Abstract
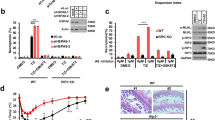
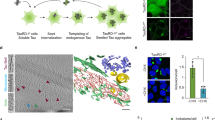
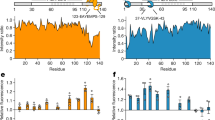
Excessive aggregation of proteins has a major impact on cell fate and is a hallmark of amyloid diseases in humans. To resolve insoluble deposits and to maintain protein homeostasis, all cells use dedicated protein disaggregation, protein folding and protein degradation factors. Despite intense recent research, the underlying mechanisms controlling this key metabolic event are not well understood. Here, we analyzed how a single factor, the highly conserved serine protease HTRA1, degrades amyloid fibrils in an ATP-independent manner. This PDZ protease solubilizes protein fibrils and disintegrates the fibrillar core structure, allowing productive interaction of aggregated polypeptides with the active site for rapid degradation. The aggregate burden in a cellular model of cytoplasmic tau aggregation is thus reduced. Mechanistic aspects of ATP-independent proteolysis and its implications in amyloid diseases are discussed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Wang, L. Towards revealing the structure of bacterial inclusion bodies. Prion 3, 139–145 (2009).
Friedman, R. Aggregation of amyloids in a cellular context: modelling and experiment. Biochem. J. 438, 415–426 (2011).
Akerfelt, M., Morimoto, R.I. & Sistonen, L. Heat shock factors: integrators of cell stress, development and lifespan. Nat. Rev. Mol. Cell Biol. 11, 545–555 (2010).
Saibil, H. Chaperone machines for protein folding, unfolding and disaggregation. Nat. Rev. Mol. Cell Biol. 14, 630–642 (2013).
Clausen, T., Southan, C. & Ehrmann, M. The HtrA family of proteases. Implications for protein composition and cell fate. Mol. Cell 10, 443–455 (2002).
Clausen, T., Kaiser, M., Huber, R. & Ehrmann, M. HTRA proteases: regulated proteolysis in protein quality control. Nat. Rev. Mol. Cell Biol. 12, 152–162 (2011).
Truebestein, L. et al. Substrate-induced remodeling of the active site regulates human HTRA1 activity. Nat. Struct. Mol. Biol. 18, 386–388 (2011).
Chien, J. et al. Serine protease HtrA1 associates with microtubules and inhibits cell migration. Mol. Cell. Biol. 29, 4177–4187 (2009).
Tiaden, A.N. & Richards, P.J. The emerging roles of HTRA1 in musculoskeletal disease. Am. J. Pathol. 182, 1482–1488 (2013).
Jones, A. et al. Increased expression of multifunctional serine protease, HTRA1, in retinal pigment epithelium induces polypoidal choroidal vasculopathy in mice. Proc. Natl. Acad. Sci. USA 108, 14578–14583 (2011).
Vierkotten, S., Muether, P.S. & Fauser, S. Overexpression of HTRA1 leads to ultrastructural changes in the elastic layer of Bruch′s membrane via cleavage of extracellular matrix components. PLoS ONE 6, e22959 (2011).
Grau, S. et al. The role of human HtrA1 in arthritic disease. J. Biol. Chem. 281, 6124–6129 (2006).
Tsuchiya, A. et al. Expression of mouse HtrA1 serine protease in normal bone and cartilage and its upregulation in joint cartilage damaged by experimental arthritis. Bone 37, 323–336 (2005).
Chamberland, A. et al. Identification of a novel HtrA1-susceptible cleavage site in human aggrecan: evidence for the involvement of HtrA1 in aggrecan proteolysis in vivo. J. Biol. Chem. 284, 27352–27359 (2009).
Campioni, M. et al. The serine protease HtrA1 specifically interacts and degrades the tuberous sclerosis complex 2 protein. Mol. Cancer Res. 8, 1248–1260 (2010).
Clawson, G.A., Bui, V., Xin, P., Wang, N. & Pan, W. Intracellular localization of the tumor suppressor HtrA1/Prss11 and its association with HPV16 E6 and E7 proteins. J. Cell. Biochem. 105, 81–88 (2008).
Chien, J., He, X. & Shridhar, V. Identification of tubulins as substrates of serine protease HtrA1 by mixture-based oriented peptide library screening. J. Cell. Biochem. 107, 253–263 (2009).
Chien, J., Campioni, M., Shridhar, V. & Baldi, A. HtrA serine proteases as potential therapeutic targets in cancer. Curr. Cancer Drug Targets 9, 451–468 (2009).
Yang, Z. et al. A variant of the HTRA1 gene increases susceptibility to age-related macular degeneration. Science 314, 992–993 (2006).
Grau, S. et al. Implications of the serine protease HtrA1 in amyloid precursor protein processing. Proc. Natl. Acad. Sci. USA 102, 6021–6026 (2005).
Milner, J.M., Patel, A. & Rowan, A.D. Emerging roles of serine proteinases in tissue turnover in arthritis. Arthritis Rheum. 58, 3644–3656 (2008).
Hara, K. et al. Association of HTRA1 mutations and familial ischemic cerebral small-vessel disease. N. Engl. J. Med. 360, 1729–1739 (2009).
Gamblin, T.C. et al. Caspase cleavage of tau: linking amyloid and neurofibrillary tangles in Alzheimer's disease. Proc. Natl. Acad. Sci. USA 100, 10032–10037 (2003).
Wang, Y.P., Biernat, J., Pickhardt, M., Mandelkow, E. & Mandelkow, E.M. Stepwise proteolysis liberates tau fragments that nucleate the Alzheimer-like aggregation of full-length tau in a neuronal cell model. Proc. Natl. Acad. Sci. USA 104, 10252–10257 (2007).
Spillantini, M.G. & Goedert, M. Tau pathology and neurodegeneration. Lancet Neurol. 12, 609–622 (2013).
Tennstaedt, A. et al. Human high temperature requirement serine protease A1 (HTRA1) degrades tau protein aggregates. J. Biol. Chem. 287, 20931–20941 (2012).
Li, W. & Lee, V.M. Characterization of two VQIXXK motifs for tau fibrillization in vitro. Biochemistry 45, 15692–15701 (2006).
Bibow, S. et al. The dynamic structure of filamentous tau. Angew. Chem. Int. Edn Engl. 50, 11520–11524 (2011).
Crowther, R.A. Straight and paired helical filaments in Alzheimer disease have a common structural unit. Proc. Natl. Acad. Sci. USA 88, 2288–2292 (1991).
Guo, J.L. & Lee, V.M. Seeding of normal Tau by pathological Tau conformers drives pathogenesis of Alzheimer-like tangles. J. Biol. Chem. 286, 15317–15331 (2011).
Mukrasch, M.D. et al. Structural polymorphism of 441-residue tau at single residue resolution. PLoS Biol. 7, e34 (2009).
Kuperstein, I. et al. Neurotoxicity of Alzheimer's disease Abeta peptides is induced by small changes in the Abeta42 to Abeta40 ratio. EMBO J. 29, 3408–3420 (2010).
Wilken, C., Kitzing, K., Kurzbauer, R., Ehrmann, M. & Clausen, T. Crystal structure of the DegS stress sensor: how a PDZ domain recognizes misfolded protein and activates a protease domain. Cell 117, 483–494 (2004).
Krojer, T. et al. Interplay of PDZ and protease domain of DegP ensures efficient elimination of misfolded proteins. Proc. Natl. Acad. Sci. USA 105, 7702–7707 (2008).
Meltzer, M. et al. Allosteric activation of HtrA protease DegP by stress signals during bacterial protein quality control. Angew. Chem. Int. Edn Engl. 47, 1332–1334 (2008).
Merdanovic, M. et al. Determinants of structural and functional plasticity of a widely conserved protease chaperone complex. Nat. Struct. Mol. Biol. 17, 837–843 (2010).
Eigenbrot, C. et al. Structural and functional analysis of HtrA1 and its subdomains. Structure 20, 1040–1050 (2012).
Risør, M.W. et al. The autolysis of human HtrA1 is governed by the redox state of its N-terminal domain. Biochemistry 53, 3851–3857 (2014).
Jaru-Ampornpan, P. et al. ATP-independent reversal of a membrane protein aggregate by a chloroplast SRP subunit. Nat. Struct. Mol. Biol. 17, 696–702 (2010).
Spiess, C., Beil, A. & Ehrmann, M. A temperature-dependent switch from chaperone to protease in a widely conserved heat shock protein. Cell 97, 339–347 (1999).
Krojer, T. et al. Structural basis for the regulated protease and chaperone function of DegP. Nature 453, 885–890 (2008).
Malet, H. et al. Binding of substrate proteins inside the molecular cage of the chaperone-protease DegQ. Nat. Struct. Mol. Biol. 19, 152–157 (2012).
Miyata, Y., Koren, J., Kiray, J., Dickey, C.A. & Gestwicki, J.E. Molecular chaperones and regulation of tau quality control: strategies for drug discovery in tauopathies. Future Med. Chem. 3, 1523–1537 (2011).
Dou, F. et al. Chaperones increase association of tau protein with microtubules. Proc. Natl. Acad. Sci. USA 100, 721–726 (2003).
DeSantis, M.E. et al. Operational plasticity enables hsp104 to disaggregate diverse amyloid and nonamyloid clients. Cell 151, 778–793 (2012).
Johnson, G.V. Tau phosphorylation and proteolysis: insights and perspectives. J. Alzheimers Dis. 9, 243–250 (2006).
Hegde, A.N. & Upadhya, S.C. Role of ubiquitin-proteasome-mediated proteolysis in nervous system disease. Biochim. Biophys. Acta 1809, 128–140 (2011).
Karring, H. et al. Composition and proteolytic processing of corneal deposits associated with mutations in the TGFBI gene. Exp. Eye Res. 96, 163–170 (2012).
Hardy, J. & Revesz, T. The spread of neurodegenerative disease. N. Engl. J. Med. 366, 2126–2128 (2012).
Searcy, J.L., Le Bihan, T., Salvadores, N., McCulloch, J. & Horsburgh, K. Impact of age on the cerebrovascular proteomes of wild-type and Tg-SwDI mice. PLoS ONE 9, e89970 (2014).
Schneider, C.A., Rasband, W.S. & Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Thévenaz, P., Ruttimann, U.E. & Unser, M. A pyramid approach to subpixel registration based on intensity. IEEE Trans. Image Process. 7, 27–41 (1998).
Rappsilber, J., Mann, M. & Ishihama, Y. Protocol for micro-purification, enrichment, pre-fractionation and storage of peptides for proteomics using StageTips. Nat. Protoc. 2, 1896–1906 (2007).
Michalski, A. et al. Ultra high resolution linear ion trap Orbitrap mass spectrometer (Orbitrap Elite) facilitates top down LC MS/MS and versatile peptide fragmentation modes. Mol. Cell. Proteomics 11, O111013698 (2012).
Olsen, J.V. et al. Parts per million mass accuracy on an Orbitrap mass spectrometer via lock mass injection into a C-trap. Mol. Cell. Proteomics 4, 2010–2021 (2005).
Perkins, D.N., Pappin, D.J., Creasy, D.M. & Cottrell, J.S. Probability-based protein identification by searching sequence databases using mass spectrometry data. Electrophoresis 20, 3551–3567 (1999).
Acknowledgements
We thank N. Schulze and M. Graessl for help with TIRFM, D. Grum and M. Merdanovic with ITC, S. Blaskowski with MS, V. Lux (University Duisburg-Essen) for providing purified PDZ domain and M. Breiden (University Duisburg-Essen) for providing tau. This work was supported by grants EH 100/14-1 (to M.E.) and SFB 1093 (to M.E., M.K. and B.S.) and INST 20876/127-1 FUGG (to M.K.) from Deutsche Forschungsgemeinschaft. The Research Institute of Molecular Pathology is funded by Boehringer Ingelheim.
Author information
Authors and Affiliations
Contributions
S.P. designed and carried out experiments, analyzed data and wrote the paper; A.S. and B.S. carried out AFM; F.K. and M.K. carried out MS; C.G. and S.R. carried out EM; T.C. performed biochemistry; and M.E. outlined the work and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Table 1 and Supplementary Figures 1–9. (PDF 21400 kb)
Supplementary Data Set 1
Uncut gel images: full gels of Figs 1a, 2c, 3a–c, 6a and 6e. (PDF 7454 kb)
Rights and permissions
About this article
Cite this article
Poepsel, S., Sprengel, A., Sacca, B. et al. Determinants of amyloid fibril degradation by the PDZ protease HTRA1. Nat Chem Biol 11, 862–869 (2015). https://doi.org/10.1038/nchembio.1931
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1931
This article is cited by
-
HTRA1 disaggregates α-synuclein amyloid fibrils and converts them into non-toxic and seeding incompetent species
Nature Communications (2024)
-
Proteomic profiling in cerebral amyloid angiopathy reveals an overlap with CADASIL highlighting accumulation of HTRA1 and its substrates
Acta Neuropathologica Communications (2022)
-
Dissecting the clinical heterogeneity of early-onset Alzheimer’s disease
Molecular Psychiatry (2022)
-
Allosteric inhibition of HTRA1 activity by a conformational lock mechanism to treat age-related macular degeneration
Nature Communications (2022)
-
Tau-proximity ligation assay reveals extensive previously undetected pathology prior to neurofibrillary tangles in preclinical Alzheimer’s disease
Acta Neuropathologica Communications (2021)