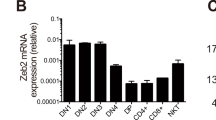
Abstract
The transcription factors GATA-3 and ThPOK are required for intrathymic differentiation of CD4+ T cells, but their precise functions in this process remain unclear. Here we show that, contrary to previous findings, Gata3 disruption blocked differentiation into the CD4+ T cell lineage before commitment to the CD4+ lineage and in some contexts permitted the 'redirection' of major histocompatibility complex class II–restricted thymocytes into the CD8+ lineage. GATA-3 promoted ThPOK expression and bound to a region of the locus encoding ThPOK established as being critical for ThPOK expression. Finally, ThPOK promoted differentiation into the CD4+ lineage in a way dependent on GATA-3 but inhibited differentiation into the CD8+ lineage independently of GATA-3. We propose that GATA-3 acts as a specification factor for the CD4+ lineage 'upstream' of the ThPOK-controlled CD4+ commitment checkpoint.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Starr, T.K., Jameson, S.C. & Hogquist, K.A. Positive and negative selection of T cells. Annu. Rev. Immunol. 21, 139–176 (2003).
Bosselut, R. CD4/CD8-lineage differentiation in the thymus: from nuclear effectors to membrane signals. Nat. Rev. Immunol. 4, 529–540 (2004).
Laky, K. & Fowlkes, B. Receptor signals and nuclear events in CD4 and CD8 T cell lineage commitment. Curr. Opin. Immunol. 17, 116–121 (2005).
He, X. et al. The zinc finger transcription factor Th-POK regulates CD4 versus CD8 T-cell lineage commitment. Nature 433, 826–833 (2005).
Sun, G. et al. The zinc finger protein cKrox directs CD4 lineage differentiation during intrathymic T cell positive selection. Nat. Immunol. 6, 373–381 (2005).
Bilic, I. & Ellmeier, W. The role of BTB domain-containing zinc finger proteins in T cell development and function. Immunol. Lett. 108, 1–9 (2007).
Ting, C.N., Olson, M.C., Barton, K.P. & Leiden, J.M. Transcription factor GATA-3 is required for development of the T-cell lineage. Nature 384, 474–478 (1996).
Zhu, J., Yamane, H., Cote-Sierra, J., Guo, L. & Paul, W.E. GATA-3 promotes Th2 responses through three different mechanisms: induction of Th2 cytokine production, selective growth of Th2 cells and inhibition of Th1 cell-specific factors. Cell Res. 16, 3–10 (2006).
Ho, I.C. & Pai, S.Y. GATA-3—not just for Th2 cells anymore. Cell. Mol. Immunol. 4, 15–29 (2007).
Hendriks, R.W. et al. Expression of the transcription factor GATA-3 is required for the development of the earliest T cell progenitors and correlates with stages of cellular proliferation in the thymus. Eur. J. Immunol. 29, 1912–1918 (1999).
Hernandez-Hoyos, G., Anderson, M.K., Wang, C., Rothenberg, E.V. & Alberola-Ila, J. GATA-3 expression is controlled by TCR signals and regulates CD4/CD8 differentiation. Immunity 19, 83–94 (2003).
Pai, S.Y. et al. Critical roles for transcription factor GATA-3 in thymocyte development. Immunity 19, 863–875 (2003).
Zhu, J. et al. Conditional deletion of Gata3 shows its essential function in TH1-TH2 responses. Nat. Immunol. 5, 1157–1165 (2004).
Ling, K.W. et al. GATA3 controls the expression of CD5 and the T cell receptor during CD4 T cell lineage development. Eur. J. Immunol. 37, 1043–1052 (2007).
Wolfe, S.A., Nekludova, L. & Pabo, C.O. DNA recognition by Cys2His2 zinc finger proteins. Annu. Rev. Biophys. Biomol. Struct. 29, 183–212 (2000).
Galera, P., Musso, M., Ducy, P. & Karsenty, G. c-Krox, a transcriptional regulator of type I collagen gene expression, is preferentially expressed in skin. Proc. Natl. Acad. Sci. USA 91, 9372–9376 (1994).
Seder, R.A., Paul, W.E., Davis, M.M. & Fazekas de St Groth, B. The presence of interleukin 4 during in vitro priming determines the lymphokine-producing potential of CD4+ T cells from T cell receptor transgenic mice. J. Exp. Med. 176, 1091–1098 (1992).
Rothenberg, E.V. Stepwise specification of lymphocyte developmental lineages. Curr. Opin. Genet. Dev. 10, 370–379 (2000).
Ko, L.J. & Engel, J.D. DNA-binding specificities of the GATA transcription factor family. Mol. Cell. Biol. 13, 4011–4022 (1993).
Merika, M. & Orkin, S.H. Functional synergy and physical interactions of the erythroid transcription factor GATA-1 with the Kruppel family proteins Sp1 and EKLF. Mol. Cell. Biol. 15, 2437–2447 (1995).
He, X. et al. CD4–CD8 lineage commitment is regulated by a silencer element at the ThPOK transcription-factor locus. Immunity 28, 346–358 (2008).
Setoguchi, R. et al. Repression of the transcription factor Th-POK by Runx complexes in cytotoxic T cell development. Science 319, 822–825 (2008).
Ellmeier, W., Sunshine, M.J., Losos, K., Hatam, F. & Littman, D.R. An enhancer that directs lineage-specific expression of CD8 in positively selected thymocytes and mature T cells. Immunity 7, 537–547 (1997).
Hostert, A. et al. A region in the CD8 gene locus that directs expression to the mature CD8 T cell subset in transgenic mice. Immunity 7, 525–536 (1997).
Aliahmad, P. & Kaye, J. Development of all CD4 T lineages requires nuclear factor TOX. J. Exp. Med. 205, 245–256 (2008).
Bender, T.P., Kremer, C.S., Kraus, M., Buch, T. & Rajewsky, K. Critical functions for c-Myb at three checkpoints during thymocyte development. Nat. Immunol. 5, 721–729 (2004).
Maurice, D., Hooper, J., Lang, G. & Weston, K. c-Myb regulates lineage choice in developing thymocytes via its target gene Gata3. EMBO J. 26, 3629–3640 (2007).
Busslinger, M. Transcriptional control of early B cell development. Annu. Rev. Immunol. 22, 55–79 (2004).
Hagman, J. & Lukin, K. Transcription factors drive B cell development. Curr. Opin. Immunol. 18, 127–134 (2006).
Brugnera, E. et al. Coreceptor reversal in the thymus: signaled CD4+8+ thymocytes initially terminate CD8 transcription even when differentiating into CD8+ T cells. Immunity 13, 59–71 (2000).
Yasutomo, K., Doyle, C., Miele, L., Fuchs, C. & Germain, R.N. The duration of antigen receptor signalling determines CD4+ versus CD8+ T-cell lineage fate. Nature 404, 506–510 (2000).
Liu, X. & Bosselut, R. Duration of TCR signaling controls CD4–CD8 lineage differentiation in vivo. Nat. Immunol. 5, 280–288 (2004).
Itano, A. et al. The cytoplasmic domain of CD4 promotes the development of CD4 lineage T cells. J. Exp. Med. 183, 731–741 (1996).
Hernandez-Hoyos, G., Sohn, S.J., Rothenberg, E.V. & Alberola-Ila, J. Lck activity controls CD4/CD8 T cell lineage commitment. Immunity 12, 313–322 (2000).
Azzam, H.S. et al. Fine tuning of TCR signaling by CD5. J. Immunol. 166, 5464–5472 (2001).
Nawijn, M.C. et al. Enforced expression of GATA-3 during T cell development inhibits maturation of CD8 single-positive cells and induces thymic lymphoma in transgenic mice. J. Immunol. 167, 715–723 (2001).
Atherly, L.O. et al. The Tec family tyrosine kinases Itk and Rlk regulate the development of conventional CD8+ T cells. Immunity 25, 79–91 (2006).
Dubois, S., Waldmann, T.A. & Muller, J.R. ITK and IL-15 support two distinct subsets of CD8+ T cells. Proc. Natl. Acad. Sci. USA 103, 12075–12080 (2006).
Horai, R. et al. Requirements for selection of conventional and innate T lymphocyte lineages. Immunity 27, 775–785 (2007).
Yu, Q., Erman, B., Bhandoola, A., Sharrow, S.O. & Singer, A. In vitro evidence that cytokine receptor signals are required for differentiation of double positive thymocytes into functionally mature CD8+ T cells. J. Exp. Med. 197, 475–487 (2003).
Taniuchi, I. et al. Differential requirements for Runx proteins in CD4 repression and epigenetic silencing during T lymphocyte development. Cell 111, 621–633 (2002).
Woolf, E. et al. Runx3 and Runx1 are required for CD8 T cell development during thymopoiesis. Proc. Natl. Acad. Sci. USA 100, 7731–7736 (2003).
Jenkinson, S.R. et al. Expression of the transcription factor cKrox in peripheral CD8 T cells reveals substantial postthymic plasticity in CD4–CD8 lineage differentiation. J. Exp. Med. 204, 267–272 (2007).
Wildt, K.F. et al. The transcription factor Zbtb7b promotes CD4 expression by antagonizing Runx-mediated activation of the CD4 silencer. J. Immunol. 179, 4405–4414 (2007).
Egawa, T., Tillman, R.E., Naoe, Y., Taniuchi, I. & Littman, D.R. The role of the Runx transcription factors in thymocyte differentiation and in homeostasis of naive T cells. J. Exp. Med. 204, 1945–1957 (2007).
Ma, W. et al. Hepatic vascular tumors, angiectasis in multiple organs, and impaired spermatogenesis in mice with conditional inactivation of the VHL gene. Cancer Res. 63, 5320–5328 (2003).
Lee, P.P. et al. A critical role for Dnmt1 and DNA methylation in T cell development, function, and survival. Immunity 15, 763–774 (2001).
Kaye, J. et al. Selective development of CD4+ T cells in transgenic mice expressing a class II MHC-restricted antigen receptor. Nature 341, 746–749 (1989).
Liu, X. et al. Restricting Zap70 expression to CD4+CD8+ thymocytes reveals a T cell receptor-dependent proofreading mechanism controlling the completion of positive selection. J. Exp. Med. 197, 363–373 (2003).
Swat, W. & Dessing, M. von, B. H. & Kisielow, P. CD69 expression during selection and maturation of CD4+8+ thymocytes. Eur. J. Immunol. 23, 739–746 (1993).
Acknowledgements
We thank Y. Xiong, R. Jenkinson and J. Rozenberg for assistance with ChIP assays; E. Southon for embryonic stem cell recombination; M. Pellegrini and A. Nussenzweig for assistance with recombination-mediated genetic engineering; E. Castro and G. Sanchez for mouse technical assistance; B. Taylor and S. Banerjee for cell sorting; and A. Gégonne and A. Singer for reading the manuscript. Supported by the Intramural Research Programs of the National Cancer Institute, Center for Cancer Research, and of the National Institute of Allergy and Infectious Diseases, National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figure
Supplementary Figures 1–11, Tables 1–3 and Methods (PDF 4474 kb)
Rights and permissions
About this article
Cite this article
Wang, L., Wildt, K., Zhu, J. et al. Distinct functions for the transcription factors GATA-3 and ThPOK during intrathymic differentiation of CD4+ T cells. Nat Immunol 9, 1122–1130 (2008). https://doi.org/10.1038/ni.1647
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1647
This article is cited by
-
Markers and makers of NKT17 cells
Experimental & Molecular Medicine (2023)
-
Reversal of the T cell immune system reveals the molecular basis for T cell lineage fate determination in the thymus
Nature Immunology (2022)
-
TCF-1: a maverick in T cell development and function
Nature Immunology (2022)
-
TCF1 in T cell immunity: a broadened frontier
Nature Reviews Immunology (2022)
-
GATA-3 is a proto-oncogene in T-cell lymphoproliferative neoplasms
Blood Cancer Journal (2022)