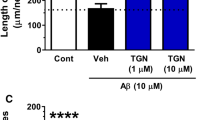
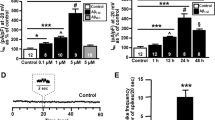
Abstract
Here, we report a novel target of the drug memantine, ATP-sensitive K+ (KATP) channels, potentially relevant to memory improvement. We confirmed that memantine antagonizes memory impairment in Alzheimer’s model APP23 mice. Memantine increased CaMKII activity in the APP23 mouse hippocampus, and memantine-induced enhancement of hippocampal long-term potentiation (LTP) and CaMKII activity was totally abolished by treatment with pinacidil, a specific opener of KATP channels. Memantine also inhibited Kir6.1 and Kir6.2 KATP channels and elevated intracellular Ca2+ concentrations in neuro2A cells overexpressing Kir6.1 or Kir6.2. Kir6.2 was preferentially expressed at postsynaptic regions of hippocampal neurons, whereas Kir6.1 was predominant in dendrites and cell bodies of pyramidal neurons. Finally, we confirmed that Kir6.2 mutant mice exhibit severe memory deficits and impaired hippocampal LTP, impairments that cannot be rescued by memantine administration. Altogether, our studies show that memantine modulates Kir6.2 activity, and that the Kir6.2 channel is a novel target for therapeutics to improve memory impairment in Alzheimer disease patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Greenamyre JT, Penney JB, D’Amato CJ, Young AB . Dementia of the Alzheimer’s type: changes in hippocampal L-[3H] glutamate binding. J Neurochem 1987; 48: 543–551.
Chen HS, Lipton SA . The chemical biology of clinically tolerated NMDA receptor antagonists. J Neurochem 2006; 97: 1611–1626.
Chen HS, Pellegrini JW, Aggarwai SK, Lei SZ, Warach S, Jensen F et al. Open-channel block of N-methyl-d-aspartate (NMDA) responses by memantine: therapeutic advantage against NMDA receptor-mediated neurotoxicity. J Neurosci 1992; 12: 4427–2236.
Lipton SA . Paradigm shift in NMDA receptor antagonist drug development: molecular mechanism of uncompetitive inhibition by memantine in the treatment of Alzheimer’s disease and other neurologic disorders. J Alzheimers Dis 2004; 6: S61–S74.
Fukunaga K, Stoppini L, Miyamoto E, Muller D . Long-term potentiation is associated with a increased activity of Ca2+/calmodulin-dependent protein kinase II. J Biol Chem 1993; 268: 7863–7867.
Silva AJ, Paylor R, Wehner JM, Tonegawa S . Impaired spatial learning in alpha-calcium-calmodulin kinase II mutant mice. Science 1992; 257: 206–211.
Babenko AP, Aguilar-Bryan L, Bryan J . A view of sur/KIR6.X, KATP channels. Annu Rev Physiol 1998; 60: 667–687.
Zawar C, Plant TD, Schirra C, Konnerth A, Neumcke B . Cell-type specific expression of ATP-sensitive potassium channels in the rat hippocampus. J Physiol 1999; 514: 327–341.
Thomzig A, Laube G, Pruss H, Veh RW . Pore-forming subunits of K-ATP channels, Kir6.1 and Kir6.2, display prominent differences in regional and cellular distribution. J Comp Neurol 2005; 484: 313–330.
Sun XL, Hu G . ATP-sensitive potassium channels: a promissing target for protecting neurovascular unit function in stroke. Clin Exp Pharmacol Physiol 2010; 37: 243–252.
Moriguchi S, Nishi M, Komazaki S, Sakagami H, Miyazaki T, Masumiya H et al. Functional uncoupling between Ca2+ release and afterhyperpolarization in mutant hippocampal neurons lacking junctophilins. Proc Natl Acad Sci USA 2006; 103: 10811–10816.
Hokama M, Oka S, Leon J, Ninomiya T, Honda H, Sasaki K et al. Altered expression of diabetes-related genes in Alzheimer’s disease brains: the Hisayama study. Cerebral Cortex 2014; 24: 2476–2488.
Miki T, Nagashima K, Tashiro F, Kotake K, Yoshitomi H, Tamamoto A et al. Defective insulin secretion and enhanced insulin action in KATP channel deficient mice. Proc Natl Acad Sci USA 1998; 95: 10402–10406.
Miki T, Suzuki M, Shibasaki T, Uemura H, Sato T, Yamaguchi K et al. Mouse model of prinzmetal angina by disruption of the inward rectifier Kir6.1. Nat Med 2002; 8: 466–472.
Sturchler-Pierrat C, Abramowski D, Duke M, Wiederhold KH, Mistl C, Rothacher S et al. Two amyloid precursor protein transgenic mouse models with Alzheimer’s disease-like pathology. Proc Natl Acad Sci USA 1997; 94: 13287–13292.
Yamasaki N, Maekawa M, Kobayashi K, Kajii Y, Maeda J, Soma M et al. Alpha-CaMKII deficiency causes immature dentate gyrus, a novel candidate endophenotype of psychiatric disorders. Mol Brain 2008; 1: 6.
Takao K, Tanda K, Nakamura K, Kasahara J, Nakao K, Katsuki M et al. Comprehensive behavioral analysis of calcium/calmodulin-dependent protein kinase IV knockout mice. PLoS ONE 2010; 5: e9460.
Fukunaga K, Muller D, Miyamoto E . Increased phosphorylation of Ca2+/calmodulin-dependent protein kinase II and its endgeneous substrates in the induction of long term potentiation. J Biol Chem 1995; 270: 6119–6124.
Fukunaga K, Horikawa K, Shibata S, Takeuchi Y, Miyamoto E . Ca2+/calmodulin-dependent protein kinase II-dependent long-term potentiation in the rat suprachiasmatic nucleus and its inhibition by melatonin. J Neurosci Res 2002; 70: 799–807.
Sturchler-Pierrat C, Staufenbiel M . Pathogenic mechanisms of Alzheimer’s disease analyzed in the APP23 transgenic mouse model. Ann NY Acad Sci 2000; 920: 134–139.
Rossor MN, Newman S, Frackowiak RS, Lantos P, Kennedy AM . Alzheimer’s disease families with amyloid precursor protein mutations. Ann NY Acad Sci 1993; 695: 198–202.
Kasahara J, Fukunaga K, Miyamoto E . Activation of calcium/calmodulin-dependent protein kinase IV in long term potentiation in the rat hippocampal CA1 region. J Biol Chem 2001; 276: 24044–24050.
Bliss TV, Collingridge GL . A synaptic model of memory: long-term potentiation in the hippocampus. Nature 1993; 361: 31–39.
Shi SH, Hayashi Y, Petralia RS, Zaman SH, Wenthold RJ, Svoboda K et al. Rapid spine delivery and redistribution of AMPA receptors after synaptic NMDA receptor activation. Science 1999; 284: 1811–1816.
Scheetz AJ, Nairn AC, Constantine-Paton M . NMDA receptor-mediated control of protein synthesis at developing synapses. Nat Neurosci 2000; 3: 211–216.
Almeida RC, Souza DG, Soletti RC, Lopez MG, Rodrigues ALS, Gabilan NH . Involvement of PKA, MAPK/ERK and CaMKII, but not PKC in the acute antidepressant-like effect of memantine in mice. Neurosci Lett 2006; 395: 93–97.
Zini S, Tremblay E, Pollard H, Moreau J, Ben-Ari Y . Regional distribution of sulfonylurea receptors in the brain of rodent and primate. Neuroscience 1993; 55: 1085–1091.
Karschin C, Ecke C, Ashcroft FM, Karschin A . Overlapping distribution of KATP channel-forming Kir6.2 subunit and the sulfonylurea receptor SUR1 in rodent brain. FEBS Lett 1997; 401: 59–64.
Seino S, Iwanaga T, Nagashima K, Miki T . Diverse roles of KATP channels learned from Kir6.2 genetically engineered mice. Diabetes 2000; 49: 311–318.
Yang JZ, Huang X, Zhao FF, Xu Q, Hu G . Iptakalim enhances adult mouse hippocampal neurogenesis via opening Kir6.1-composed K-ATP channels expressed in neural stem cells. CNS Neurosci Ther 2012; 18: 737–744.
Choeiri C, Staines WA, Miki T, Seino S, Renaud JM, Teutenberg K et al. Cerebral glucose transporters expression and spatial learning in the K-ATP Kir6.2−/− knockout mice. Behav Brain Res 2006; 172: 233–239.
Betourne A, Bertholet AM, Labroue E, Halley H, Sun HS, Lorsignol A et al. Involvement of hippocampal CA3 KATP channels in contextual memory. Neuropharmacology 2009; 56: 615–625.
Schroder UH, Hock FJ, Wirth K, Englert HC, Reymann KG . The ATP-regulated K+-channel inhibitor HMR-1372 affects synaptic plasticity in hippocampal slices. Eur J Pharmacol 2004; 502: 99–104.
Kline CF, Wright PJ, Koval OM, Zmuda EJ, Johnson BL, Anderson ME et al. βIV-spectrin and CaMKII facilitate Kir6.2 regulation in pancreatic beta cells. Proc Natl Acad Sci USA 2013; 110: 17576–17581.
Sierra A, Zhu Z, Sapay N, Sharotri V, Kline CF, Luczak ED et al. Regulation of cardiac ATP-sensitive potassium channel surface expression by calcium/calmodulin-dependent protein kinase II. J Biol Chem 2013; 288: 1568–1581.
Ishikawa R, Kim R, Namba T, Kohsaka S, Uchino S, Kida S . Time-dependent enhancement of hippocampus-dependent memory after treatment with memantine: Implications for enhanced hippocampal adult neurogenesis. Hippocampus 2014; 24: 784–793.
Moriguchi S, Sakagami H, Yabuki Y, Sasaki Y, Izumi H, Zhang C et al. Stimulation of sigma-1 receptor ameliorates depressive-like behaviors in CaMKIV null mice. Mol Neurobiol 2015; 52: 1210–1222.
Akhtar MW, Sanz-Blasco S, Dolatabadi N, Parker J, Chon K, Lee MS et al. Elevated glucose and oligomeric β-amyloid disrupt synapses via a common pathway of aberrant protein S-nitrosylation. Nat Commun 2016; 7: 10242.
Xu XH, Pan YP, Wang XL . mRNA expression alterations of inward rectifier potassium channels in rat brain with cholinergic impairment. Neurosci Lett 2002; 322: 25–28.
Gloyn AL, Pearson ER, Antcliff JF, Proks P, Bruining GJ, Slingerland AS et al. Activating mutations in the gene encoding the ATP-sensitive potassium-channel subunit Kir6.2 and permanent neonatal diabetes. N Engl J Med 2004; 350: 1838–1849.
Slingerland AS, Hurkx W, Noordam K, Flanagan SE, Jukema JW, Meiners LC et al. Sulphonylurea therapy improves cognition in a patient with the V59M KCNJ11 mutation. Diabetes Med 2008; 25: 277–281.
Acknowledgements
This work was supported in part by grants from the Ministry of Education, Culture, Sports, Science and Technology, and the Ministry of Health and Welfare of Japan (22390109 to KF; 20790398 to SM). Also, thanks to Professor Tsuyoshi Miyakawa for providing CaMKIIα +/− mice and Professor Susumu Seino for providing Kir6.1 +/− and Kir6.2 −/− mice. We also thank Novartis Pharma for providing APP23 mice.
Author Contributions
SM, NS, HT, YY, YS, and HS performed the experiments. TI and HY provided recombinant Kir6.1 or Kir6.2. H.S. provided knockout mice. JZY and TN provided critical reagents and advice. SM and KF wrote the manuscript and designed the study.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Supplementary information
Rights and permissions
About this article
Cite this article
Moriguchi, S., Ishizuka, T., Yabuki, Y. et al. Blockade of the KATP channel Kir6.2 by memantine represents a novel mechanism relevant to Alzheimer’s disease therapy. Mol Psychiatry 23, 211–221 (2018). https://doi.org/10.1038/mp.2016.187
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2016.187
This article is cited by
-
Propolis Promotes Memantine-Dependent Rescue of Cognitive Deficits in APP-KI Mice
Molecular Neurobiology (2022)
-
Combination of Donepezil and Memantine Attenuated Cognitive Impairment Induced by Mixed Endocrine-Disrupting Chemicals: an In Silico Study
Neurotoxicity Research (2022)
-
Kir6.1 Heterozygous Mice Exhibit Aberrant Amygdala-Dependent Cued Fear Memory
Molecular Neurobiology (2020)
-
State-of-the-art: functional fluorescent probes for bioimaging and pharmacological research
Acta Pharmacologica Sinica (2019)
-
Aberrant Amygdala-Dependent Cued Fear Memory in Na+/Ca2+ Exchanger 1 Heterozygous Mice
Molecular Neurobiology (2019)