Abstract
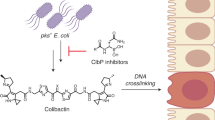
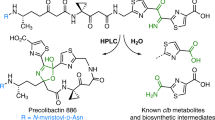
Colibactin is an as-yet-uncharacterized genotoxic secondary metabolite produced by human gut bacteria. Here we report the biosynthetic discovery of two new precolibactin molecules from Escherichia coli, including precolibactin-886, which uniquely incorporates the highly sought genotoxicity-associated aminomalonate building block into its unprecedented macrocyclic structure. This work provides new insights into the biosynthetic logic and mode of action of this colorectal-cancer-linked microbial chemical.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Nougayrède, J.P. et al. Science 313, 848–851 (2006).
Bode, H.B. Angew. Chem. Int. Ed. Engl. 54, 10408–10411 (2015).
Balskus, E.P. Nat. Prod. Rep. 32, 1534–1540 (2015).
Arthur, J.C. et al. Science 338, 120–123 (2012).
Welch, R.A. et al. Proc. Natl. Acad. Sci. USA 99, 17020–17024 (2002).
Olier, M. et al. Gut Microbes 3, 501–509 (2012).
Vizcaino, M.I. & Crawford, J.M. Nat. Chem. 7, 411–417 (2015).
Li, Z.R. et al. ChemBioChem 16, 1715–1719 (2015).
Engel, P., Vizcaino, M.I. & Crawford, J.M. Appl. Environ. Microbiol. 81, 1502–1512 (2015).
Brotherton, C.A. & Balskus, E.P. J. Am. Chem. Soc. 135, 3359–3362 (2013).
Bian, X. et al. ChemBioChem 14, 1194–1197 (2013).
Brachmann, A.O. et al. Chem. Commun. (Camb.) 51, 13138–13141 (2015).
Zha, L., Wilson, M.R., Brotherton, C.A. & Balskus, E.P. ACS Chem. Biol. 11, 1287–1295 (2016).
Vizcaino, M.I., Engel, P., Trautman, E. & Crawford, J.M. J. Am. Chem. Soc. 136, 9244–9247 (2014).
Brotherton, C.A., Wilson, M., Byrd, G. & Balskus, E.P. Org. Lett. 17, 1545–1548 (2015).
Bian, X., Plaza, A., Zhang, Y. & Müller, R. Chem. Sci. 6, 3154–3160 (2015).
Li, Y. et al. Sci. Rep. 5, 9383 (2015).
Du, L. & Lou, L. Nat. Prod. Rep. 27, 255–278 (2010).
Patonay, T. & Hoffman, R.V. J. Org. Chem. 60, 2368–2377 (1995).
Pinho e Melo, T.M.V.D. et al. J. Org. Chem. 67, 66–71 (2002).
Healy, A.R., Vizcaino, M.I., Crawford, J.M. & Herzon, S.B. J. Am. Chem. Soc. 138, 5426–5432 (2016).
Aslan, K. et al. Anal. Chem. 80, 4125–4132 (2008).
Gust, B., Challis, G.L., Fowler, K., Kieser, T. & Chater, K.F. Proc. Natl. Acad. Sci. USA 100, 1541–1546 (2003).
Kumar, S., Stecher, G. & Tamura, K. Mol. Biol. Evol. 33, 1870–1874 (2016).
Okonechnikov, K., Golosova, O. & Fursov, M. Bioinformatics 28, 1166–1167 (2012).
Datsenko, K.A. & Wanner, B.L. Proc. Natl. Acad. Sci. USA 97, 6640–6645 (2000).
Kouprina, N. & Larionov, V. Nat. Protoc. 3, 371–377 (2008).
Yamanaka, K. et al. Proc. Natl. Acad. Sci. USA 111, 1957–1962 (2014).
Liu, T. et al. Chem. Biol. 13, 945–955 (2006).
Rohm, B., Scherlach, K. & Hertweck, C. Org. Biomol. Chem. 8, 1520–1522 (2010).
Fujii, K. et al. Anal. Chem. 69, 5146–5151 (1997).
Ross, A.C. et al. J. Am. Chem. Soc. 135, 1155–1162 (2013).
Lock, R.B. & Stribinskiene, L. Cancer Res. 56, 4006–4012 (1996).
Acknowledgements
This work was generously supported by grants from the China Ocean Mineral Resources Research and Development Association (13.5 program to P.-Y.Q.) and the NIH (R01-GM85770 to B.S.M.). We thank P.R. Jensen, J. Busch, C.B. Naman, and Y.K. Tam for technical advice and access to equipment.
Author information
Authors and Affiliations
Contributions
Z.-R.L., J.L., J.-P.G., J.Y.H.L., B.M.D., W.-P.Z., Z.-L.L., Y.-X.L., R.-B.T., Y.X., and D.-H.L. performed experiments and collected data; all authors analyzed data; Z.-R.L., J.L., B.S.M., and P.-Y.Q. designed the study and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Tables 1–3 and Supplementary Figures 1–18. (PDF 4521 kb)
Supplementary Note
Supplementary Notes 1–3. (PDF 6943 kb)
Source data
Rights and permissions
About this article
Cite this article
Li, ZR., Li, J., Gu, JP. et al. Divergent biosynthesis yields a cytotoxic aminomalonate-containing precolibactin. Nat Chem Biol 12, 773–775 (2016). https://doi.org/10.1038/nchembio.2157
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2157
This article is cited by
-
Mutational signature in colorectal cancer caused by genotoxic pks+ E. coli
Nature (2020)
-
The mysteries of macrocyclic colibactins
Nature Chemistry (2019)
-
Macrocyclic colibactin induces DNA double-strand breaks via copper-mediated oxidative cleavage
Nature Chemistry (2019)
-
Synthesis and reactivity of precolibactin 886
Nature Chemistry (2019)
-
Refining and expanding nonribosomal peptide synthetase function and mechanism
Journal of Industrial Microbiology and Biotechnology (2019)