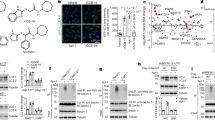
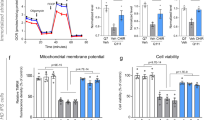
Abstract
A hallmark of many neurodegenerative diseases is accumulation of misfolded proteins within neurons, leading to cellular dysfunction and cell death. Although several mechanisms have been proposed to link protein misfolding to cellular toxicity, the connection remains enigmatic. Here, we report a cell death pathway involving protein disulfide isomerase (PDI), a protein chaperone that catalyzes isomerization, reduction and oxidation of disulfides. Through a small molecule screening approach, we discovered five structurally distinct compounds that prevent apoptosis induced by mutant huntingtin protein. Using modified Huisgen cycloaddition chemistry, we then identified PDI as the molecular target of these small molecules. Expression of polyglutamine-expanded huntingtin exon 1 in PC12 cells caused PDI to accumulate at mitochondrial-associated ER membranes and trigger apoptotic cell death via mitochondrial outer-membrane permeabilization. Inhibiting PDI in rat brain cells suppressed the toxicity of mutant huntingtin exon 1 and Aβ peptides processed from the amyloid precursor protein. This pro-apoptotic function of PDI represents a new mechanism linking protein misfolding and apoptotic cell death.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Gregersen, N. Protein misfolding disorders: pathogenesis and intervention. J. Inherit. Metab. Dis. 29, 456–470 (2006).
Orr, H.T. & Zoghbi, H.Y. Trinucleotide repeat disorders. Annu. Rev. Neurosci. 30, 575–621 (2007).
Hickey, M.A. & Chesselet, M.F. Apoptosis in Huntington's disease. Prog. Neuropsychopharmacol. Biol. Psychiatry 27, 255–265 (2003).
Thomas, L.B. et al. DNA end labeling (TUNEL) in Huntington's disease and other neuropathological conditions. Exp. Neurol. 133, 265–272 (1995).
Reddy, P.H. et al. Behavioural abnormalities and selective neuronal loss in HD transgenic mice expressing mutated full-length HD cDNA. Nat. Genet. 20, 198–202 (1998).
Hodgson, J.G. et al. A YAC mouse model for Huntington's disease with full-length mutant huntingtin, cytoplasmic toxicity, and selective striatal neurodegeneration. Neuron 23, 181–192 (1999).
Kiechle, T. et al. Cytochrome C and caspase-9 expression in Huntington's disease. Neuromolecular Med. 1, 183–195 (2002).
Yu, Z.X. et al. Mutant huntingtin causes context-dependent neurodegeneration in mice with Huntington's disease. J. Neurosci. 23, 2193–2202 (2003).
Ciammola, A. et al. Increased apoptosis, Huntingtin inclusions and altered differentiation in muscle cell cultures from Huntington's disease subjects. Cell Death Differ. 13, 2068–2078 (2006).
Bredesen, D.E., Rao, R.V. & Mehlen, P. Cell death in the nervous system. Nature 443, 796–802 (2006).
Chipuk, J.E. & Green, D.R. How do BCL-2 proteins induce mitochondrial outer membrane permeabilization? Trends Cell Biol. 18, 157–164 (2008).
Leber, B., Lin, J. & Andrews, D.W. Embedded together: the life and death consequences of interaction of the Bcl-2 family with membranes. Apoptosis 12, 897–911 (2007).
Jiang, X. & Wang, X. Cytochrome C-mediated apoptosis. Annu. Rev. Biochem. 73, 87–106 (2004).
Chipuk, J.E., Bouchier-Hayes, L. & Green, D.R. Mitochondrial outer membrane permeabilization during apoptosis: the innocent bystander scenario. Cell Death Differ. 13, 1396–1402 (2006).
Aiken, C.T., Tobin, A.J. & Schweitzer, E.S. A cell-based screen for drugs to treat Huntington's disease. Neurobiol. Dis. 16, 546–555 (2004).
Huisgen, R. in 1,3-Dipolar Cycloaddition Chemistry (ed. Padwa, A.) 1–176 (Wiley, New York, 1984).
Rostovtsev, V.V., Green, L.G., Fokin, V.V. & Sharpless, K.B. A stepwise huisgen cycloaddition process: copper(I)-catalyzed regioselective “ligation” of azides and terminal alkynes. Angew. Chem. Int. Edn Engl. 41, 2596–2599 (2002).
Tornøe, C.W., Christensen, C. & Meldal, M. Peptidotriazoles on solid phase: [1,2,3]-triazoles by regiospecific copper(i)-catalyzed 1,3-dipolar cycloadditions of terminal alkynes to azides. J. Org. Chem. 67, 3057–3064 (2002).
Wang, Q. et al. Bioconjugation by copper(I)-catalyzed azide-alkyne [3 + 2] cycloaddition. J. Am. Chem. Soc. 125, 3192–3193 (2003).
Speers, A.E. & Cravatt, B.F. Profiling enzyme activities in vivo using click chemistry methods. Chem. Biol. 11, 535–546 (2004).
Karpuj, M.V. et al. Prolonged survival and decreased abnormal movements in transgenic model of Huntington disease, with administration of the transglutaminase inhibitor cystamine. Nat. Med. 8, 143–149 (2002).
Dedeoglu, A. et al. Therapeutic effects of cystamine in a murine model of Huntington's disease. J. Neurosci. 22, 8942–8950 (2002).
Van Raamsdonk, J.M. et al. Cystamine treatment is neuroprotective in the YAC128 mouse model of Huntington disease. J. Neurochem. 95, 210–220 (2005).
Tremblay, M.E. et al. Neuroprotective effects of cystamine in aged parkinsonian mice. Neurobiol. Aging 27, 862–870 (2006).
Wilkinson, B. & Gilbert, H.F. Protein disulfide isomerase. Biochim. Biophys. Acta 1699, 35–44 (2004).
Ellgaard, L. & Ruddock, L.W. The human protein disulphide isomerase family: substrate interactions and functional properties. EMBO Rep. 6, 28–32 (2005).
Rigobello, M.P., Donella-Deana, A., Cesaro, L. & Bindoli, A. Distribution of protein disulphide isomerase in rat liver mitochondria. Biochem. J. 356, 567–570 (2001).
Turano, C., Coppari, S., Altieri, F. & Ferraro, A. Proteins of the PDI family: unpredicted non-ER locations and functions. J. Cell. Physiol. 193, 154–163 (2002).
Vance, J.E. Phospholipid synthesis in a membrane fraction associated with mitochondria. J. Biol. Chem. 265, 7248–7256 (1990).
Rizzuto, R. et al. Close contacts with the endoplasmic reticulum as determinants of mitochondrial Ca2+ responses. Science 280, 1763–1766 (1998).
Hayashi, T. & Su, T.P. Sigma-1 receptor chaperones at the ER-mitochondrion interface regulate Ca2+ signaling and cell survival. Cell 131, 596–610 (2007).
Hayashi, T., Rizzuto, R., Hajnoczky, G. & Su, T.P. MAM: more than just a housekeeper. Trends Cell Biol. 19, 81–88 (2009).
Lindsten, T. et al. The combined functions of proapoptotic Bcl-2 family members bak and bax are essential for normal development of multiple tissues. Mol. Cell 6, 1389–1399 (2000).
Nie, C. et al. Cysteine 62 of Bax is critical for its conformational activation and its proapoptotic activity in response to H2O2-induced apoptosis. J. Biol. Chem. 283, 15359–15369 (2008).
Khoshnan, A. et al. Activation of the IκB kinase complex and nuclear factor-κB contributes to mutant huntingtin neurotoxicity. J. Neurosci. 24, 7999–8008 (2004).
Varma, H., Cheng, R., Voisine, C., Hart, A.C. & Stockwell, B.R. Inhibitors of metabolism rescue cell death in Huntington's disease models. Proc. Natl. Acad. Sci. USA 104, 14525–14530 (2007).
Southwell, A.L. et al. Intrabodies binding the proline-rich domains of mutant huntingtin increase its turnover and reduce neurotoxicity. J. Neurosci. 28, 9013–9020 (2008).
Thinakaran, G. & Koo, E.H. Amyloid precursor protein trafficking, processing, and function. J. Biol. Chem. 283, 29615–29619 (2008).
Braithwaite, S.P. et al. Inhibition of c-Jun kinase provides neuroprotection in a model of Alzheimer's disease. Neurobiol. Dis. 39, 311–317 (2010).
Friedlander, R.M., Brown, R.H., Gagliardini, V., Wang, J. & Yuan, J. Inhibition of ICE slows ALS in mice. Nature 388, 31 (1997).
Ona, V.O. et al. Inhibition of caspase-1 slows disease progression in a mouse model of Huntington's disease. Nature 399, 263–267 (1999).
Chen, M. et al. Minocycline inhibits caspase-1 and caspase-3 expression and delays mortality in a transgenic mouse model of Huntington disease. Nat. Med. 6, 797–801 (2000).
Zhu, S. et al. Minocycline inhibits cytochrome c release and delays progression of amyotrophic lateral sclerosis in mice. Nature 417, 74–78 (2002).
Graham, R.K. et al. Cleavage at the caspase-6 site is required for neuronal dysfunction and degeneration due to mutant huntingtin. Cell 125, 1179–1191 (2006).
Galvan, V. et al. Reversal of Alzheimer's-like pathology and behavior in human APP transgenic mice by mutation of Asp664. Proc. Natl. Acad. Sci. USA 103, 7130–7135 (2006).
Atkin, J.D. et al. Induction of the unfolded protein response in familial amyotrophic lateral sclerosis and association of protein-disulfide isomerase with superoxide dismutase 1. J. Biol. Chem. 281, 30152–30165 (2006).
Conn, K.J. et al. Identification of the protein disulfide isomerase family member PDIp in experimental Parkinson's disease and Lewy body pathology. Brain Res. 1022, 164–172 (2004).
Yoo, B.C. et al. Overexpressed protein disulfide isomerase in brains of patients with sporadic Creutzfeldt-Jakob disease. Neurosci. Lett. 334, 196–200 (2002).
Uehara, T. et al. S-nitrosylated protein-disulphide isomerase links protein misfolding to neurodegeneration. Nature 441, 513–517 (2006).
Bailey, C.D. & Johnson, G.V. The protective effects of cystamine in the R6/2 Huntington's disease mouse involve mechanisms other than the inhibition of tissue transglutaminase. Neurobiol. Aging 27, 871–879 (2006).
Acknowledgements
We thank E. Schweitzer (University of California–Los Angeles) for the transfected PC12 cell lines; C. Ross (Johns Hopkins University) for HTT DNAs upon which the constructs used here were based; I. Smukste for assistance with organic synthesis; A. Bauer for compound library assembly library; D. Dunn for additional brain-slice experiments; C. Thompson (University of Pennsylvania) for the Bax/Bak double-knockout MEF cells; L. Moore (Gage laboratory, Salk Institute) for tebufenozide; A. Speers, J. Alexander and B. Cravatt for reagents and advice on the cycloaddition reactions; E. Miller, E. Signer, A. Tobin, N. Wexler, C. Johnson, R. Pacifici and M. Finn for useful discussions; and E. Miller and S. Hoffschmidt for editorial comments on the manuscript. This research was supported by grants from the US National Institutes of Health (NIGMS 1RO1GM085081 to B.R.S. and NINDS R21NS048181 to D.C.L.), the Hereditary Disease Foundation (D.C.L., B.R.S.), the High Q Foundation (B.R.S.), CHDI Foundation (D.C.L., B.R.S.), the Arnold and Mabel Beckman Foundation (B.R.S.), the Training Program in Molecular Biophysics (T32GM008281 to A.K.) and a Burroughs Wellcome Fund Career Award at the Scientific Interface (B.R.S.). B.G.H. was supported in part by a postdoctoral fellowship from the High Q Foundation. B.R.S. is an Early Career Scientist of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
B.G.H. and B.R.S designed the experiments. B.G.H. performed the experiments and analyzed the data with B.R.S. A.K. assisted with the library screening, characterization of hit compounds, analysis of synthesized analogs, validation studies and structural identification of securinine. R.L. performed the PDI and Bcl-2 overexpression experiments. R.S.S., G.J.T. and D.C.L. provided the cortical-striatal brain-slice data. B.G.H. and B.R.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Methods, Supplementary Tables 1–3 and Supplementary Figures 1–10 (PDF 1975 kb)
Rights and permissions
About this article
Cite this article
Hoffstrom, B., Kaplan, A., Letso, R. et al. Inhibitors of protein disulfide isomerase suppress apoptosis induced by misfolded proteins. Nat Chem Biol 6, 900–906 (2010). https://doi.org/10.1038/nchembio.467
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.467
This article is cited by
-
The novel role of ER protein TXNDC5 in the pathogenesis of organ fibrosis: mechanistic insights and therapeutic implications
Journal of Biomedical Science (2022)
-
Protection against glutathione depletion-associated oxidative neuronal death by neurotransmitters norepinephrine and dopamine: Protein disulfide isomerase as a mechanistic target for neuroprotection
Acta Pharmacologica Sinica (2022)
-
PDIA3 inhibits mitochondrial respiratory function in brain endothelial cells and C. elegans through STAT3 signaling and decreases survival after OGD
Cell Communication and Signaling (2021)
-
STIM1, STIM2, and PDI Participate in Cellular Fate Decisions in Low Energy Availability Induced by 3-NP in Male Rats
Neurotoxicity Research (2021)
-
Protein disulfide isomerase in cardiovascular disease
Experimental & Molecular Medicine (2020)