Abstract
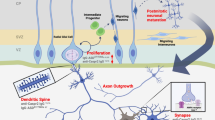
Autism spectrum disorder (ASD) occurs in 1 in 68 births, preferentially affecting males. It encompasses a group of neurodevelopmental abnormalities characterized by impaired social interaction and communication, stereotypic behaviors and motor dysfunction. Although recent advances implicate maternal brain-reactive antibodies in a causative role in ASD, a definitive assessment of their pathogenic potential requires cloning of such antibodies. Here, we describe the isolation and characterization of monoclonal brain-reactive antibodies from blood of women with brain-reactive serology and a child with ASD. We further demonstrate that male but not female mice exposed in utero to the C6 monoclonal antibody, binding to contactin-associated protein-like 2 (Caspr2), display abnormal cortical development, decreased dendritic complexity of excitatory neurons and reduced numbers of inhibitory neurons in the hippocampus, as well as impairments in sociability, flexible learning and repetitive behavior. Anti-Caspr2 antibodies are frequent in women with brain-reactive serology and a child with ASD. Together these studies provide a methodology for obtaining monclonal brain-reactive antibodies from blood B cells, demonstrate that ASD can result from in utero exposure to maternal brain-reactive antibodies of single specificity and point toward the exciting possibility of prognostic and protective strategies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Diagnostic and Statistical Manual of Mental Disorders, 5th edn. American Psychiatric Publishing: Arlington, VA, USA, 2013.
Developmental Disabilities Monitoring Network Surveillance Year 2010 Principal Investigators; Centers for Disease Control and Prevention (CDC). Prevalence of autism spectrum disorder among children aged 8 years - autism and developmental disabilities monitoring network, 11 sites, United States, 2010. MMWR Surveill Summ 2014; 63: 1–21.
Risch N, Hoffmann TJ, Anderson M, Croen LA, Grether JK, Windham GC . Familial recurrence of autism spectrum disorder: evaluating genetic and environmental contributions. Am J Psychiatry 2014; 171: 1206–1213.
Patterson PH . Maternal infection and autism. Brain Behav Immun 2012; 26: 393.
Smith SE, Li J, Garbett K, Mirnics K, Patterson PH . Maternal immune activation alters fetal brain development through interleukin-6. J Neurosci 2007; 27: 10695–10702.
Choi GB, Yim YS, Wong H, Kim S, Kim H, Kim SV et al. The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science 2016; 351: 933–939.
Atladottir HO, Henriksen TB, Schendel DE, Parner ET . Autism after infection, febrile episodes, and antibiotic use during pregnancy: an exploratory study. Pediatrics 2012; 130: e1447–e1454.
Mazina V, Gerdts J, Trinh S, Ankenman K, Ward T, Dennis MY et al. Epigenetics of autism-related impairment: copy number variation and maternal infection. J Dev Behav Pediatr 2015; 36: 61–67.
Brimberg L, Sadiq A, Gregersen PK, Diamond B . Brain-reactive IgG correlates with autoimmunity in mothers of a child with an autism spectrum disorder. Mol Psychiatry 2013; 18: 1171–1177.
Croen LA, Braunschweig D, Haapanen L, Yoshida CK, Fireman B, Grether JK et al. Maternal mid-pregnancy autoantibodies to fetal brain protein: the early markers for autism study. Biol Psychiatry 2008; 64: 583–588.
Braunschweig D, Ashwood P, Krakowiak P, Hertz-Picciotto I, Hansen R, Croen LA et al. Autism: maternally derived antibodies specific for fetal brain proteins. Neurotoxicology 2008; 29: 226–231.
Singer HS, Morris CM, Gause CD, Gillin PK, Crawford S, Zimmerman AW . Antibodies against fetal brain in sera of mothers with autistic children. J Neuroimmunol 2008; 194: 165–172.
Dalton P, Deacon R, Blamire A, Pike M, McKinlay I, Stein J et al. Maternal neuronal antibodies associated with autism and a language disorder. Ann Neurol 2003; 53: 533–537.
Braniste V, Al-Asmakh M, Kowal C, Anuar F, Abbaspour A, Toth M et al. The gut microbiota influences blood-brain barrier permeability in mice. Sci Transl Med 2014; 6: 263ra158.
Bauman MD, Iosif AM, Ashwood P, Braunschweig D, Lee A, Schumann CM et al. Maternal antibodies from mothers of children with autism alter brain growth and social behavior development in the rhesus monkey. Transl Psychiatry 2013; 3: e278.
Singer HS, Morris C, Gause C, Pollard M, Zimmerman AW, Pletnikov M . Prenatal exposure to antibodies from mothers of children with autism produces neurobehavioral alterations: a pregnant dam mouse model. J Neuroimmunol 2009; 211: 39–48.
Braunschweig D, Krakowiak P, Duncanson P, Boyce R, Hansen RL, Ashwood P et al. Autism-specific maternal autoantibodies recognize critical proteins in developing brain. Transl Psychiatry 2013; 3: e277.
Strauss KA, Puffenberger EG, Huentelman MJ, Gottlieb S, Dobrin SE, Parod JM et al. Recessive symptomatic focal epilepsy and mutant contactin-associated protein-like 2. N Engl J Med 2006; 354: 1370–1377.
Poliak S, Gollan L, Martinez R, Custer A, Einheber S, Salzer JL et al. Caspr2, a new member of the neurexin superfamily, is localized at the juxtaparanodes of myelinated axons and associates with K+ channels. Neuron 1999; 24: 1037–1047.
Penagarikano O, Abrahams BS, Herman EI, Winden KD, Gdalyahu A, Dong H et al. Absence of CNTNAP2 leads to epilepsy, neuronal migration abnormalities, and core autism-related deficits. Cell 2011; 147: 235–246.
Fischbach GD, Lord C . The Simons Simplex Collection: a resource for identification of autism genetic risk factors. Neuron 2010; 68: 192–195.
Zhang J, Jacobi AM, Mackay M, Aranow C, Wang T, Chinnasamy P et al. Identification of DNA-reactive B cells in patients with systemic lupus erythematosus. J Immunol Methods 2008; 338: 79–84.
Tiller T, Meffre E, Yurasov S, Tsuiji M, Nussenzweig MC, Wardemann H . Efficient generation of monoclonal antibodies from single human B cells by single cell RT-PCR and expression vector cloning. J Immunol Methods 2008; 329: 112–124.
Lee JY, Huerta PT, Zhang J, Kowal C, Bertini E, Volpe BT et al. Neurotoxic autoantibodies mediate congenital cortical impairment of offspring in maternal lupus. Nat Med 2009; 15: 91–96.
Mader S, Lutterotti A, Di Pauli F, Kuenz B, Schanda K, Aboul-Enein F et al. Patterns of antibody binding to aquaporin-4 isoforms in neuromyelitis optica. PLoS One 2010; 5: e10455.
Bradl M, Misu T, Takahashi T, Watanabe M, Mader S, Reindl M et al. Neuromyelitis optica: pathogenicity of patient immunoglobulin in vivo. Ann Neurol 2009; 66: 630–643.
Wang L, Zhou D, Lee J, Niu H, Faust TW, Frattini S et al. Female mouse fetal loss mediated by maternal autoantibody. The J Exp Med 2012; 209: 1083–1089.
Anderson GR, Galfin T, Xu W, Aoto J, Malenka RC, Sudhof TC . Candidate autism gene screen identifies critical role for cell-adhesion molecule CASPR2 in dendritic arborization and spine development. Proc Natl Acad Sci USA 2012; 109: 18120–18125.
Varea O, Martin-de-Saavedra MD, Kopeikina KJ, Schurmann B, Fleming HJ, Fawcett-Patel JM et al. Synaptic abnormalities and cytoplasmic glutamate receptor aggregates in contactin associated protein-like 2/Caspr2 knockout neurons. Proc Natl Acad Sci USA 2015; 112: 6176–6181.
Poliak S, Salomon D, Elhanany H, Sabanay H, Kiernan B, Pevny L et al. Juxtaparanodal clustering of Shaker-like K+ channels in myelinated axons depends on Caspr2 and TAG-1. J Cell Biol 2003; 162: 1149–1160.
Gdalyahu A, Lazaro M, Penagarikano O, Golshani P, Trachtenberg JT, Geschwind DH . The autism related protein contactin-associated protein-like 2 (CNTNAP2) stabilizes new spines: an in vivo mouse study. PLoS One 2015; 10: e0125633.
Alarcon M, Abrahams BS, Stone JL, Duvall JA, Perederiy JV, Bomar JM et al. Linkage, association, and gene-expression analyses identify CNTNAP2 as an autism-susceptibility gene. Am J Hum Genet 2008; 82: 150–159.
Falivelli G, De Jaco A, Favaloro FL, Kim H, Wilson J, Dubi N et al. Inherited genetic variants in autism-related CNTNAP2 show perturbed trafficking and ATF6 activation. Hum Mol Genet 2012; 21: 4761–4773.
Verkerk AJ, Mathews CA, Joosse M, Eussen BH, Heutink P, Oostra BA . CNTNAP2 is disrupted in a family with Gilles de la Tourette syndrome and obsessive compulsive disorder. Genomics 2003; 82: 1–9.
Simister NE . Placental transport of immunoglobulin G. Vaccine 2003; 21: 3365–3369.
Casanova JR, Nishimura M, Swann JW . The effects of early-life seizures on hippocampal dendrite development and later-life learning and memory. Brain Res Bull 2014; 103: 39–48.
Chavan SS, Huerta PT, Robbiati S, Valdes-Ferrer SI, Ochani M, Dancho M et al. HMGB1 mediates cognitive impairment in sepsis survivors. Mol Med 2012; 18: 930–937.
Silverman JL, Yang M, Lord C, Crawley JN . Behavioural phenotyping assays for mouse models of autism. Nat Rev Neurosci 2010; 11: 490–502.
McFarlane HG, Kusek GK, Yang M, Phoenix JL, Bolivar VJ, Crawley JN . Autism-like behavioral phenotypes in BTBR T+tf/J mice. Genes Brain Behav 2008; 7: 152–163.
Deacon RM . Digging and marble burying in mice: simple methods for in vivo identification of biological impacts. Nat Protoc 2006; 1: 122–124.
Thomas A, Burant A, Bui N, Graham D, Yuva-Paylor LA, Paylor R . Marble burying reflects a repetitive and perseverative behavior more than novelty-induced anxiety. Psychopharmacology (Berl) 2009; 204: 361–373.
Chang EH, Volpe BT, Mackay M, Aranow C, Watson P, Kowal C et al. Selective impairment of spatial cognition caused by autoantibodies to the N-methyl-d-aspartate receptor. EBioMed 2015; 2: 755–764.
Tan GC, Doke TF, Ashburner J, Wood NW, Frackowiak RS . Normal variation in fronto-occipital circuitry and cerebellar structure with an autism-associated polymorphism of CNTNAP2. Neuroimage 2010; 53: 1030–1042.
Simms ML, Kemper TL, Timbie CM, Bauman ML, Blatt GJ . The anterior cingulate cortex in autism: heterogeneity of qualitative and quantitative cytoarchitectonic features suggests possible subgroups. Acta Neuropathol 2009; 118: 673–684.
Denaxa M, Chan CH, Schachner M, Parnavelas JG, Karagogeos D . The adhesion molecule TAG-1 mediates the migration of cortical interneurons from the ganglionic eminence along the corticofugal fiber system. Development 2001; 128: 4635–4644.
Furley AJ, Morton SB, Manalo D, Karagogeos D, Dodd J, Jessell TM . The axonal glycoprotein TAG-1 is an immunoglobulin superfamily member with neurite outgrowth-promoting activity. Cell 1990; 61: 157–170.
Kogan JH, Frankland PW, Silva AJ . Long-term memory underlying hippocampus-dependent social recognition in mice. Hippocampus 2000; 10: 47–56.
Gordon A, Salomon D, Barak N, Pen Y, Tsoory M, Kimchi T et al. Expression of Cntnap2 (Caspr2) in multiple levels of sensory systems. Mol Cell Neurosci 2016; 70: 42–53.
Hoffman EJ, Turner KJ, Fernandez JM, Cifuentes D, Ghosh M, Ijaz S et al. Estrogens suppress a behavioral phenotype in zebrafish mutants of the autism risk gene, CNTNAP2. Neuron 2016; 89: 725–733.
Lancaster E, Huijbers MG, Bar V, Boronat A, Wong A, Martinez-Hernandez E et al. Investigations of caspr2, an autoantigen of encephalitis and neuromyotonia. Ann Neurol 2011; 69: 303–311.
Irani SR, Alexander S, Waters P, Kleopa KA, Pettingill P, Zuliani L et al. Antibodies to Kv1 potassium channel-complex proteins leucine-rich, glioma inactivated 1 protein and contactin-associated protein-2 in limbic encephalitis, Morvan's syndrome and acquired neuromyotonia. Brain 2010; 133: 2734–2748.
Acknowledgements
We thank the Simons Foundation and Elena Kowalsky at the Feinstein Institute for help obtaining blood samples. We also thank Dr Kevin J Tracey for helpful comments. The research was funded by the Department of Defense (AR130137), NIH (R43 MH106195), and The Nancy Lurie Marks Foundation and The Simons Foundation. LB is a recipient of a Brain and Behavior NARSAD Young Investigator Foundation Grant. We thank Dr. Czeslawa Kowal for her assistance in perfusing embryos.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Rights and permissions
About this article
Cite this article
Brimberg, L., Mader, S., Jeganathan, V. et al. Caspr2-reactive antibody cloned from a mother of an ASD child mediates an ASD-like phenotype in mice. Mol Psychiatry 21, 1663–1671 (2016). https://doi.org/10.1038/mp.2016.165
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2016.165
This article is cited by
-
Maternal brain reactive antibodies profile in autism spectrum disorder: an update
Translational Psychiatry (2023)
-
Sexually dimorphic neuroanatomical differences relate to ASD-relevant behavioral outcomes in a maternal autoantibody mouse model
Molecular Psychiatry (2021)
-
Autoantibodies in neurological disease
Nature Reviews Immunology (2021)
-
Risk assessment analysis for maternal autoantibody-related autism (MAR-ASD): a subtype of autism
Molecular Psychiatry (2021)
-
DNA methylation signatures in autism spectrum disorders
The Nucleus (2021)