.D.V. is the principal investigator, and led the research team.
Corresponding author
Ethics declarations
Competing interests
A.P.R., V.N.P., S.D.S. and O.D.V. declare potential financial interests in the future development and commercialization of similar nanomaterials. NC State University has filed a patent application (PCT/US2014/022382) and has licensed the EbNP technology to a commercial entity.
Supplementary information
Rights and permissions
About this article
Cite this article
Richter, A., Brown, J., Bharti, B. et al. An environmentally benign antimicrobial nanoparticle based on a silver-infused lignin core. Nature Nanotech 10, 817–823 (2015). https://doi.org/10.1038/nnano.2015.141
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2015.141