Abstract
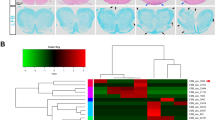
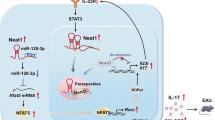
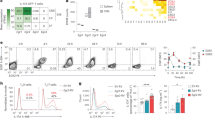
Interleukin 17 (IL-17)-producing T helper cells (TH-17 cells) are increasingly recognized as key participants in various autoimmune diseases, including multiple sclerosis. Although sets of transcription factors and cytokines are known to regulate TH-17 differentiation, the role of noncoding RNA is poorly understood. Here we identify a TH-17 cell–associated microRNA, miR-326, whose expression was highly correlated with disease severity in patients with multiple sclerosis and mice with experimental autoimmune encephalomyelitis (EAE). In vivo silencing of miR-326 resulted in fewer TH-17 cells and mild EAE, and its overexpression led to more TH-17 cells and severe EAE. We also found that miR-326 promoted TH-17 differentiation by targeting Ets-1, a negative regulator of TH-17 differentiation. Our data show a critical role for microRNA in TH-17 differentiation and the pathogenesis of multiple sclerosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
04 December 2009
In the version of this article initially published, the Actin loading control blot for the top blot is missing from Figure 5c. The error has been corrected in the HTML and PDF versions of the article. In the version of the ONLINE METHODS originally posted online, the description of the lentiviral vector was incorrect. The text in the left column, line 28, should read "...was cloned, downstream of the cytomegalovirus promoter, into a modified lentiviral vector generated from pCDH-CMVMCS-EF1-copGFP (CD511B-1; System Bioscience), in which EF1-copGFP was replaced with IRES-eGFP." The error has been corrected in the HTML and PDF versions of the article.
References
Korn, T., Bettelli, E., Oukka, M. & Kuchroo, V.K. IL-17 and Th17 cells. Annu. Rev. Immunol. 27, 485–517 (2009).
Dong, C. TH17 cells in development: an updated view of their molecular identity and genetic programming. Nat. Rev. Immunol. 8, 337–348 (2008).
Zhou, L. & Littman, D.R. Transcriptional regulatory networks in Th17 cell differentiation. Curr. Opin. Immunol. 21, 146–152 (2009).
Veldhoen, M. et al. The aryl hydrocarbon receptor links TH17-cell-mediated autoimmunity to environmental toxins. Nature 453, 106–109 (2008).
Brustle, A. et al. The development of inflammatory TH-17 cells requires interferon-regulatory factor 4. Nat. Immunol. 8, 958–966 (2007).
Zhou, L. et al. TGF-β-induced Foxp3 inhibits TH17 cell differentiation by antagonizing RORγt function. Nature 453, 236–240 (2008).
Moisan, J., Grenningloh, R., Bettelli, E., Oukka, M. & Ho, I.C. Ets-1 is a negative regulator of Th17 differentiation. J. Exp. Med. 204, 2825–2835 (2007).
Compston, A. & Coles, A. Multiple sclerosis. Lancet 359, 1221–1231 (2002).
Frohman, E.M., Racke, M.K. & Raine, C.S. Multiple sclerosis–the plaque and its pathogenesis. N. Engl. J. Med. 354, 942–955 (2006).
Kantarci, O.H. Genetics and natural history of multiple sclerosis. Semin. Neurol. 28, 7–16 (2008).
Polman, C.H. et al. Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria”. Ann. Neurol. 58, 840–846 (2005).
Sospedra, M. & Martin, R. Immunology of multiple sclerosis. Annu. Rev. Immunol. 23, 683–747 (2005).
Pettinelli, C.B. & McFarlin, D.E. Adoptive transfer of experimental allergic encephalomyelitis in SJL/J mice after in vitro activation of lymph node cells by myelin basic protein: requirement for Lyt1+2− T lymphocytes. J. Immunol. 127, 1420–1423 (1981).
Langrish, C.L. et al. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J. Exp. Med. 201, 233–240 (2005).
Ivanov, I.I. et al. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 126, 1121–1133 (2006).
Tzartos, J.S. et al. Interleukin-17 production in central nervous system-infiltrating T cells and glial cells is associated with active disease in multiple sclerosis. Am. J. Pathol. 172, 146–155 (2008).
Bartel, D.P. MicroRNAs: target recognition and regulatory functions. Cell 136, 215–233 (2009).
Ma, L., Teruya-Feldstein, J. & Weinberg, R.A. Tumour invasion and metastasis initiated by microRNA-10b in breast cancer. Nature 449, 682–688 (2007).
Thum, T. et al. MicroRNA-21 contributes to myocardial disease by stimulating MAP kinase signalling in fibroblasts. Nature 456, 980–984 (2008).
Lu, J. et al. MicroRNA expression profiles classify human cancers. Nature 435, 834–838 (2005).
Slack, F.J. & Weidhaas, J.B. MicroRNA in cancer prognosis. N. Engl. J. Med. 359, 2720–2722 (2008).
Zhou, X. et al. Selective miRNA disruption in T reg cells leads to uncontrolled autoimmunity. J. Exp. Med. 205, 1983–1991 (2008).
Chong, M.M., Rasmussen, J.P., Rudensky, A.Y. & Littman, D.R. The RNAseIII enzyme Drosha is critical in T cells for preventing lethal inflammatory disease. J. Exp. Med. 205, 2005–2017 (2008).
Wingerchuk, D.M., Lennon, V.A., Lucchinetti, C.F., Pittock, S.J. & Weinshenker, B.G. The spectrum of neuromyelitis optica. Lancet Neurol. 6, 805–815 (2007).
Acosta-Rodriguez, E.V. et al. Surface phenotype and antigenic specificity of human interleukin 17-producing T helper memory cells. Nat. Immunol. 8, 639–646 (2007).
Ebert, M.S., Neilson, J.R. & Sharp, P.A. MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells. Nat. Methods 4, 721–726 (2007).
Pan, D. et al. Biodistribution and toxicity studies of VSVG-pseudotyped lentiviral vector after intravenous administration in mice with the observation of in vivo transduction of bone marrow. Mol. Ther. 6, 19–29 (2002).
Lewis, B.P., Shih, I.H., Jones-Rhoades, M.W., Bartel, D.P. & Burge, C.B. Prediction of mammalian microRNA targets. Cell 115, 787–798 (2003).
John, B., Sander, C. & Marks, D.S. Prediction of human microRNA targets. Methods Mol. Biol. 342, 101–113 (2006).
Tesmer, L.A., Lundy, S.K., Sarkar, S. & Fox, D.A. Th17 cells in human disease. Immunol. Rev. 223, 87–113 (2008).
Miossec, P., Korn, T. & Kuchroo, V.K. Interleukin-17 and type 17 helper T cells. N. Engl. J. Med. 361, 888–898 (2009).
Lennon, V.A. et al. A serum autoantibody marker of neuromyelitis optica: distinction from multiple sclerosis. Lancet 364, 2106–2112 (2004).
Xiao, C. & Rajewsky, K. MicroRNA control in the immune system: basic principles. Cell 136, 26–36 (2009).
Hoefig, K.P. & Heissmeyer, V. MicroRNAs grow up in the immune system. Curr. Opin. Immunol. 20, 281–287 (2008).
Li, Q.J. et al. miR-181a is an intrinsic modulator of T cell sensitivity and selection. Cell 129, 147–161 (2007).
Lin, Y.C. et al. c-Myb is an evolutionary conserved miR-150 target and miR-150/c-Myb interaction is important for embryonic development. Mol. Biol. Evol. 25, 2189–2198 (2008).
Xiao, C. et al. MiR-150 controls B cell differentiation by targeting the transcription factor c-Myb. Cell 131, 146–159 (2007).
Schrempf, W. & Ziemssen, T. Glatiramer acetate: mechanisms of action in multiple sclerosis. Autoimmun. Rev. 6, 469–475 (2007).
Brusaferri, F. & Candelise, L. Steroids for multiple sclerosis and optic neuritis: a meta-analysis of randomized controlled clinical trials. J. Neurol. 247, 435–442 (2000).
Stromnes, I.M. & Goverman, J.M. Active induction of experimental allergic encephalomyelitis. Nat. Protoc. 1, 1810–1819 (2006).
Chen, J.F. et al. The role of microRNA-1 and microRNA-133 in skeletal muscle proliferation and differentiation. Nat. Genet. 38, 228–233 (2006).
Acknowledgements
We thank C. Dong (Anderson Cancer Center, Houston) for retrovector RV-GFP; X. Liu (Shanghai Institute for Biological Sciences) for fluorescein isothiocyanate–conjugated antibody to mouse T cell antigen receptor γδ; C. Wu (Sun Yat-sen University) for phycoerythrin-conjugated antibody to human CCR6; J. Zhao, L. Wei, Y. Shi, Q. Jing and G. Gao for discussions; and J. Zou, S. Xin, X. Zeng and S. Chen for technical assistance. Supported by the Ministry of Science and Technology (2005CB522406 and 2009CB941100), the National Natural Science Foundation of China (30621091, 30625014, 30623003, 30871285, 90713047 and 90919028), the Shanghai Municipal Commission for Science and Technology (07PJ14099) and the Chinese Academy of Sciences (2007KIP204 and SIBS2008001).
Author information
Authors and Affiliations
Contributions
C.D., C.L., J.K. and G.P. designed the study; C.D., C.L. and S.H. did the experiments; Z.Y. contributed to the bioinformatics analysis target prediction; Z.W., Z.L. and G.Z. provided clinical samples; G.P. supervised the project; and C.L., C.D. and J.K. contributed to the writing of the paper.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–14 and Supplementary Methods (PDF 1271 kb)
Rights and permissions
About this article
Cite this article
Du, C., Liu, C., Kang, J. et al. MicroRNA miR-326 regulates TH-17 differentiation and is associated with the pathogenesis of multiple sclerosis. Nat Immunol 10, 1252–1259 (2009). https://doi.org/10.1038/ni.1798
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1798
This article is cited by
-
MicroRNAs expression in peripheral blood mononuclear cells of patients with multiple sclerosis propose
Molecular Biology Reports (2023)
-
MiR-326 regulates cell proliferation and apoptosis in fibroblast-like synoviocytes in rheumatoid arthritis
Human Cell (2023)
-
MicroRNAs: a crossroad that connects obesity to immunity and aging
Immunity & Ageing (2022)
-
Association between miRNA-145 and miRNA-155 expression in peripheral blood mononuclear cells of patients with multiple sclerosis: a case-control study
BMC Neurology (2022)
-
CNS microRNA profiles: a database for cell type enriched microRNA expression across the mouse central nervous system
Scientific Reports (2020)