Abstract
Deregulation of the TH17 subset of helper T cells is closely linked with immunological disorders and inflammatory diseases. However, the mechanism by which TH17 cells are regulated remains elusive. Here we found that the phosphatase DUSP2 (PAC1) negatively regulated the development of TH17 cells. DUSP2 was directly associated with the signal transducer and transcription activator STAT3 and attenuated its activity through dephosphorylation of STAT3 at Tyr705 and Ser727. DUSP2-deficient mice exhibited severe susceptibility to experimental colitis, with enhanced differentiation of TH17 cells and secretion of proinflammatory cytokines. In clinical patients with ulcerative colitis, DUSP2 was downregulated by DNA methylation and was not induced during T cell activation. Our data demonstrate that DUSP2 is a true STAT3 phosphatase that modulates the development of TH17 cells in the autoimmune response and inflammation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Accession codes
Change history
10 November 2015
In the version of this article initially published online, in Figure 2a, the middle plot in the bottom row was a duplicate of another plot, and in Figure 5c, the top right plot was a duplicate of another plot. These errors have been corrected for the print, PDF and HTML versions of this article.
References
Korn, T., Bettelli, E., Oukka, M. & Kuchroo, V.K. IL-17 and Th17 cells. Annu. Rev. Immunol. 27, 485–517 (2009).
Yang, X.O. et al. T helper 17 lineage differentiation is programmed by orphan nuclear receptors RORα and RORγ. Immunity 28, 29–39 (2008).
Yang, X.O. et al. STAT3 regulates cytokine-mediated generation of inflammatory helper T cells. J. Biol. Chem. 282, 9358–9363 (2007).
Yang, X.P. et al. Opposing regulation of the locus encoding IL-17 through direct, reciprocal actions of STAT3 and STAT5. Nat. Immunol. 12, 247–254 (2011).
Chaudhry, A. et al. CD4+ regulatory T cells control TH17 responses in a Stat3-dependent manner. Science 326, 986–991 (2009).
Abraham, C. & Cho, J.H. Inflammatory bowel disease. N. Engl. J. Med. 361, 2066–2078 (2009).
Lovato, P. et al. Constitutive STAT3 activation in intestinal T cells from patients with Crohn′s disease. J. Biol. Chem. 278, 16777–16781 (2003).
Musso, A. et al. Signal transducers and activators of transcription 3 signaling pathway: an essential mediator of inflammatory bowel disease and other forms of intestinal inflammation. Inflamm. Bowel Dis. 11, 91–98 (2005).
Barrett, J.C. et al. Genome-wide association defines more than 30 distinct susceptibility loci for Crohn′s disease. Nat. Genet. 40, 955–962 (2008).
McGovern, D.P. et al. Genome-wide association identifies multiple ulcerative colitis susceptibility loci. Nat. Genet. 42, 332–337 (2010).
Durant, L. et al. Diverse targets of the transcription factor STAT3 contribute to T cell pathogenicity and homeostasis. Immunity 32, 605–615 (2010).
Danial, N.N., Pernis, A. & Rothman, P.B. Jak-STAT signaling induced by the v-abl oncogene. Science 269, 1875–1877 (1995).
Jain, N., Zhang, T., Kee, W.H., Li, W. & Cao, X. Protein kinase C δ associates with and phosphorylates Stat3 in an interleukin-6-dependent manner. J. Biol. Chem. 274, 24392–24400 (1999).
Wei, W. et al. Dual-specificity phosphatases 2: surprising positive effect at the molecular level and a potential biomarker of diseases. Genes Immun. 14, 1–6 (2013).
Jeffrey, K.L., Camps, M., Rommel, C. & Mackay, C.R. Targeting dual-specificity phosphatases: manipulating MAP kinase signalling and immune responses. Nat. Rev. Drug Discov. 6, 391–403 (2007).
Huang, G., Wang, Y., Shi, L.Z., Kanneganti, T.D. & Chi, H. Signaling by the phosphatase MKP-1 in dendritic cells imprints distinct effector and regulatory T cell fates. Immunity 35, 45–58 (2011).
Holmes, D.A., Yeh, J.H., Yan, D., Xu, M. & Chan, A.C. Dusp5 negatively regulates IL-33-mediated eosinophil survival and function. EMBO J. 34, 218–235 (2015).
Huang, C.Y. et al. DUSP4 deficiency enhances CD25 expression and CD4+ T-cell proliferation without impeding T-cell development. Eur. J. Immunol. 42, 476–488 (2012).
Yang, C.Y. et al. Dual-specificity phosphatase 14 (DUSP14/MKP6) negatively regulates TCR signaling by inhibiting TAB1 activation. J. Immunol. 192, 1547–1557 (2014).
Li, J.P. et al. The phosphatase JKAP/DUSP22 inhibits T-cell receptor signalling and autoimmunity by inactivating Lck. Nat. Commun. 5, 3618 (2014).
Rohan, P.J. et al. PAC-1: a mitogen-induced nuclear protein tyrosine phosphatase. Science 259, 1763–1766 (1993).
Yin, Y., Liu, Y.X., Jin, Y.J., Hall, E.J. & Barrett, J.C. PAC1 phosphatase is a transcription target of p53 in signalling apoptosis and growth suppression. Nature 422, 527–531 (2003).
Wu, J., Jin, Y.J., Calaf, G.M., Huang, W.L. & Yin, Y. PAC1 is a direct transcription target of E2F–1 in apoptotic signaling. Oncogene 26, 6526–6535 (2007).
Chu, Y., Solski, P.A., Khosravi-Far, R., Der, C.J. & Kelly, K. The mitogen-activated protein kinase phosphatases PAC1, MKP-1, and MKP-2 have unique substrate specificities and reduced activity in vivo toward the ERK2 sevenmaker mutation. J. Biol. Chem. 271, 6497–6501 (1996).
Jeffrey, K.L. et al. Positive regulation of immune cell function and inflammatory responses by phosphatase PAC-1. Nat. Immunol. 7, 274–283 (2006).
Kinney, C.M. et al. Histone H3 as a novel substrate for MAP kinase phosphatase-1. Am. J. Physiol. Cell Physiol. 296, C242–C249 (2009).
Li, J.P., Fu, Y.N., Chen, Y.R. & Tan, T.H. JNK pathway-associated phosphatase dephosphorylates focal adhesion kinase and suppresses cell migration. J. Biol. Chem. 285, 5472–5478 (2010).
Wirtz, S., Neufert, C., Weigmann, B. & Neurath, M.F. Chemically induced mouse models of intestinal inflammation. Nat. Protoc. 2, 541–546 (2007).
Ma, H.L. et al. IL-22 is required for Th17 cell-mediated pathology in a mouse model of psoriasis-like skin inflammation. J. Clin. Invest. 118, 597–607 (2008).
Ostanin, D.V. et al. T cell transfer model of chronic colitis: concepts, considerations, and tricks of the trade. Am. J. Physiol. Gastrointest. Liver Physiol. 296, G135–G146 (2009).
Lee, Y. et al. Induction and molecular signature of pathogenic TH17 cells. Nat. Immunol. 13, 991–999 (2012).
Ghoreschi, K. et al. Generation of pathogenic TH17 cells in the absence of TGF-β signalling. Nature 467, 967–971 (2010).
El-Behi, M. et al. The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23-induced production of the cytokine GM-CSF. Nat. Immunol. 12, 568–575 (2011).
Farooq, A. et al. Solution structure of the MAPK phosphatase PAC-1 catalytic domain. Insights into substrate-induced enzymatic activation of MKP. Structure 11, 155–164 (2003).
Lin, S.C. et al. Suppression of dual-specificity phosphatase-2 by hypoxia increases chemoresistance and malignancy in human cancer cells. J. Clin. Invest. 121, 1905–1916 (2011).
Hammer, M. et al. Increased inflammation and lethality of Dusp1−/− mice in polymicrobial peritonitis models. Immunology 131, 395–404 (2010).
Vang, T. et al. Protein tyrosine phosphatases in autoimmunity. Annu. Rev. Immunol. 26, 29–55 (2008).
Zhang, Y. et al. Regulation of innate and adaptive immune responses by MAP kinase phosphatase 5. Nature 430, 793–797 (2004).
Okamoto, K. et al. IkappaBzeta regulates TH17 development by cooperating with ROR nuclear receptors. Nature 464, 1381–1385 (2010).
Wei, L., Laurence, A., Elias, K.M. & O'Shea, J.J. IL-21 is produced by Th17 cells and drives IL-17 production in a STAT3-dependent manner. J. Biol. Chem. 282, 34605–34610 (2007).
Neurath, M.F. Cytokines in inflammatory bowel disease. Nat. Rev. Immunol. 14, 329–342 (2014).
Ivanov, I.I. et al. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 126, 1121–1133 (2006).
Alex, P. et al. Distinct cytokine patterns identified from multiplex profiles of murine DSS and TNBS-induced colitis. Inflamm. Bowel Dis. 15, 341–352 (2009).
van Loosdregt, J. et al. Stabilization of the transcription factor Foxp3 by the deubiquitinase USP7 increases Treg-cell-suppressive capacity. Immunity 39, 259–271 (2013).
Gu, T. et al. CREB is a novel nuclear target of PTEN phosphatase. Cancer Res. 71, 2821–2825 (2011).
Lu, D. et al. The tumor-suppressive function of UNC5D and its repressed expression in renal cell carcinoma. Clin. Cancer Res. 19, 2883–2892 (2013).
Acknowledgements
We thank C. Huang for help with the generation of Dusp2−/− mice; Y. Li for technical support; Y. Zhou for analysis of mouse phenotypes; J. Zhang for critical reading of the manuscript and comments; and C.Y. Wang for long-term support. Supported by the National Natural Science Foundation of China (81430056, 31420103905 and 81372491 to Y.Y.), the China National Major Scientific Program (2010CB912202 to Y.Y.), the Shu Fan Education Foundation and the Lam Chung Nin Foundation for Systems Biomedicine.
Author information
Authors and Affiliations
Contributions
D.L. and L.L. designed the study, performed most of the experiments, and analyzed the data; X.J. helped with some experiments; Y.G. and Yu Liu contributed to the flow cytometry and enzyme-linked immunosorbent assays; X.C. provided animal care and technical assistance; X.Z. did the mass spectrum analysis; Yang Liu provided clinical material; Yunqiao Li prepared the polyclonal antibody to DUSP2; Yan Li and Y.J. provided technical support; Y.Z. participated in designing the study and provided expertise; M.A.M. discussed and revised the manuscript; Y.Y. designed the project and supervised the experiments; and D.L., L.L. and Y. Y. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Dusp2 is induced during T cell activation.
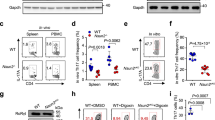
(a) CD4-CD8-, CD4+CD8+, CD4+CD8-, CD4-CD8+ and CD4+CD25+Foxp3+ T cells were isolated from the thymus. CD4+CD8- and CD4-CD8+ T cells, naive (CD4+CD25-CD62LhiCD44low) T cells and B220+ B cells from peripheral lymph nodes (LN) were sorted by flow cytometry. TH0, TH1, TH2, TH17 and iTreg cells were prepared by culturing naive T cells in polarizing conditions. Dusp2 mRNA was evaluated by qRT-PCR. (b) Naive T cells from LN were cultured under stimulation of anti-CD3 plus anti-CD28 (α-CD3 + α-CD28) and TH17- or Treg-skewing conditions, and qRT-PCR was performed at indicated time points during culture to assess Dusp2 or Cd69. (c) Immunoblot analysis of the time course of DUSP2 expression in naive T cells stimulated with PMA and ionomycin (PMA + iono). (d) Naive T cells from LN were left untreated or simulated with anti-CD3 plus anti-CD28 or PMA and ionomycin for 2 h and transcript levels of DUSP family members were measured by qRT-PCR. (e) Naive T cells were stimulated with anti-CD3 and anti-CD28 for various lengths of time, and mRNA levels of Dusp1, Dusp4, Dusp5 or Dusp10 were evaluated by qRT-PCR. (f) Naive T cells were sorted by flow cytometry from LN. TH0, TH1, TH2, TH17 and iTreg cells were prepared by culturing naive T cells in polarizing conditions for 72 h. Total RNA was isolated and mRNA expression of the indicated genes was evaluated by qRT-PCR. Data are representative of three (b,c) or two (a,d-f) independent experiments with three (a) mice per group or with two replicates from three (b,d,e) mice per group (error bars, s.e.m.).
Supplementary Figure 2 Construction of Dusp2−/− mice and characterization of lymphocyte compartment in wild-type and Dusp2−/− mice.
(a) Targeting strategy for generation of Dusp2−/− mice. (b) Dusp2 mRNA expression in thymus (Thy) and LN from wild-type and Dusp2−/− mice was evaluated with qRT-PCR. (c) DUSP2 protein expression. MACS sorted total CD4+ T cells were stimulated with PMA and ionomycin for 4 h and expression of DUSP2 and GAPDH protein was assessed by immunoblotting using antibodies against either DUSP2 or GAPDH. (d) Bone marrow (BM), thymus, spleen (Spl) and peripheral lymph node cellularity. Each symbol represents an individual mouse. (e) Thymic cells from wild-type (WT) and Dusp2−/− mice were prepared and stained for surface CD4, CD8 or CD25 and intracellular Foxp3, followed by analysis with flow cytometry. (f) Peripheral LN from Dusp2−/− and wild-type controls were collected and stained for CD4, CD8, B220, CD62L, CD44, CD25 and Foxp3, followed by flow cytometry analysis. (g) Splenic cells from Dusp2−/− and wild-type controls were collected and the cells were stained for CD4, CD8, B220, CD62L, CD44, CD25 and Foxp3, followed by flow cytometry analysis. (h) Mononuclear cells from LN and Spl from Dusp2−/− and wild-type mice were prepared and activated with PMA and ionomycin for 5 h, followed by staining for IFN-γ and IL-17A and flow cytometry analysis. Data are representative of two (b-h) independent experiments with four (d-g) or three (b,h) mice per group (error bars, s.e.m.).
Supplementary Figure 3 Characterization of Dusp2−/− T cells in vitro.
(a) qRT-PCR analysis of Il2 expression in wild-type and Dusp2−/− T cells stimulated with anti-CD3 and anti-CD28 for various lengths of time. (b) Flow cytometry analysis of CFSE dilution in wild-type and Dusp2−/− CD4+ T cells isolated from LN left untreated (grey shaded curves) or stimulated for 48 h with anti-CD3 and anti-CD28. (c) Naive CD4+ T cells from wild-type and Dusp2−/− mice were polarized in TH17-skewing conditions with various doses of IL-6, followed by intracellular staining of IL-17A. (d) Naive CD4+ T cells from wild-type and Dusp2−/− mice were polarized in Treg-skewing conditions with various doses of TGF-β, followed by flow cytometry analysis of expression of CD25 and Foxp3. (e) Naive CD4+ T cells from wild-type and Dusp2−/− mice were cultured under Treg-polarizing conditions in the presence of various concentrations of IL-2, followed by staining for CD25 and Foxp3. (f) Wild-type or Dusp2−/− CD4+ T cells were stimulated with various doses of IL-6 for 30 min, followed by flow cytometry analysis of expression of phosphorylated STAT3 (Tyr705). (g) Wild-type or Dusp2−/− CD4+ T cells were stimulated with various doses of IL-2 for 15 min, followed by flow cytometry analysis of expression of phosphorylated STAT5 (Tyr694). Each symbol (b-g) represents an individual mouse. NS, not significant; * P < 0.05, ** P < 0.01 and *** P < 0.001 (unpaired Student’s t-test). Data are representative of two (a-g) independent experiments with four (c-e,g) or three (a,b,f) mice per group (error bars, s.e.m.).
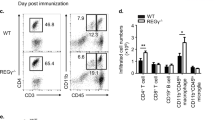
Supplementary Figure 4 Deletion of DUSP2 results in enhanced generation of TH17 cells and T cell response after immunization with KLH.
(a) Draining lymph node cells were prepared from wild-type and Dusp2−/− mice 7 d after subcutaneous immunization with KLH in CFA, followed by stimulation with PMA and ionomycin for 5 h. IL-17A-, IFN-γ- or Foxp3-expressing cells were quantified by intracellular staining. Each symbol represents an individual mouse. (b) Sera were obtained from wild-type and Dusp2−/− mice as in a, and KLH-specific antibodies were measured in sera by ELISA. NS, not significant; * P < 0.05 (unpaired Student’s t-test). Data are representative of three (a,b) independent experiments with four (a) or three (b) mice per group measured in duplicate (b) (error bars, s.e.m.).
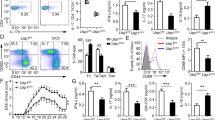
Supplementary Figure 5 DUSP2 deficiency exacerbates DSS-induced colitis in mice.
(a) Mean changes in blood biochemical indices (blood glucose, serum albumin and total cholesterol) of wild-type and Dusp2−/− mice untreated (UT) or treated with 3% DSS for 4 days. (b) Mean changes in body weight of wild-type and Dusp2−/− mice given 5% DSS for 6 days followed by untreated drinking water for 10 days. (c) Survival curves for wild-type and Dusp2−/− mice treated with 3 cycles of DSS (6 days of 2.5% DSS, 14 days of regular water). Log-rank (Mantel-Cox) test. (d) Wild-type and Dusp2−/− mice were administrated with 2.5% DSS for 6 days, followed by 14 days of untreated water, and this cycle was repeated. Mice were sacrificed on day 8 after the beginning of the second DSS cycle and colon shortening was examined. (e) Hematoxylin-and-eosin staining of colons from mice as in d. Scale bars, 50 μm. (f) Mean histologic scores of colonic sections from wild-type and Dusp2−/− mice as in d. Each symbol (a,f) represents an individual mouse. NS, not significant; * P < 0.05, ** P < 0.01 and *** P < 0.001 (unpaired Student’s t-test). Data are representative of two (a,c-f) or three (b) independent experiments with nine (c), five (d-f), four (b) or three (a) mice per group (error bars, s.e.m.).
Supplementary Figure 6 DUSP2 deficiency promotes TH17 differentiation during acute DSS-induced colitis.
Wild-type and Dusp2−/− mice were treated with 3% DSS and sacrificed on day 8. Mononuclear cells from mesenteric lymph nodes (MLN), Peyer’s patches (PP) and the lamina propria (LP) were isolated, followed by surface staining for CD4 and intracellular staining for IFN-γ, IL-17A or Foxp3, and the percentages of different cell populations were calculated. Each symbol represents an individual mouse. NS, not significant; * P < 0.05 and ** P < 0.01 (unpaired Student’s t-test). Data are representative of three independent experiments with four mice per group (error bars, s.e.m.).
Supplementary Figure 7 DUSP2 is not required for the suppressive function of Treg cells.
(a) Mean change in body weight of SCID mice that received wild-type CD45.1+CD4+CD45RBhi cells (RBhi) alone or in combination with CD4+CD25+ cells (Treg) from wild-type or Dusp2−/− mice. (b) Morphologic examination of colons in recipient mice at 8 weeks after transfer of RBhi cells alone or together with Treg cells from wild-type or Dusp2−/− mice. (c) Flow cytometry analysis of Foxp3 expression in CD4+ cells from the MLN and Spl of recipients as in b. Each symbol represents an individual mouse, and horizontal lines indicate the mean. Data are representative of two (a,b) independent experiments with four (a,b) mice per group or are pooled from two (c) independent experiments with eight (c) mice per group (error bars, s.e.m.).
Supplementary Figure 8 Role of DUSP2 in modulating TH17 differentiation.
(a) Model for the role of DUSP2 in modulation of T cell homeostasis. (b) HEK293T cells were transfected with Vector control or DUSP2-FLAG. Cell lysates were subjected to immunoblotting using the anti-DUSP2 antibody (left) or immunoprecipatated with anti-DUSP2, followed by immunoblot analysis with anti-FLAG (right). Data are representative of two independent experiments (b).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Tables 1 and 2 (PDF 5721 kb)
Source data
Rights and permissions
About this article
Cite this article
Lu, D., Liu, L., Ji, X. et al. The phosphatase DUSP2 controls the activity of the transcription activator STAT3 and regulates TH17 differentiation. Nat Immunol 16, 1263–1273 (2015). https://doi.org/10.1038/ni.3278
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3278
This article is cited by
-
Bioinformatic analysis and experimental validation of the potential gene in the airway inflammation of steroid-resistant asthma
Scientific Reports (2023)
-
Novel targets for immunotherapy associated with exhausted CD8 + T cells in cancer
Journal of Cancer Research and Clinical Oncology (2023)
-
Resilient T-cell responses in patients with advanced cancers
International Journal of Hematology (2023)
-
Gastric cancer-derived exosomal miR-519a-3p promotes liver metastasis by inducing intrahepatic M2-like macrophage-mediated angiogenesis
Journal of Experimental & Clinical Cancer Research (2022)
-
Targeting protein phosphatases for the treatment of inflammation-related diseases: From signaling to therapy
Signal Transduction and Targeted Therapy (2022)