Abstract
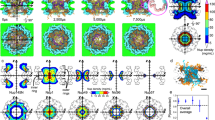
The nuclear pore complex (NPC) is the gate for transport between the cell nucleus and the cytoplasm. Small molecules cross the NPC by passive diffusion, but molecules larger than ∼5 nm must bind to nuclear transport receptors to overcome a selective barrier within the NPC1. Although the structure and shape of the cytoplasmic ring of the NPC are relatively well characterized2,3,4,5, the selective barrier is situated deep within the central channel of the NPC and depends critically on unstructured nuclear pore proteins5,6, and is therefore not well understood. Here, we show that stiffness topography7 with sharp atomic force microscopy tips can generate nanoscale cross-sections of the NPC. The cross-sections reveal two distinct structures, a cytoplasmic ring and a central plug structure, which are consistent with the three-dimensional NPC structure derived from electron microscopy2,3,4,5. The central plug persists after reactivation of the transport cycle and resultant cargo release, indicating that the plug is an intrinsic part of the NPC barrier. Added nuclear transport receptors accumulate on the intact transport barrier and lead to a homogenization of the barrier stiffness. The observed nanomechanical properties in the NPC indicate the presence of a cohesive barrier to transport and are quantitatively consistent with the presence of a central condensate of nuclear pore proteins in the NPC channel.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Mohr, D., Frey, S., Fischer, T., Güttler, T. & Görlich, D. Characterisation of the passive permeability barrier of nuclear pore complexes. EMBO J. 28, 2541–2553 (2009).
Fahrenkrog, B. & Aebi, U. The nuclear pore complex: nucleocytoplasmic transport and beyond. Nature Rev. Mol. Cell Biol. 4, 757–766 (2003).
Alber, F. et al. The molecular architecture of the nuclear pore complex. Nature 450, 695–701 (2007).
Beck, M., Lucic, V., Forster, F., Baumeister, W. & Medalia, O. Snapshots of nuclear pore complexes in action taken by cryoelectron tomography. Nature 449, 611–615 (2007).
Hoelz, A., Debler, E. W. & Blobel, G. Structure of the nuclear pore complex. Annu. Rev. Biochem. 80, 613–643 (2011).
Tetenbaum-Novatt, J. & Rout, M. P. The mechanism of nucleocytoplasmic transport through the nuclear pore complex. Cold Spring Harb. Symp. Quant. Biol. 75, 567–584 (2011).
Roduit, C. et al. Stiffness tomography by atomic force microscopy. Biophys. J. 97, 674–677 (2009).
Adams, R. L. & Wente, S. R. Uncovering nuclear pore complexity with innovation. Cell 152, 1218–1221 (2013).
Lowe, A. R. et al. Selectivity mechanism of the nuclear pore complex characterized by single cargo tracking. Nature 467, 600–603 (2010).
Goryaynov, A., Ma, J. & Yang, W. Single-molecule studies of nucleocytoplasmic transport: from one dimension to three dimensions. Integr. Biol. (Camb.) 4, 10–21 (2012).
Macara, I. G. Transport into and out of the nucleus. Microbiol. Mol. Biol. Rev. 65, 570–594 (2001).
Rout, M. P., Aitchison, J. D., Magnasco, M. O. & Chait, B. T. Virtual gating and nuclear transport: the hole picture. Trends Cell Biol. 13, 622–628 (2003).
Lim, R. Y. et al. Nanomechanical basis of selective gating by the nuclear pore complex. Science 318, 640–643 (2007).
Patel, S. S., Belmont, B. J., Sante, J. M. & Rexach, M. F. Natively unfolded nucleoporins gate protein diffusion across the nuclear pore complex. Cell 129, 83–96 (2007).
Frey, S. & Görlich, D. A saturated FG-repeat hydrogel can reproduce the permeability properties of nuclear pore complexes. Cell 130, 512–523 (2007).
Jäggi, R. D. et al. Modulation of nuclear pore topology by transport modifiers. Biophys. J. 84, 665–670 (2003).
Kramer, A. et al. Apoptosis leads to a degradation of vital components of active nuclear transport and a dissociation of the nuclear lamina. Proc. Natl Acad. Sci. USA 105, 11236–11241 (2008).
Keminer, O., Siebrasse, J. P., Zerf, K. & Peters, R. Optical recording of signal-mediated protein transport through single nuclear pore complexes. Proc. Natl Acad. Sci. USA 96, 11842–11847 (1999).
Eisele, N. B., Labokha, A. A., Frey, S., Görlich, D. & Richter, R. P. Cohesiveness tunes assembly and morphology of FG nucleoporin domain meshworks—implications for nuclear pore permeability. Biophys. J. 105, 1860–1870 (2013).
Rangl, M. et al. Single-molecule analysis of the recognition forces underlying nucleo-cytoplasmic transport. Angew. Chem. Int. Ed. 52, 10356–10359 (2013).
Tetard, L., Passian, A. & Thundat, T. New modes for subsurface atomic force microscopy through nanomechanical coupling. Nature Nanotech. 5, 105–109 (2010).
Wilson, N. R. & Macpherson, J. V. Carbon nanotube tips for atomic force microscopy. Nature Nanotech. 4, 483–491 (2009).
Yamada, J. et al. A bimodal distribution of two distinct categories of intrinsically disordered structures with separate functions in FG nucleoporins. Mol. Cell Proteomics 9, 2205–2224 (2010).
Osmanović, D. et al. Bistable collective behavior of polymers tethered in a nanopore. Phys. Rev. E 85, 061917 (2012).
Schoch, R. L., Kapinos, L. E. & Lim, R. Y. Nuclear transport receptor binding avidity triggers a self-healing collapse transition in FG-nucleoporin molecular brushes. Proc. Natl Acad. Sci. USA 109, 16911–16916 (2012).
Jovanovic-Talisman, T. et al. Artificial nanopores that mimic the transport selectivity of the nuclear pore complex. Nature 457, 1023–1027 (2009).
Tagliazucchi, M., Peleg, O., Kröger, M., Rabin, Y. & Szleifer, I. Effect of charge, hydrophobicity, and sequence of nucleoporins on the translocation of model particles through the nuclear pore complex. Proc. Natl Acad. Sci. USA 110, 3363–3368 (2013).
Peleg, O., Tagliazucchi, M., Kröger, M., Rabin, Y. & Szleifer, I. Morphology control of hairy nanopores. ACS Nano 5, 4737–4747 (2011).
Osmanović, D., Ford, I. J. & Hoogenboom, B. W. Model inspired by nuclear pore complex suggests possible roles for nuclear transport receptors in determining its structure. Biophys. J. 105, 2781–2789 (2013).
Zaitseva, L., Myers, R. & Fassati, A. tRNAs promote nuclear import of HIV-1 intracellular reverse transcription complexes. PLoS Biol. 4, e332 (2006).
Acknowledgements
The authors thank H. Oberleithner for the use of laboratory facilities, P. Vergani for assistance with the preparation of X. laevis oocytes, D. Görlich for the expression plasmids for importin β and Ran-mix, S. Frey for Rch1-IBB-MBP-GFP protein, R.P. Richter for providing the data presented in Supplementary Fig. 8, J. Grech for the electron microscopy image in Fig. 1a, M. Goldberg for the protocol for removal of the nuclear basket from the NPCs, J. Bailey and A.H. Harker for support in initializing the modelling work, R. Thorogate for assistance with the confocal microscopy measurements, C. Leung for assistance in formatting the figures and T. Duke (deceased) and G. Aeppli for proofreading the manuscript. This work was partially funded by the European Molecular Biology Organization (ALTF 757-2008 to A.K.), the Kazakh Ministry of Education and Science (A.B.), the Sackler Foundation (D.O.), the UK Biotechnology and Biological Sciences Research Council (BB/G011729/1 to B.W.H.) and the Wellcome Trust (083810/Z/07/Z to A.F.). G.C. is a Royal Society University Research Fellow.
Author information
Authors and Affiliations
Contributions
A.B., A.K., A.L., I.L., G.C., A.F. and B.W.H. designed the experiments. A.B., A.K., A.L. and I.L. performed the experiments. A.B., A.K. and B.W.H., with support from E.V.O., performed the data analysis. D.O., I.J.F. and B.W.H. designed and performed the polymer modelling. A.F. and B.W.H. wrote the manuscript. All authors read and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary Information (PDF 1849 kb)
Supplementary information
Supplementary Movie 1 (AVI 1632 kb)
Supplementary information
Supplementary Movie 2 (AVI 1632 kb)
Rights and permissions
About this article
Cite this article
Bestembayeva, A., Kramer, A., Labokha, A. et al. Nanoscale stiffness topography reveals structure and mechanics of the transport barrier in intact nuclear pore complexes. Nature Nanotech 10, 60–64 (2015). https://doi.org/10.1038/nnano.2014.262
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2014.262
This article is cited by
-
A simple thermodynamic description of phase separation of Nup98 FG domains
Nature Communications (2022)
-
The mechanical stability of proteins regulates their translocation rate into the cell nucleus
Nature Physics (2019)
-
Imaging modes of atomic force microscopy for application in molecular and cell biology
Nature Nanotechnology (2017)
-
Visualization of Au Nanoparticles Buried in a Polymer Matrix by Scanning Thermal Noise Microscopy
Scientific Reports (2017)
-
HIV-1 capsid is involved in post-nuclear entry steps
Retrovirology (2016)