Abstract
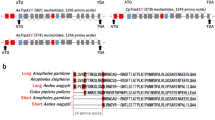
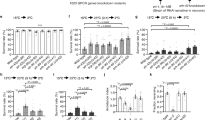
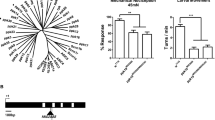
Discriminating among sensory stimuli is critical for animal survival. This discrimination is particularly essential when evaluating whether a stimulus is noxious or innocuous. From insects to humans, transient receptor potential (TRP) channels are key transducers of thermal, chemical and other sensory cues1,2. Many TRPs are multimodal receptors that respond to diverse stimuli1,2,3, but how animals distinguish sensory inputs activating the same TRP is largely unknown. Here we determine how stimuli activating Drosophila TRPA1 are discriminated. Although Drosophila TRPA1 responds to both noxious chemicals4 and innocuous warming5, we find that TRPA1-expressing chemosensory neurons respond to chemicals but not warmth, a specificity conferred by a chemosensory-specific TRPA1 isoform with reduced thermosensitivity compared to the previously described isoform. At the molecular level, this reduction results from a unique region that robustly reduces the channel’s thermosensitivity. Cell-type segregation of TRPA1 activity is critical: when the thermosensory isoform is expressed in chemosensors, flies respond to innocuous warming with regurgitation, a nocifensive response. TRPA1 isoform diversity is conserved in malaria mosquitoes, indicating that similar mechanisms may allow discrimination of host-derived warmth—an attractant—from chemical repellents. These findings indicate that reducing thermosensitivity can be critical for TRP channel functional diversification, facilitating their use in contexts in which thermal sensitivity can be maladaptive.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dhaka, A., Viswanath, V. & Patapoutian, A. TRP ion channels and temperature sensation. Annu. Rev. Neurosci. 29, 135–161 (2006)
Wu, L. J., Sweet, T. B. & Clapham, D. E. International Union of Basic and Clinical Pharmacology. LXXVI. Current progress in the mammalian TRP ion channel family. Pharmacol. Rev. 62, 381–404 (2010)
Daniels, R. L. & McKemy, D. D. Mice left out in the cold: commentary on the phenotype of TRPM8-nulls. Mol. Pain 3, 23 (2007)
Kang, K. et al. Analysis of Drosophila TRPA1 reveals an ancient origin for human chemical nociception. Nature 464, 597–600 (2010)
Hamada, F. N. et al. An internal thermal sensor controlling temperature preference in Drosophila . Nature 454, 217–220 (2008)
Patapoutian, A., Tate, S. & Woolf, C. J. Transient receptor potential channels: targeting pain at the source. Nature Rev. Drug Discov. 8, 55–68 (2009)
Viswanath, V. et al. Opposite thermosensor in fruitfly and mouse. Nature 423, 822–823 (2003)
Cohet, Y. Epigenetic influences on the lifespan of the Drosophila: existence of an optimal growth temperature for adult longevity. Exp. Gerontol. 10, 181–184 (1975)
Hodgson, E. S., Lettvin, J. Y. & Roeder, K. D. Physiology of a primary chemoreceptor unit. Science 122, 417–418 (1955)
Weiss, L. A., Dahanukar, A., Kwon, J. Y., Banerjee, D. & Carlson, J. R. The molecular and cellular basis of bitter taste in Drosophila . Neuron 69, 258–272 (2011)
von Philipsborn, A. C. et al. Neuronal control of Drosophila courtship song. Neuron 69, 509–522 (2011)
Graveley, B. R. et al. The developmental transcriptome of Drosophila melanogaster . Nature 471, 473–479 (2011)
Vyklicky, L. et al. Temperature coefficient of membrane currents induced by noxious heat in sensory neurones in the rat. J. Physiol. (Lond.) 517, 181–192 (1999)
Tracey, W. D., Jr, Wilson, R. I., Laurent, G. & Benzer, S. painless, a Drosophila gene essential for nociception. Cell 113, 261–273 (2003)
Talavera, K. et al. Heat activation of TRPM5 underlies thermal sensitivity of sweet taste. Nature 438, 1022–1025 (2005)
Gracheva, E. O. et al. Molecular basis of infrared detection by snakes. Nature 464, 1006–1011 (2010)
Gracheva, E. O. et al. Ganglion-specific splicing of TRPV1 underlies infrared sensation in vampire bats. Nature 476, 88–91 (2011)
Marella, S. et al. Imaging taste responses in the fly brain reveals a functional map of taste category and behavior. Neuron 49, 285–295 (2006)
Lu, G., Henderson, D., Liu, L., Reinhart, P. H. & Simon, S. A. TRPV1b, a functional human vanilloid receptor splice variant. Mol. Pharmacol. 67, 1119–1127 (2005)
Vos, M. H. et al. TRPV1b overexpression negatively regulates TRPV1 responsiveness to capsaicin, heat and low pH in HEK293 cells. J. Neurochem. 99, 1088–1102 (2006)
Brauchi, S., Orio, P. & Latorre, R. Clues to understanding cold sensation: thermodynamics and electrophysiological analysis of the cold receptor TRPM8. Proc. Natl Acad. Sci. USA 101, 15494–15499 (2004)
Liu, B., Hui, K. & Qin, F. Thermodynamics of heat activation of single capsaicin ion channels VR1. Biophys. J. 85, 2988–3006 (2003)
Wang, G. et al. Anopheles gambiae TRPA1 is a heat-activated channel expressed in thermosensitive sensilla of female antennae. Eur. J. Neurosci. 30, 967–974 (2009)
Maekawa, E. et al. The role of proboscis of the malaria vector mosquito Anopheles stephensi in host-seeking behavior. Parasit. Vectors 4, 10 (2011)
Uchida, K. & Tominaga, M. The role of thermosensitive TRP (transient receptor potential) channels in insulin secretion. Endocr. J. 10.1507/endocrj.EJ11-0130 (2011)
Grandl, J. et al. Pore region of TRPV3 ion channel is specifically required for heat activation. Nature Neurosci. 11, 1007–1013 (2008)
Grandl, J. et al. Temperature-induced opening of TRPV1 ion channel is stabilized by the pore domain. Nature Neurosci. 13, 708–714 (2010)
Yang, F., Cui, Y., Wang, K. & Zheng, J. Thermosensitive TRP channel pore turret is part of the temperature activation pathway. Proc. Natl Acad. Sci. USA 107, 7083–7088 (2011)
Yao, J., Liu, B. & Qin, F. Modular thermal sensors in temperature-gated transient receptor potential (TRP) channels. Proc. Natl Acad. Sci. USA 108, 11109–11114 (2011)
Groth, A. C., Fish, M., Nusse, R. & Calos, M. P. Construction of transgenic Drosophila by using the site-specific integrase from phage φC31. Genetics 166, 1775–1782 (2004)
Markstein, M., Pitsouli, C., Villalta, C., Celniker, S. E. & Perrimon, N. Exploiting position effects and the gypsy retrovirus insulator to engineer precisely expressed transgenes. Nature Genet. 40, 476–483 (2008)
Wieczorek, H. & Wolff, G. The labellar sugar receptor of Drosophila . J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 164, 825–834 (1989)
Edgar, R. C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797 (2004)
Acknowledgements
We thank the P.A.G. laboratory, H. Garrity, L. Griffith, L. Huang, J. Rodriguez and M. Rosbash for helpful comments, and F. Marion-Poll and A. Dahanukar for guidance with tip-recording. This work was supported by grants from the National Science Foundation (IOS-1025307), National Institute of Mental Health (EUREKA R01 MH094721) and National Institute of Neurological Disorders and Stroke (NINDS) (PO1 NS044232) to P.A.G., a National Research Service Award from NINDS to V.C.P. (F31 NS071897-02) and the Boston College DeLuca Professorship to M.A.T.M.
Author information
Authors and Affiliations
Contributions
K.K., V.C.P. and P.A.G. designed experiments. K.K. performed molecular biology, genetics and oocyte physiology. V.C.P. performed genetics and sensory neuron electrophysiology. E.C.C. performed genetics and behavioural experiments. A.M.D. performed behavioural experiments. L.N. performed immunohistochemistry. A.M.J., K.R. and M.A.T.M. grew and harvested mosquitoes. P.A.G. performed bioinformatics. K.K., V.C.P. and P.A.G. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Figures 1-10 with legends. (PDF 1266 kb)
Supplementary Movie 1
This movie 'warmth triggered regurgitation in a TRPA1(B) rescue fly' shows a Gr66a>TRPA1(B); TrpA1ins mutant rescue animal being heated from room temperature (~23˚C) to ~32˚C over 30 seconds. The fly responds to warming by initiating proboscis movements and eventually regurgitates previously ingested liquid. (MOV 8091 kb)
Rights and permissions
About this article
Cite this article
Kang, K., Panzano, V., Chang, E. et al. Modulation of TRPA1 thermal sensitivity enables sensory discrimination in Drosophila. Nature 481, 76–80 (2012). https://doi.org/10.1038/nature10715
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10715
This article is cited by
-
Molecular architecture and gating mechanisms of the Drosophila TRPA1 channel
Cell Discovery (2023)
-
TRPA1-mediated repellency behavior in the red flour beetle Tribolium castaneum
Scientific Reports (2022)
-
The human TRPA1 intrinsic cold and heat sensitivity involves separate channel structures beyond the N-ARD domain
Nature Communications (2022)
-
Subsecond multichannel magnetic control of select neural circuits in freely moving flies
Nature Materials (2022)
-
Sonogenetic control of mammalian cells using exogenous Transient Receptor Potential A1 channels
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.