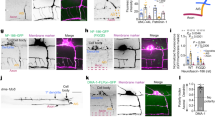
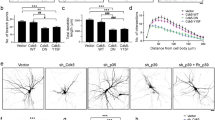
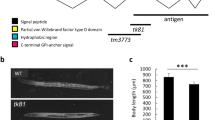
Abstract
Dendrites often adopt complex branched structures. The development and organization of these arbors fundamentally determine the potential input and connectivity of a given neuron. The cell-surface receptors that control dendritic branching remain poorly understood. We found that, in Caenorhabditis elegans, a previously uncharacterized transmembrane protein containing extracellular leucine-rich repeat (LRR) domains, which we named DMA-1 (dendrite-morphogenesis-abnormal), promotes dendrite branching and growth. Sustained expression of dma-1 was found only in the elaborately branched sensory neurons PVD and FLP. Genetic analysis revealed that the loss of dma-1 resulted in much reduced dendritic arbors, whereas overexpression of dma-1 resulted in excessive branching. Forced expression of dma-1 in neurons with simple dendrites was sufficient to promote ectopic branching. Worms lacking dma-1 were defective in sensing harsh touch. DMA-1 is the first transmembrane LRR protein to be implicated in dendritic branching and expands the breadth of roles of LRR receptors in nervous system development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Masland, R.H. Neuronal cell types. Curr. Biol. 14, R497–R500 (2004).
Ramon y Cajal, S. Histology of the Nervous System of Man and Vertebrates (Oxford University Press, 1995).
Wen, Q., Stepanyants, A., Elston, G.N., Grosberg, A.Y. & Chklovskii, D.B. Maximization of the connectivity repertoire as a statistical principle governing the shapes of dendritic arbors. Proc. Natl. Acad. Sci. USA 106, 12536–12541 (2009).
Aizawa, H. et al. Dendrite development regulated by CREST, a calcium-regulated transcriptional activator. Science 303, 197–202 (2004).
Gaudillière, B., Konishi, Y., de la Iglesia, N., Yao, G. & Bonni, A.A. CaMKII-NeuroD signaling pathway specifies dendritic morphogenesis. Neuron 41, 229–241 (2004).
Morita, A. et al. Regulation of dendritic branching and spine maturation by semaphorin3A-Fyn signaling. J. Neurosci. 26, 2971–2980 (2006).
Grueber, W.B. et al. Projections of Drosophila multidendritic neurons in the central nervous system: links with peripheral dendrite morphology. Development 134, 55–64 (2007).
Jan, Y.N. & Jan, L.Y. Branching out: mechanisms of dendritic arborization. Nat. Rev. Neurosci. 11, 316–328 (2010).
Grueber, W.B., Jan, L.Y. & Jan, Y.N. Different levels of the homeodomain protein cut regulate distinct dendrite branching patterns of Drosophila multidendritic neurons. Cell 112, 805–818 (2003).
Sugimura, K., Satoh, D., Estes, P., Crews, S. & Uemura, T. Development of morphological diversity of dendrites in Drosophila by the BTB-zinc finger protein abrupt. Neuron 43, 809–822 (2004).
Satoh, D. et al. Spatial control of branching within dendritic arbors by dynein-dependent transport of Rab5-endosomes. Nat. Cell Biol. 10, 1164–1171 (2008).
Zheng, Y. et al. Dynein is required for polarized dendritic transport and uniform microtubule orientation in axons. Nat. Cell Biol. 10, 1172–1180 (2008).
Sweeney, N.T., Brenman, J.E., Jan, Y.N. & Gao, F.B. The coiled-coil protein shrub controls neuronal morphogenesis in Drosophila. Curr. Biol. 16, 1006–1011 (2006).
Ye, B. et al. Growing dendrites and axons differ in their reliance on the secretory pathway. Cell 130, 717–729 (2007).
Jaworski, J., Spangler, S., Seeburg, D.P., Hoogenraad, C.C. & Sheng, M. Control of dendritic arborization by the phosphoinositide-3′-kinase-Akt-mammalian target of rapamycin pathway. J. Neurosci. 25, 11300–11312 (2005).
Emoto, K. et al. Control of dendritic branching and tiling by the Tricornered-kinase/Furry signaling pathway in Drosophila sensory neurons. Cell 119, 245–256 (2004).
Ye, B. et al. Nanos and Pumilio are essential for dendrite morphogenesis in Drosophila peripheral neurons. Curr. Biol. 14, 314–321 (2004).
Albeg, A. et al. C. elegans multi-dendritic sensory neurons: morphology and function. Mol. Cell Neurosci. 46, 308–317 (2010).
Smith, C.J. et al. Time-lapse imaging and cell-specific expression profiling reveal dynamic branching and molecular determinants of a multi-dendritic nociceptor in C. elegans. Dev. Biol. 345, 18–33 (2010).
Shen, K. & Cowan, C.W. Guidance molecules in synapse formation and plasticity. Cold Spring Harb. Perspect. Biol. 2, a001842 (2010).
Shapiro, L., Love, J. & Colman, D.R. Adhesion molecules in the nervous system: structural insights into function and diversity. Annu. Rev. Neurosci. 30, 451–474 (2007).
Dolan, J. et al. The extracellular leucine-rich repeat superfamily; a comparative survey and analysis of evolutionary relationships and expression patterns. BMC Genomics 8, 320 (2007).
Chen, Y., Aulia, S., Li, L. & Tang, B.L. AMIGO and friends: an emerging family of brain-enriched, neuronal growth modulating, type I transmembrane proteins with leucine-rich repeats (LRR) and cell adhesion molecule motifs. Brain Res. Rev. 51, 265–274 (2006).
de Wit, J. et al. LRRTM2 interacts with Neurexin1 and regulates excitatory synapse formation. Neuron 64, 799–806 (2009).
Hong, W. et al. Leucine-rich repeat transmembrane proteins instruct discrete dendrite targeting in an olfactory map. Nat. Neurosci. 12, 1542–1550 (2009).
Ko, J., Fuccillo, M.V., Malenka, R.C. & Sudhof, T.C. LRRTM2 functions as a neurexin ligand in promoting excitatory synapse formation. Neuron 64, 791–798 (2009).
Linhoff, M.W. et al. An unbiased expression screen for synaptogenic proteins identifies the LRRTM protein family as synaptic organizers. Neuron 61, 734–749 (2009).
Tursun, B., Cochella, L., Carrera, I. & Hobert, O. A toolkit and robust pipeline for the generation of fosmid-based reporter genes in C. elegans. PLoS ONE 4, e4625 (2009).
Oren-Suissa, M., Hall, D.H., Treinin, M., Shemer, G. & Podbilewicz, B. The fusogen EFF-1 controls sculpting of mechanosensory dendrites. Science 328, 1285–1288 (2010).
Tsalik, E.L. et al. LIM homeobox gene–dependent expression of biogenic amine receptors in restricted regions of the C. elegans nervous system. Dev. Biol. 263, 81–102 (2003).
Frøkjær-Jensen, C. et al. Targeted gene deletions in C. elegans using transposon excision. Nat. Methods 7, 451–453 (2010).
Way, J.C. & Chalfie, M. The mec-3 gene of Caenorhabditis elegans requires its own product for maintained expression and is expressed in three neuronal cell types. Genes Dev. 3, 1823–1833 (1989).
Li, W., Kang, L., Piggott, B.J., Feng, Z. & Xu, X.Z. The neural circuits and sensory channels mediating harsh touch sensation in Caenorhabditis elegans. Nat. Commun. 2, 315 (2011).
Hutter, H., Ng, M.P. & Chen, N. GExplore: a web server for integrated queries of protein domains, gene expression and mutant phenotypes. BMC Genomics 10, 529 (2009).
Hattori, D., Millard, S.S., Wojtowicz, W.M. & Zipursky, S.L. Dscam-mediated cell recognition regulates neural circuit formation. Annu. Rev. Cell Dev. Biol. 24, 597–620 (2008).
Dimitrova, S., Reissaus, A. & Tavosanis, G. Slit and Robo regulate dendrite branching and elongation of space-filling neurons in Drosophila. Dev. Biol. 324, 18–30 (2008).
McAllister, A.K., Katz, L.C. & Lo, D.C. Opposing roles for endogenous BDNF and NT-3 in regulating cortical dendritic growth. Neuron 18, 767–778 (1997).
Long, H., Ou, Y., Rao, Y. & van Meyel, D.J. Dendrite branching and self-avoidance are controlled by Turtle, a conserved IgSF protein in Drosophila. Development 136, 3475–3484 (2009).
Bella, J., Hindle, K.L., McEwan, P.A. & Lovell, S.C. The leucine-rich repeat structure. Cell. Mol. Life Sci. 65, 2307–2333 (2008).
Morlot, C. et al. Structural insights into the Slit-Robo complex. Proc. Natl. Acad. Sci. USA 104, 14923–14928 (2007).
Schubert, W.D. et al. Structure of internalin, a major invasion protein of Listeria monocytogenes, in complex with its human receptor E-cadherin. Cell 111, 825–836 (2002).
Caldwell, J.C., Fineberg, S.K. & Eberl, D.F. reduced ocelli encodes the leucine rich repeat protein Pray For Elves in Drosophila melanogaster. Fly (Austin) 1, 146–152 (2007).
Oldenburg, K.R., Vo, K.T., Michaelis, S. & Paddon, C. Recombination-mediated PCR-directed plasmid construction in vivo in yeast. Nucleic Acids Res. 25, 451–452 (1997).
Mello, C. & Fire, A. DNA transformation. Methods Cell Biol. 48, 451–482 (1995).
Wang, G.J. et al. GRLD-1 regulates cell-wide abundance of glutamate receptor through post-transcriptional regulation. Nat. Neurosci. 13, 1489–1495 (2010).
Acknowledgements
We thank C. Gao and Y. Fu for technical assistance, C. Chen, K. Mizumoto, P. Chia and A. Hellman for critical reading of the manuscript, and the members of the Shen laboratory for helpful discussion. We would also like to thank the members of the Hobert and the Jorgensen laboratories for sharing plasmids and expertise regarding fosmid recombineering and MosDel. This work was supported by grants from the Howard Hughes Medical Institute and the US National Institutes of Health to K.S., and a postdoctoral fellowship from the Jane Coffin Childs Memorial Fund to O.W.L.
Author information
Authors and Affiliations
Contributions
O.W.L. conducted the experiments. K.S. supervised the project. O.W.L. and K.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Table 1 (PDF 1359 kb)
Rights and permissions
About this article
Cite this article
Liu, O., Shen, K. The transmembrane LRR protein DMA-1 promotes dendrite branching and growth in C. elegans. Nat Neurosci 15, 57–63 (2012). https://doi.org/10.1038/nn.2978
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2978
This article is cited by
-
Ribo-On and Ribo-Off tools using a self-cleaving ribozyme allow manipulation of endogenous gene expression in C. elegans
Communications Biology (2023)
-
Endocytosis in the axon initial segment maintains neuronal polarity
Nature (2022)
-
CPB-3 and CGH-1 localize to motile particles within dendrites in C. elegans PVD sensory neurons
BMC Research Notes (2021)
-
Deep learning-enabled analysis reveals distinct neuronal phenotypes induced by aging and cold-shock
BMC Biology (2020)
-
Atlastin-1 regulates morphology and function of endoplasmic reticulum in dendrites
Nature Communications (2019)