Abstract
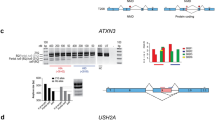
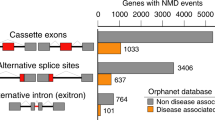
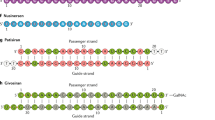
We describe a new technology for recruiting specific proteins to RNA through selective recognition of heteroduplexes formed with chemically modified antisense oligonucleotides (ASOs). Typically, ASOs function by hybridizing to their RNA targets and blocking the binding of single-stranded RNA–binding proteins. Unexpectedly, we found that ASOs with 2′-deoxy-2′-fluoro (2′-F) nucleotides, but not with other 2′ chemical modifications, have an additional property: they form heteroduplexes with RNA that are specifically recognized by the interleukin enhancer-binding factor 2 and 3 complex (ILF2/3). 2′-F ASO–directed recruitment of ILF2/3 to RNA can be harnessed to control gene expression by modulating alternative splicing of target transcripts. ILF2/3 recruitment to precursor mRNA near an exon results in omission of the exon from the mature mRNA, both in cell culture and in mice. We discuss the possibility of using chemically engineered ASOs that recruit specific proteins to modulate gene expression for therapeutic intervention.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Glisovic, T., Bachorik, J.L., Yong, J. & Dreyfuss, G. RNA-binding proteins and post-transcriptional gene regulation. FEBS Lett. 582, 1977–1986 (2008).
Licatalosi, D.D. & Darnell, R.B. RNA processing and its regulation: global insights into biological networks. Nat. Rev. Genet. 11, 75–87 (2010).
Calarco, J.A., Zhen, M. & Blencowe, B.J. Networking in a global world: establishing functional connections between neural splicing regulators and their target transcripts. RNA 17, 775–791 (2011).
Cooper, T.A., Wan, L. & Dreyfuss, G. RNA and disease. Cell 136, 777–793 (2009).
Lunde, B.M., Moore, C. & Varani, G. RNA-binding proteins: modular design for efficient function. Nat. Rev. Mol. Cell Biol. 8, 479–490 (2007).
Auweter, S.D., Oberstrass, F.C. & Allain, F.H. Sequence-specific binding of single-stranded RNA: is there a code for recognition? Nucleic Acids Res. 34, 4943–4959 (2006).
Tian, B., Bevilacqua, P.C., Diegelman-Parente, A. & Mathews, M.B. The double-stranded-RNA-binding motif: interference and much more. Nat. Rev. Mol. Cell Biol. 5, 1013–1023 (2004).
Wahl, M.C., Will, C.L. & Luhrmann, R. The spliceosome: design principles of a dynamic RNP machine. Cell 136, 701–718 (2009).
Wang, Y., Cheong, C.G., Hall, T.M. & Wang, Z. Engineering splicing factors with designed specificities. Nat. Methods 6, 825–830 (2009).
Mackay, J.P., Font, J. & Segal, D.J. The prospects for designer single-stranded RNA-binding proteins. Nat. Struct. Mol. Biol. 18, 256–261 (2011).
Culler, S.J., Hoff, K.G. & Smolke, C.D. Reprogramming cellular behavior with RNA controllers responsive to endogenous proteins. Science 330, 1251–1255 (2010).
Baughan, T.D., Dickson, A., Osman, E.Y. & Lorson, C.L. Delivery of bifunctional RNAs that target an intronic repressor and increase SMN levels in an animal model of spinal muscular atrophy. Hum. Mol. Genet. 18, 1600–1611 (2009).
Cartegni, L. & Krainer, A.R. Correction of disease-associated exon skipping by synthetic exon-specific activators. Nat. Struct. Biol. 10, 120–125 (2003).
Skordis, L.A., Dunckley, M.G., Yue, B., Eperon, I.C. & Muntoni, F. Bifunctional antisense oligonucleotides provide a trans-acting splicing enhancer that stimulates SMN2 gene expression in patient fibroblasts. Proc. Natl. Acad. Sci. USA 100, 4114–4119 (2003).
Villemaire, J., Dion, I., Elela, S.A. & Chabot, B. Reprogramming alternative pre-messenger RNA splicing through the use of protein-binding antisense oligonucleotides. J. Biol. Chem. 278, 50031–50039 (2003).
Goraczniak, R., Behlke, M.A. & Gunderson, S.I. Gene silencing by synthetic U1 adaptors. Nat. Biotechnol. 27, 257–263 (2009).
Bennett, C.F. & Swayze, E.E. RNA targeting therapeutics: molecular mechanisms of antisense oligonucleotides as a therapeutic platform. Annu. Rev. Pharmacol. Toxicol. 50, 259–293 (2010).
Bauman, J., Jearawiriyapaisarn, N. & Kole, R. Therapeutic potential of splice-switching oligonucleotides. Oligonucleotides 19, 1–13 (2009).
Bennett, C.F. et al. Antisense oligonucleotide-based therapeutics. in Gene and Cell Therapy: Therapeutic Mechanisms and Strategies (ed. Templeton, N.S.) 497–522 (Taylor & Francis Group, 2009).
Swayze, E.E. & Balkrishen, B. The medicinal chemistry of oligonucleotides. in Antisense Drug Technology: Principles, Strategies and Applications (ed. Crooke, S.T.) 143–182 (Taylor & Francis Group, 2008).
Seth, P.P. et al. Synthesis and biophysical evaluation of 2′,4′-constrained 2′O-methoxyethyl and 2′,4′-constrained 2′O-ethyl nucleic acid analogues. J. Org. Chem. 75, 1569–1581 (2010).
Levin, A.A., Yu, R.Z. & Geary, R.S. Basic principles of the pharmacokinetics of antisense oligonucleotide drugs. in Antisense Drug Technology: Principles, Strategies and Applications (ed. Crooke, S.T.) 183–215 (Taylor & Francis Group, 2008).
Chen, M. & Manley, J.L. Mechanisms of alternative splicing regulation: insights from molecular and genomics approaches. Nat. Rev. Mol. Cell Biol. 10, 741–754 (2009).
Hua, Y., Vickers, T.A., Okunola, H.L., Bennett, C.F. & Krainer, A.R. Antisense masking of an hnRNP A1/A2 intronic splicing silencer corrects SMN2 splicing in transgenic mice. Am. J. Hum. Genet. 82, 834–848 (2008).
Lorson, C.L., Hahnen, E., Androphy, E.J. & Wirth, B. A single nucleotide in the SMN gene regulates splicing and is responsible for spinal muscular atrophy. Proc. Natl. Acad. Sci. USA 96, 6307–6311 (1999).
Pandya-Jones, A. & Black, D.L. Co-transcriptional splicing of constitutive and alternative exons. RNA 15, 1896–1908 (2009).
Noguchi, T., Inoue, H. & Tanaka, T. The M1- and M2-type isozymes of rat pyruvate kinase are produced from the same gene by alternative RNA splicing. J. Biol. Chem. 261, 13807–13812 (1986).
Christofk, H.R. et al. The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumour growth. Nature 452, 230–233 (2008).
Boise, L.H. et al. bcl-x, a bcl-2–related gene that functions as a dominant regulator of apoptotic cell death. Cell 74, 597–608 (1993).
Goedert, M., Spillantini, M.G., Potier, M.C., Ulrich, J. & Crowther, R.A. Cloning and sequencing of the cDNA encoding an isoform of microtubule-associated protein tau containing four tandem repeats: differential expression of tau protein mRNAs in human brain. EMBO J. 8, 393–399 (1989).
Rademakers, R., Cruts, M. & van Broeckhoven, C. The role of tau (MAPT) in frontotemporal dementia and related tauopathies. Hum. Mutat. 24, 277–295 (2004).
Singh, N.K., Singh, N.N., Androphy, E.J. & Singh, R.N. Splicing of a critical exon of human Survival Motor Neuron is regulated by a unique silencer element located in the last intron. Mol. Cell. Biol. 26, 1333–1346 (2006).
Guan, D. et al. Nuclear factor 45 (NF45) is a regulatory subunit of complexes with NF90/110 involved in mitotic control. Mol. Cell. Biol. 28, 4629–4641 (2008).
Liao, H.J., Kobayashi, R. & Mathews, M.B. Activities of adenovirus virus-associated RNAs: purification and characterization of RNA binding proteins. Proc. Natl. Acad. Sci. USA 95, 8514–8519 (1998).
Kawasaki, A.M. et al. Uniformly modified 2′-deoxy-2′-fluoro phosphorothioate oligonucleotides as nuclease-resistant antisense compounds with high affinity and specificity for RNA targets. J. Med. Chem. 36, 831–841 (1993).
Teplova, M. et al. Crystal structure and improved antisense properties of 2′-O-(2-methoxyethyl)-RNA. Nat. Struct. Biol. 6, 535–539 (1999).
Li, F. et al. 2′-Fluoroarabino- and arabinonucleic acid show different conformations, resulting in deviating RNA affinities and processing of their heteroduplexes with RNA by RNase H. Biochemistry 45, 4141–4152 (2006).
Inoue, H. et al. Synthesis and hybridization studies on two complementary nona(2′-O-methyl)ribonucleotides. Nucleic Acids Res. 15, 6131–6148 (1987).
Wilds, C.J. & Damha, M.J. 2′-Deoxy-2′-fluoro-β-D-arabinonucleosides and oligonucleotides (2′F-ANA): synthesis and physicochemical studies. Nucleic Acids Res. 28, 3625–3635 (2000).
Vester, B. & Wengel, J. LNA (locked nucleic acid): high-affinity targeting of complementary RNA and DNA. Biochemistry 43, 13233–13241 (2004).
Jepsen, J.S., Sorensen, M.D. & Wengel, J. Locked nucleic acid: a potent nucleic acid analog in therapeutics and biotechnology. Oligonucleotides 14, 130–146 (2004).
Liu, J., Hu, J. & Corey, D.R. Expanding the action of duplex RNAs into the nucleus: redirecting alternative splicing. Nucleic Acids Res. 40, 1240–1250 (2011).
Cohen, J.S. Informational drugs: a new concept in pharmacology. Antisense Res. Dev. 1, 191–193 (1991).
Wang, G.S. & Cooper, T.A. Splicing in disease: disruption of the splicing code and the decoding machinery. Nat. Rev. Genet. 8, 749–761 (2007).
Barber, G.N. The NFAR's (nuclear factors associated with dsRNA): evolutionarily conserved members of the dsRNA binding protein family. RNA Biol. 6, 35–39 (2009).
Win, M.N., Liang, J.C. & Smolke, C.D. Frameworks for programming biological function through RNA parts and devices. Chem. Biol. 16, 298–310 (2009).
Hua, Y. et al. Antisense correction of SMN2 splicing in the CNS rescues necrosis in a type III SMA mouse model. Genes Dev. 24, 1634–1644 (2010).
Acknowledgements
We thank A. Berdeja for Tm measurements and E. Swayze, W. Lima and D. Corey for helpful discussions.
Author information
Authors and Affiliations
Contributions
F.R., Y.H., T.P.P., A.R.K. and C.F.B. conceived of and designed the experiments. F.R. and S.J.C. performed the experiments. F.R. and C.F.B. analyzed the data. T.P.P. synthesized the ASOs. F.R. and C.F.B. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
F.R., S.J.C., T.P.P. and C.F.B. are employees of ISIS Pharmaceuticals. A.R.K. serves on the scientific advisory board of two nonprofit spinal muscular atrophy foundations and is a consultant for ISIS Pharmaceuticals.
Supplementary information
Supplementary Text and Figures
Supplementary Methods and Supplementary Results (PDF 1587 kb)
Rights and permissions
About this article
Cite this article
Rigo, F., Hua, Y., Chun, S. et al. Synthetic oligonucleotides recruit ILF2/3 to RNA transcripts to modulate splicing. Nat Chem Biol 8, 555–561 (2012). https://doi.org/10.1038/nchembio.939
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.939
This article is cited by
-
Effects of combinations of gapmer antisense oligonucleotides on the target reduction
Molecular Biology Reports (2023)
-
An LNA-amide modification that enhances the cell uptake and activity of phosphorothioate exon-skipping oligonucleotides
Nature Communications (2022)
-
Systematic screening identifies therapeutic antisense oligonucleotides for Hutchinson–Gilford progeria syndrome
Nature Medicine (2021)
-
Systematic evaluation of 2′-Fluoro modified chimeric antisense oligonucleotide-mediated exon skipping in vitro
Scientific Reports (2019)
-
Non-coding RNAs as drug targets
Nature Reviews Drug Discovery (2017)