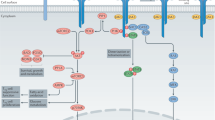
Abstract
The outcomes of clinical trials provide the most convincing data to clarify the role of particular cytokines in the pathogenesis of human diseases. The immunology community, for a variety of practical reasons, spends most of its research time and funds on studies in model systems, mainly mice. In this perspective I discuss results of clinical trials assessing the effect of blocking the differentiation and/or function of interleukin-17–producing CD4+ T cells on human autoimmune disease, and devote more limited attention to corroborating preclinical studies from animal models. Thus far, these outcomes in human trials have been mixed, with notable success in psoriasis and Crohn's disease but a negative result in relapsing-remitting multiple sclerosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Katie Vicari

Katie Vicari
Similar content being viewed by others
References
Feldmann, M. & Steinman, L. Design of effective immunotherapy for human autoimmunity. Nature 435, 612–619 (2005).
Nishimoto, N. & Kishimoto, T. Humanized antihuman IL-6 receptor antibody, tocilizumab. Handb. Exp. Pharmacol. 181, 151–160 (2008).
Church, L.D., Cook, G.P. & McDermott, M.F. Primer: inflammosomes and interleukin 1β in inflammatory disorders. Nat. Clin. Pract. Rheumatol. 4, 34–42 (2008).
Boniface, K., Blom, B., Liu, Y. & Malefyt R.d.W. From interleukin 23 to T-helper 17 cells: human T-helper cell differentiation revisited. Immunol. Rev. 226, 132–146 (2008).
Steinman, L. A brief history of TH17, the first major revision in the TH1/TH2 hypothesis of T cell–mediated tissue damage. Nat. Med. 13, 139–145 (2007).
Billiau, A. et al. Enhancement of experimental allergic encephalomyelitis in mice by antibodies against IFN-gamma. J. Immunol. 140, 1506–1510 (1988).
Ferber, I.A. et al. Mice with a disrupted interferon-γ gene are susceptible to the induction of experimental autoimmune encephalomyelitis (EAE). J. Immunol. 156, 5–7 (1996).
Steinman, L. A rush to judgment on Th17. J. Exp. Med. 205, 1517–1522 (2008).
Kroenke, M.A., Carlson, T.J., Adnjelkovic, A.V. & Segal, B.M. IL12– and IL-23–modulated T cells induce distinct types of EAE based on histology, CNS chemokine profile and response to cytokine inhibition. J. Exp. Med. 205, 1535–1541 (2008).
Yang, Y. et al. T-bet is essential for encephalitogenicity of both Th1 and Th17 cells. J. Exp. Med. 206, 1549–1564 (2009).
Lafaille, J.J. et al. Myelin basic protein–specific T helper 2 (Th2) cells cause experimental autoimmune encephalomyelitis in immunodeficient hosts rather than protect them from the disease. J. Exp. Med. 186, 307–312 (1997).
Annunziato, F. et al. Phenotypic and functional features of human Th17 cells. J. Exp. Med. 204, 1849–1861 (2007).
Lock, C. et al. Gene microarray analysis of multiple sclerosis lesions yields new targets validated in autoimmune encephalomyelitis. Nat. Med. 8, 500–508 (2002).
Li, Y. et al. Increased IL-23p19 expression in multiple sclerosis lesions and its induction in microglia. Brain 130, 490–501 (2007).
Cua, D.J. et al. Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature 421, 744–748 (2003).
't Hart, B.A. et al. Suppression of ongoing disease in a nonhuman primate model of multiple sclerosis by a human-anti-human IL-12p40 antibody. J. Immunol. 175, 4761–4768 (2005).
Segal, B.M. et al. Repeated subcutaneous injections of IL12/23 p40 neutralising antibody, ustekinumab, in patients with relapsing-remitting multiple sclerosis: a phase II, double-blind, placebo-controlled, randomised, dose-ranging study. Lancet Neurol. 7, 796–804 (2008).
Longbrake, E.E. & Racke M. Why did IL-12/IL-23 antibody therapy fail in multiple sclerosis? Expert Rev. Neurother. 9, 319–321 (2009).
Steinman, L. Multiple sclerosis: a two-stage disease. Nat. Immunol. 2, 762–765 (2001).
Steinman, L. Blocking adhesion molecules as therapy for multiple sclerosis: natalizumab. Nat. Rev. Drug Discov. 4, 510–518 (2005).
Polman, C.H. et al. A randomized, placebo-controlled trial of natalizumab for relapsing multiple sclerosis. N. Engl. J. Med. 354, 899–910 (2006).
Lenercept Multiple Sclerosis Study Group & University of British Columbia MS/MRI Analysis Group. TNF neutralization in MS: results of a randomized, placebo-controlled multicenter study. Neurology 53, 457–465 (1999).
van Oosten, B.W. et al. Increased MRI activity and immune activation in two multiple sclerosis patients treated with the monoclonal antitumor necrosis factor antibody cA2. Neurology 47, 1531–1534 (1996).
Krueger, G.G. et al. A human interleukin-12/23 monoclonal antibody for the treatment of psoriasis. N. Engl. J. Med. 356, 580–592 (2007).
Kimball, A.B. et al. Safety and efficacy of ABT-874, a fully human interleukin 12/23 monoclonal antibody, in the treatment of moderate to severe chronic plaque psoriasis. Results of a randomized, placebo-controlled, phase 2 trial. Arch. Dermatol. 144, 200–207 (2008).
Chan, J.R. et al. IL-23 stimulates epidermal hyperplasia via TNF and IL-20R2-dependent mechanisms with implications for psoriasis pathogenesis. J. Exp. Med. 203, 2577–2587 (2006).
Lee, E. et al. Increased expression of interleukin 23 p19 and p40 in lesional skin of patients with psoriasis vulgaris. J. Exp. Med. 199, 125–130 (2004).
Capon, F. et al. Sequence variants in the genes for the interleukin-23 receptor (IL23R) and its ligand (IL12B) confer protection against psoriasis. Hum. Genet. 122, 201–206 (2007).
Cargill, M. et al. A large-scale genetic association study confirms IL12B and leads to the identification of IL23R as psoriasis-risk genes. Am. J. Hum. Genet. 80, 273–290 (2007).
Duerr, R.H. et al. A genome-wide association study identifies IL23R as an inflammatory bowel disease gene. Science 314, 1461–1463 (2006).
Holtta, V. et al. IL-23/IL-17 immunity as a hallmark of Crohn's disease. Inflamm. Bowel Dis. 14, 1175–1184 (2008).
Pene, J. et al. Chronically inflamed tissues are infiltrated by highly differentiated TH17 lymphocytes. J. Immunol. 180, 7423–7430 (2008).
Mannon, P.J. et al. Anti–interleukin-12 antibody for active Crohn's disease. N. Engl. J. Med. 351, 2069–2079 (2004).
Sandborn, W.J. et al. A randomized trial of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with moderate-to-severe Crohn's disease. Gastroenterology 135, 1130–1141 (2008).
Genovese, M.C., Van den Bosch, F., Roberson, S.A. & Sloan-Lancaster, J. Safety, tolerability and evidence of efficacy of intravenous LY2439821 in patients with rheumatoid arthritis receiving background oral DMARDs. Ann. Rheum. Dis. 68 (suppl. 3), abstract OP-1060 (2009).
Durez, P. et al. AIN457, an anti-IL-17 antibody, shows good safety and induces clinical responses in patients with active rheumatoid arthritis (RA) despite methotrexate therapy in a randomized, double-blind proof-of-concept trial. Ann. Rheum. Dis. 68 (suppl. 3), abstract LB002 (2009).
Panitch, H.S., Hirsch, R.L., Schindler, J. & Johnson, K.P. Treatment of multiple sclerosis with gamma interferon: exacerbations associated with activation of the immune system. Neurology 37, 1097–1102 (1987).
Platten, M. et al. Blocking angiotensin converting enzyme induces potent regulatory T cells and modulates TH1 and TH17-mediated autoimmunity. Proc. Natl. Acad. Sci. USA 106, 14948–14953 (2009).
Stegbauer, J. et al. Role of the renin-angiotensin system in autoimmune inflammation of the central nervous system. Proc. Natl. Acad. Sci. USA 106, 14942–14947 (2009); doi:10.1073/pnas.0903602106.
Schulze-Topphoff, U. et al. Activation of kinin receptor B1 limits encephalitogenic T lymphocyte recruitment to the central nervous system. Nat. Med. 15, 788–793 (2009).
Steinman, L. A molecular trio in relapse and remission for multiple sclerosis. Nat. Rev. Immunol. 9, 440–447 (2009).
Molloy, E.S. & Calabrese, L. Therapy targeted but not trouble free: efalizumab and PML. Nat. Rev. Rheumatol. 5, 418–419 (2009).
Han, M.H. et al. Proteomic analysis of active multiple sclerosis lesions reveals therapeutic targets. Nature 451, 1076–1081 (2008).
Yao, C. et al. Prostaglandin E2–EP4 signaling promotes immune inflammation through TH1 cell differentiation and TH17 cell expansion. Nat. Med. 15, 633–641 (2009).
Shinohara, M.L., Kim, J.-H., Garcia, V.A. & Cantor, H. Engagement of the type I interferon receptor on dendritic cells inhibits promotion of Th17 cells: role of intracellular osteopontin. Immunity 29, 68–78 (2008).
Murugaiyan, G., Mittal, A. & Weiner, H.L. Increased osteopontin expression in dendritic cells amplifies IL-17 production by CD4+ T cells in experimental autoimmune encephalomyelitis and in multiple sclerosis. J. Immunol. 181, 7480–7488 (2008).
Vogt, M.H., Lopatinskaya, L., Smits, M., Polman, C.H. & Nagelkerken, L. Elevated osteopontin levels in active relapsing-remitting multiple sclerosis. Ann. Neurol. 53, 819–822 (2003).
Comabella, M. et al. Plasma osteopontin levels in multiple sclerosis. J. Neuroimmunol. 158, 231–239 (2005).
Hur, E. et al. Osteopontin induced relapse and progression of autoimmune brain disease via enhanced survival of activated T cells. Nat. Immunol. 8, 74–83 (2007).
Chen, M. Regulatory effects of interferon-beta on production of osteopontin and IL-17 in CD4 T cells in multiple sclerosis. Eur. J. Immunol. 39, 2525–2536 (2009).
Steinman, L. Shifting therapeutic attention in multiple sclerosis to osteopontin, type 1 and type 2 IFN. Eur. J. Immunol. 39, 2358–2360 (2009).
Gaffen, S.L. Structure and signalling in the IL-17 receptor family. Nat. Rev. Immunol. 9, 556–567 (2009).
Bettelli, E., Oukka, M. & Kuchroo, V.K. TH-17 cells in the circle of immunity and autoimmunity. Nat. Immunol. 8, 345–350 (2007).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Steinman, L. Mixed results with modulation of TH-17 cells in human autoimmune diseases. Nat Immunol 11, 41–44 (2010). https://doi.org/10.1038/ni.1803
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1803
This article is cited by
-
Inflammatory milieu of muscle biopsies in juvenile dermatomyositis
Rheumatology International (2021)
-
Combined Treatment with Methylprednisolone and Human Bone Marrow-Derived Mesenchymal Stem Cells Ameliorate Experimental Autoimmune Encephalomyelitis
Tissue Engineering and Regenerative Medicine (2018)
-
The Genetic Architecture of Alopecia Areata
Journal of Investigative Dermatology Symposium Proceedings (2013)
-
Diversity of IL-17-producing T lymphocytes
Cellular and Molecular Life Sciences (2013)