Abstract
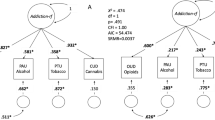
Genotype scores that predict relevant clinical outcomes may detect other disease features and help direct prevention efforts. We report data that validate a previously established v1.0 smoking cessation quit success genotype score and describe striking differences in the score in individuals who display differing developmental trajectories of use of common addictive substances. In a cessation study, v1.0 genotype scores predicted ability to quit with P=0.00056 and area under receiver-operating characteristic curve 0.66. About 43% vs 13% quit in the upper vs lower genotype score terciles. Latent class growth analyses of a developmentally assessed sample identified three latent classes based on substance use. Higher v1.0 scores were associated with (a) higher probabilities of participant membership in a latent class that displayed low use of common addictive substances during adolescence (P=0.0004) and (b) lower probabilities of membership in a class that reported escalating use (P=0.001). These results indicate that: (a) we have identified genetic predictors of smoking cessation success, (b) genetic influences on quit success overlap with those that influence the rate at which addictive substance use is taken up during adolescence and (c) individuals at genetic risk for both escalating use of addictive substances and poor abilities to quit may provide especially urgent focus for prevention efforts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Zheng J, Liu F, Lin X, Wang X, Ding Q, Jiang H et al. Predictive Performance of prostate cancer risk in Chinese men using 33 reported prostate cancer risk-associated SNPs. Prostate 2012; 72: 577–583.
Wang Y, Luk AO, Ma RC, So WY, Tam CH, Ng MC et al. Predictive role of multilocus genetic polymorphisms in cardiovascular disease and inflammation-related genes on chronic kidney disease in Type 2 diabetes–an 8-year prospective cohort analysis of 1163 patients. Nephrol Dial Transplant 2012; 27: 190–196.
Xu M, Tantisira KG, Wu A, Litonjua AA, Chu JH, Himes BE et al. Genome Wide Association Study to predict severe asthma exacerbations in children using random forests classifiers. BMC Med Genet 2011; 12: 90.
Kang J, Kugathasan S, Georges M, Zhao H, Cho JH . Improved risk prediction for Crohn’s disease with a multi-locus approach. Hum Mol Genet 2011; 20: 2435–2442.
Mealiffe ME, Stokowski RP, Rhees BK, Prentice RL, Pettinger M, Hinds DA et al. Assessment of clinical validity of a breast cancer risk model combining genetic and clinical information. J Natl Cancer Inst 2010; 102: 1618–1627.
Renstrom F, Shungin D, Johansson I MAGIC Investigators Florez JC, Hallmans G et al. Genetic predisposition to long-term nondiabetic deteriorations in glucose homeostasis: ten-year follow-up of the GLACIER study. Diabetes 2011; 60: 345–354.
Lu Y, Feskens EJ, Boer JM, Imholz S, Verschuren WM, Wijmenga C et al. Exploring genetic determinants of plasma total cholesterol levels and their predictive value in a longitudinal study. Atherosclerosis 2010; 213: 200–205.
Lluis-Ganella C, Lucas G, Subirana I, Sentí M, Jimenez-Conde J, Marrugat J et al. Additive effect of multiple genetic variants on the risk of coronary artery disease. Rev Esp Cardiol 2010; 63: 925–933.
Broms U, Silventoinen K, Madden PA, Heath AC, Kaprio J . Genetic architecture of smoking behavior: a study of Finnish adult twins. Twin Res Hum Genet 2006; 9: 64–72.
Lessov CN, Martin NG, Statham DJ, Todorov AA, Slutske WS, Bucholz KK et al. Defining nicotine dependence for genetic research: evidence from Australian twins. Psychol Med 2004; 34: 865–879.
Uhl GR, Liu QR, Drgon T, Johnson C, Walther D, Rose JE et al. Molecular genetics of successful smoking cessation: convergent genome-wide association study results. Arch Gen Psychiatry 2008; 65: 683–693.
Drgon T, Johnson C, Walther D, Albino AP, Rose JE, Uhl GR . Genome-wide association for smoking cessation success: participants in a trial with adjunctive denicotinized cigarettes. Mol Med 2009; 15: 268–274.
Uhl GR, Drgon T, Johnson C, Walther D, David SP, Aveyard P et al. Genome-wide association for smoking cessation success: participants in the Patch in Practice trial of nicotine replacement. Pharmacogenomics 2010; 11: 357–367.
Uhl GR, Drgon T, Johnson C, Ramoni MF, Behm FM, Rose JE . Genome-wide association for smoking cessation success in a trial of precessation nicotine replacement. Mol Med 2010; 16: 513–526.
Rose JE, Behm FM, Drgon T, Johnson C, Uhl GR . Personalized smoking cessation: interactions between nicotine dose, dependence and quit-success genotype score. Mol Med 2010; 16: 247–253.
Morley KI, Lynskey MT, Madden PA, Treloar SA, Heath AC, Martin NG . Exploring the inter-relationship of smoking age-at-onset, cigarette consumption and smoking persistence: genes or environment? Psychol Med 2007; 37: 1357–1367.
Lynne-Landsman SD, Graber JA, Nichols TR, Botvin GJ . Trajectories of aggression, delinquency, and substance use across middle school among urban, minority adolescents. Aggress Behav 2011; 37: 161–176.
Lynne-Landsman SD, Bradshaw CP, Ialongo NS . Testing a developmental cascade model of adolescent substance use trajectories and young adult adjustment. Dev Psychopathol 2010; 22: 933–948.
Marsiglia FF, Kulis S, Yabiku ST, Nieri TA, Coleman E . When to intervene: elementary school, middle school or both? Effects of keepin' it REAL on substance use trajectories of Mexican heritage youth. Prev Sci 2011; 12: 48–62.
Drgon T, Zhang PW, Johnson C, Walther D, Hess J, Nino M et al. Genome wide association for addiction: replicated results and comparisons of two analytic approaches. PloS one 2010; 5: e8832.
Coon KD, Myers AJ, Craig DW, Webster JA, Pearson JV, Lince DH et al. A high-density whole-genome association study reveals that APOE is the major susceptibility gene for sporadic late-onset Alzheimer's disease. J Clin Psychiatry 2007; 68: 613–618.
Kellam SG, Brown CH, Poduska JM, Ialongo NS, Wang W, Toyinbo P et al. Effects of a universal classroom behavior management program in first and second grades on young adult behavioral, psychiatric, and social outcomes. Drug Alcohol Depend 2008; 95 (Suppl 1): S5–S28.
Wang Y, Browne DC, Petras H, Stuart EA, Wagner FA, Lambert SF et al. Depressed mood and the effect of two universal first grade preventive interventions on survival to the first tobacco cigarette smoked among urban youth. Drug Alcohol Depend 2009; 100: 194–203.
Ialongo NS, Poduska JM, Werthamer-Larsson L . The distal impact of two first grade preventive interventions on conduct problems and disorder and mental health service need and utilization in early adolescence. J Emotional Behav Disord 2001; 9: 146–160.
Kellam SG, Werthamer-Larsson L, Dolan LJ, Brown CH, Mayer LS, Rebok GW et al. Developmental epidemiologically based preventive trials: baseline modeling of early target behaviors and depressive symptoms. Am J Community Psychol 1991; 19: 563–584.
Uhl GR, Liu QR, Walther D, Hess J, Naiman D . Polysubstance abuse-vulnerability genes: genome scans for association, using 1004 subjects and 1494 single-nucleotide polymorphisms. Am J Hum Genet 2001; 69: 1290–1300.
Smith SS, O’Hara BF, Persico AM, Gorelick DA, Newlin DB, Vlahov D et al. Genetic vulnerability to drug abuse. The D2 dopamine receptor Taq I B1 restriction fragment length polymorphism appears more frequently in polysubstance abusers. Arch Gen Psychiatry 1992; 49: 723–727.
Persico AM, Bird G, Gabbay FH, Uhl GR . D2 dopamine receptor gene TaqI A1 and B1 restriction fragment length polymorphisms: enhanced frequencies in psychostimulant-preferring polysubstance abusers. Biol Psychiatry 1996; 40: 776–784.
Dupont WD, Plummer WD . Power and sample size calculations. A review and computer program. Control Clin Trials 1990; 11: 116–128.
Dupont WD, Plummer WD . Power and sample size calculations for studies involving linear regression. Control Clin Trials 1998; 19: 589–601.
Muthen B, Muthen LK . Integrating person-centered and variable-centered analyses: growth mixture modeling with latent trajectory classes. Alcohol Clin Exp Res 2000; 24: 882–891.
Morley KI, Lynskey MT, Madden PA, Treloar SA, Heath AC, Martin NG . Exploring the inter-relationship of smoking age-at-onset, cigarette consumption and smoking persistence: genes or environment? Psychol Med 2007; 37: 1357–1367.
Broms U, Silventoinen K, Madden PA, Heath AC, Kaprio J . Genetic architecture of smoking behavior: a study of Finnish adult twins. Twin Res Hum Genet 2006; 9: 64–72.
Uhl GR, Drgon T, Johnson C, Rose JE . Nicotine abstinence genotyping: assessing the impact on smoking cessation clinical trials. Pharmacogenomics J 2009; 9: 111–115.
Acknowledgements
This study was supported by the National Institutes of Health (NIH)–Intramural Research Program, National Institute on Drug Abuse, Department of Health and Human Services (Dr Uhl); a grant to Duke University (PI, Dr Rose) from Philip Morris, USA for the work performed before January 2012; NIDA grants R01-DA009897 (WE) and 4R37DA011796-11 (NI). The funders had no role in the planning or execution of the study, data analysis or publication of results. We are grateful to TGEN investigators for generous access to Alzheimer’s disease GWAS genotype data and D Sisto for its analysis, to E Westman for assistance with the smoking cessation clinical trial, for each of the prevention study investigators, especially S Kellam, for the sustained cooperation of the study participants and for help and thoughtful advice from C Johnson, J Schroder, P Zandi and K Masyn. The underlying smoking cessation clinical trial was registered with clinicaltrials.gov (NCT00894166).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Drs Rose and Uhl are listed as inventors for a patent application filed by Duke University based on genomic markers that distinguish successful quitters from unsuccessful quitters in data from other clinical trials.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Supplementary information
Rights and permissions
About this article
Cite this article
Uhl, G., Walther, D., Musci, R. et al. Smoking quit success genotype score predicts quit success and distinct patterns of developmental involvement with common addictive substances. Mol Psychiatry 19, 50–54 (2014). https://doi.org/10.1038/mp.2012.155
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2012.155
Keywords
This article is cited by
-
Polygenic Score × Intervention Moderation: an Application of Discrete-Time Survival Analysis to Model the Timing of First Marijuana Use Among Urban Youth
Prevention Science (2018)
-
Biomarkers of Response to Smoking Cessation Pharmacotherapies: Progress to Date
CNS Drugs (2015)
-
Interaction between polygenic risk for cigarette use and environmental exposures in the Detroit neighborhood health study
Translational Psychiatry (2013)