Abstract
The cancer stem cell hypothesis may explain why conventional chemotherapies are unable to fully eradicate cancers. In this study, we examined both the prognostic and predictive significance of putative cancer stem cell markers in colorectal cancer. In this study, immunohistochemistry for three candidate cancer stem cell markers (CD133, Oct-4 and Sox-2) and for six other postulated prognostic markers (CK7, CK20, Cox-2, Ki-67, p27 and p53) were performed using tissue microarrays containing 501 primary colorectal cancer cases. Receiver-operating characteristic analysis was used to determine cut-off scores for positive protein expression. Multivariate analysis revealed that positive expression for CD133 and Oct-4 was associated with significantly worse survival in patients treated by surgery alone (P=0.023 and P<0.001, respectively) and in patients treated with 5-fluorouracil-based chemotherapy (P=0.001 and P=0.021, respectively). Stage III patients with negative CD133 expression showed an apparent survival benefit from 5-fluorouracil treatment (P=0.002), but not those with positive CD133 expression. Positive expression of CD133 was also associated with poorer clinical response to chemotherapy in stage IV patients (P=0.006). In summary, the putative cancer stem cell markers CD133 and Oct-4 showed strong prognostic significance in colorectal cancer. Our results show for the first time that CD133+ colorectal tumors are more resistant to 5-fluorouracil-based chemotherapy.
Similar content being viewed by others
Main
Colorectal cancer is one of the leading causes of cancer-related deaths in developed societies. Besides surgical procedures, adjuvant chemotherapy is often administered to improve survival, especially in advanced stages of the disease. In spite of the availability of new chemotherapeutic drugs, none of these treatments is fully curative, leading to a high failure rate and relatively short survival. This has raised questions regarding whether current treatments target the appropriate molecular mechanisms or tumor cell populations that are responsible for the growth and survival of these cells.1, 2
The past few years have observed an accumulation of experimental evidence in support of the hypothesis that stem cells have a major role in cancer progression. These so called cancer stem cells have self-renewing abilities and are thought to drive tumor growth.3 The cancer stem cells concept may explain the failure of current adjuvant therapies to completely eradicate metastatic colorectal cancer. This is because conventional cytotoxic chemotherapeutic agents target actively proliferating tumor cells, thus allowing the more slowly proliferating cancer stem cells subpopulation to escape death.4 As normal stem cells have multidrug resistance properties, it is conceivable that cancer stem cells may also possess similar drug resistance features.4 Consequently, the cancer stem cells compartment is increasingly being recognized as a necessary target for the effective treatment of cancers.4
One of the better studied cancer stem cells markers that has been implicated in several solid tumor types including colorectal cancer is CD133.5, 6, 7 This surface protein was reported to show a characteristic expression pattern with localization to the luminal surface of the colonic gland.7, 8, 9 Another distinctive feature of CD133 positive cells was simultaneous loss of expression of CK20, a marker of epithelial cell differentiation.7, 8 More importantly, CD133 expression has been linked to in vitro chemoresistance. CD133 positive hepatocellular carcinoma and glioblastoma cell lines show increased expression of BCRP1, a putative drug resistance protein.10, 11 The phosphorylation of Akt and subsequent accumulation of anti-apoptotic signals in Akt/PKB survival pathways have also been suggested to contribute to the chemoresistance of CD133 positive tumor cells.12 Maintenance of CD133 stem cell-like properties was recently shown to be regulated by a homeobox protein, Oct-4.13 Oct-4 has a functional role in reprogramming adult neural stem cells into pluripotent stem cells.14 In addition, Sox-2, a transcription factor involved in regulating Oct-4 expression during the maintenance of pluripotency in embryonic stem cells, has also been shown to act synergistically with Oct-4 to promote the survival of cancer cells.14
The primary aim of this study was to evaluate the prognostic and predictive significance of CD133, Oct-4 and Sox-2 expression in a well-characterized cohort of 501 primary colorectal cancer cases. Although independent prognostic significance of CD133 expression in colorectal cancer was recently reported,8 the associations of Oct-4 and Sox-2 with patient survival are currently unknown. Moreover, despite encouraging results from in vitro studies,15 the predictive significance of CD133 expression for response to chemotherapy in a clinical setting has so far not been reported. Therefore, in this study, we analyzed both the prognostic and predictive significance of three candidate cancer stem cells markers in colorectal cancer and compared these against a panel of six commonly studied protein expression markers in colorectal cancer (CK7, CK20, Cox-2, Ki-67, p27 and p53).
Materials and methods
Clinicopathological Data
The study cohort consisted of 501 consecutive colorectal cancer cases treated by surgical resection at the National University Hospital of Singapore between 1990 and 1999 inclusive. The available clinicopathological information included gender, age, tumor size, tumor stage, histological grade, vascular invasion, perineural invasion, lymphatic invasion and cancer-specific survival time. Clinicopathological data were documented at the initial diagnosis and during subsequent follow-up. Staging was based on pathological findings according to the American Joint Committee on Cancer classification.16 This work was approved by the ethics committee of the National University of Singapore (DSRB Domain B/09/284).
Treatment Schedule and Assessment of Response
Cases treated with chemotherapy underwent intravenous injection with 5-fluorouracil using the Mayo regimen. One complete cycle of treatment involved dosage administration ranging from 500 to 900 mg/m2/day for 3 to 5 consecutive days. Each cycle was repeated monthly for 6 months or until progression of disease, patient refusal or adverse reactions to the treatment. Tumor response to treatment was classified according to the standard World Health Organization response criteria.17
Tissue Microarrays
Tissue microarray blocks containing cores from 501 consecutive, primary colorectal cancer cases were constructed as described earlier.18, 19 Briefly, a needle with 0.6-mm diameter was used to punch a donor core from morphologically representative areas of a donor tissue block. The core was subsequently inserted into a recipient paraffin block using an ATA-100 tissue arrayer (Chemicon, USA). Three cores were taken from the center of tumor tissue and a single core was taken from histologically normal colon epithelium of matched cases. Consecutive tissue microarray sections of 4-μm thickness were cut and placed on slides for immunohistochemical analyses.
Immunohistochemical Procedures
All nine protein markers were assessed immunohistochemically using commercially available antibodies. Antigen retrieval was carried out with 10 mM citrate buffer (pH 6.0) in a MicroMED TT Microwave Processor (Milestone, Sorisole, Italy) for 5 min at 120 °C. Slides were then incubated with the primary antibody for 12 h at the dilutions indicated in Supplementary Table S1. Immunostaining was performed using the streptavidin-biotin kit (LSAB2, Dako, Norway) in accordance with the manufacturer's specifications and the slides were then counterstained with hematoxylin. Various human tissues or cell lines embedded in paraffin with known expression for the markers were used as positive controls. Paraffin-embedded CaCo2 cells were positive control for CD1338 and Sox-2.20 For Oct-4, human germinoma tissues were used as positive control.21 Negative controls consisted of the omission of primary antibody without any other changes to subsequent procedures.
Scoring for Protein Expression
Scoring for all nine proteins was performed using an automated imaging and scoring platform (Ariol SL-50, Applied Imaging, San-Jose, CA, USA). We have reported a high level of concordance (kappa value >0.9) between objective (automated) and subjective (manual human-observed) scoring methods for immunohistochemistry in a technical journal.22
The cut-off scores for determining positive expression for each protein were determined by receiver-operating characteristic curve analysis as outlined previously.23 Briefly, receiver-operating characteristic curves were constructed by plotting all possible sensitivity and specificity pairs. This allows selection of a threshold value that corresponds to maximum sensitivity with minimal loss of specificity. For receiver-operating characteristic analysis, scores above the cut-off were considered positive for protein expression and scores below as negative. Receiver-operating characteristic analysis also allows the identification of markers that possess discriminatory value for the determination of prognostic significance through the area under the receiver-operating characteristic curve values. Similar to the previous study,22 the clinicopathological features were dichotomized on the basis of having or not having the clinical outcome. The parameters examined include gender (female vs male), age (≥60 vs <60 years), ethnicity (Chinese vs non-Chinese), histology grade (poor vs well–moderate), staging (III–IV vs I–II), tumor site (colon vs rectal), tumor size (≥5 vs <5 cm), lymphatic invasion (yes vs no), perineural invasion (yes vs no), vascular invasion (yes vs no) and survival (death from colorectal cancer vs lost to follow-up, alive and death from other causes).
Statistical Analysis
Univariate Cox proportional hazards regression was performed to determine the prognostic value of various clinicopathological features and protein markers. To test for independence, the significant factors identified in univariate analyses were entered into a multivariate Cox regression model using a stepwise selection procedure. Survival curves were plotted using the Kaplan–Meier method and compared using the log-rank test. All statistical analyses were performed using the SPSS package (version 15.0 for Windows, SPSS, USA) with significance set at the 5% level.
Results
Protein Expression of Markers
Representative immunohistochemical stains for the three cancer stem cells proteins investigated in this study are shown in Figure 1. CD133 expression was localized to the glandular-luminal surface of tumor epithelial cells. Similar to previous study,8 the intraglandular debris of shed tumor cells also showed CD133 staining. CD133 expression in the normal colonic mucosa was absent in the majority of cases or was very mild and focal in intensity in the remainder. For both Oct-4 and Sox-2 expression, diffuse staining expression was observed in the cytoplasmic compartment of tumor epithelium. Similar staining patterns of Oct-4 and Sox-2 in rectal carcinomas have been recently reported.20 Mild cytoplasmic staining was observed in the normal colonic epithelium of a minority of the samples.
The selection of cut-off scores for defining positive protein expression was based on receiver-operating characteristic curve analysis against 5-year survival. The frequency of positive expression for each marker is shown in Table 1. Using values derived from the area under the receiver-operating characteristic curve, markers with area under the receiver-operating characteristic curve values above 0.5 indicate significant discriminatory power for survival. The three cancer stem cells markers CD133, Oct-4 and Sox-2 were all identified as having discriminatory power (area under the receiver-operating characteristic curve value >0.5). A total of 27%, 39% and 48% of patients were classified as showing positive expression for CD133, Oct-4 and Sox-2, respectively. Among the six other commonly studied protein markers in colorectal cancer, only p27 expression was identified as having significant discriminatory power for survival.
Correlations between the expressions of each marker are shown in Supplementary Table S2. Similar to previous reports,7, 8 a strong inverse correlation was found between the expression of CD133 and CK20 (Kendall's ranking coefficient=−0.68, P<0.001). The expression of CD133 and Oct-4 were both positively associated with expression of the third cancer stem cells marker, Sox-2 (Kendall's ranking coefficient of 0.24 and 0.31, respectively).
Associations between clinicopathological features and expression of the three cancer stem cells markers are shown in Supplementary Table S3. The expression of all three cancer stem cells markers showed significant associations with higher tumor stage (P<0.05), but not with any of the other clinicopathological features.
Prognostic Significance of Protein Expression
Table 2 shows the results of univariate analysis for the prognostic significance of clinicopathological features and protein expression in patients treated by surgery alone (n=310) or with 5-fluorouracil-based adjuvant chemotherapy (n=191). As expected, older patient age, poor histological grade, higher tumor stage and the presence of lymphatic, perineural and vascular invasion were all associated with worse survival in patients treated by surgery alone. CD133, Oct-4 and Sox-2 expression were also strongly associated with poor prognosis in patients treated by surgery alone, whereas p27 expression was a marker of good prognosis. All three cancer stem cells markers were also prognostic for worse survival in the subgroup of patients treated with chemotherapy.
The significant parameters identified from univariate analysis were tested for independent prognostic value in a multivariate Cox regression analysis model. Patient age, tumor stage, vascular invasion and the expression of CD133, Oct-4 and p27 were all found to be independent prognostic factors in patients treated by surgery alone (Table 3). For patients treated with chemotherapy, only tumor stage and the expression of CD133 and Oct-4 were independent factors for survival.
The survival of patient subgroups classified according to the expression of protein markers was evaluated using Kaplan–Meier analysis. Patients treated by surgery alone and with CD133+/Oct-4+/p27– tumors showed a mean survival of just 15 months compared with 50 months for those with CD133–/Oct-4–/p27+ tumors (P<0.001).
Predictive Significance of Protein Expression
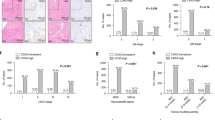
Kaplan–Meier analysis was used to compare the survival rates of stage III patients treated with or without chemotherapy and classified according to the expression of cancer stem cells proteins (Figure 2). 5-fluorouracil-based chemotherapy conferred an apparent survival benefit to patients regardless of their expression status for Oct-4 or Sox-2 (Figures 2c–f). Although patients with CD133– tumors gained significant benefit from chemotherapy (Figure 2a), no survival benefit was observed for patients with CD133+ tumors (Figure 2b), suggesting that CD133 expression is a predictive marker for survival benefit from 5-fluorouracil-based adjuvant chemotherapy.
Kaplan–Meier survival analysis of stage III colorectal cancer patients according to negative (a, c, e) or positive (b, d, f) expression of CD133, Oct-4 and Sox-2, respectively. Broken lines represent patients treated by surgery alone, while continuous lines represent patients treated with adjuvant chemotherapy in addition to surgery. An apparent survival benefit from chemotherapy is observed for all patient subgroups except those with positive CD133 expression.
Clinical data for response to chemotherapy were available for 44 stage IV patients. Using standardized criteria, half of these patients (n=22) showed clinical response to treatment and the other half did not (n=22). Of the patients with CD133+ tumors, only 7/24 (29%) showing response compared with 15/20 (75%) of the patients with CD133– tumors (P=0.006). None of the other markers showed any significant association between expression and clinical response.
Discussion
Recent reports have suggested the existence of a cancer stem cells compartment that can self-renew and differentiate into mature and diverse cancer cells capable of tumor initiation, growth, invasion and metastasis.3, 24 As cancer stem cells divide very slowly, it is believed they may be resistant to most of the current chemotherapies that target differentiated or highly proliferating tumor cells.4 The results from several in vitro studies on various cancer cell lines support this concept,11, 12, 15, 25 although to date there has not been any evidence from clinical studies. In this work, we provide the first evidence that expression of the cancer stem cells marker CD133 is associated not only with poor prognosis in colorectal cancer, but also with a poor response of these tumors to 5-fluorouracil-based chemotherapy.
Three studies have isolated tumor initiating cell populations from colorectal cancer that express the surface marker CD133 and possess apparent ‘stem cell-like’ properties.6, 7, 26 Similar to previous reports in colorectal cancer,5, 7, 8 CD133 expression in this study was found to correlate inversely with expression of the intestinal epithelial differentiation marker CK20 (Supplementary Table S2) and to localize to the glandular-luminal surface of tumor epithelial cells (Figure 1a). Using receiver-operating characteristic analysis, we estimated that 27% of colorectal cancers were CD133+ (Table 1). This compares with earlier reports for the frequency of CD133+ colorectal cancer as 258 and 15%, respectively.27 CD133 expression was an independent prognostic indicator in this study (Table 3), confirming the results of three previous studies in colorectal cancer.8, 9, 27 Oct-4 expression was shown here for the first time to have independent prognostic significance, particularly in patients treated by surgery alone (Table 3). The good prognosis associated with p27 expression confirms previous reports with this marker.28
The major finding of this study was that colorectal cancers with the CD133+ immunophenotype show resistance to 5-fluorouracil-based chemotherapy. Two separate lines of evidence were obtained in support of this. First, stage III colorectal cancer patients with CD133+ tumors did not show any apparent survival benefit from chemotherapy (Figure 2b), in contrast to patients with CD133– tumors (Figure 2a) and with all Oct-4 and Sox-2 expression subgroups (Figures 2c–f). Second, stage IV patients with CD133+ tumors showed significantly less clinical response to chemotherapy (29%) compared with patients with CD133– tumors (75%, P<0.006). Together, these results suggest that CD133 expression could be a clinically useful marker of chemoresistance in colorectal cancer and perhaps also in other tumor types.
Our observations in a clinical tumor series concur with recent findings from an in vitro study in which 5-fluorouracil and oxaliplatin-resistant clones of the HT29 colorectal cancer cell line showed 16- and 30-fold increases, respectively, in CD133 expression compared with non-resistant cells.15 These results suggest that CD133 expression may be predictive for a wide range of cytotoxic agents used in cancer treatment. Indeed, several other in vitro studies have reported chemoresistance of CD133+ ovarian cancer,25 glioblastoma11 and hepatocellular carcinoma12 cell lines to a variety of chemotherapeutic agents. The expression of CD133 has been used to isolate putative cancer stem cells from tumors of the liver,29, 30 pancreas,31 prostate,32 brain33 and lung,34 in addition to colorectal cancer. These findings suggest that CD133+ subgroups from other tumor types may also be chemoresistant.
The mechanism responsible for the resistance of CD133+ cells to conventional therapies remains to be elucidated. One area for further investigation is to define the molecular phenotype of CD133+ colorectal cancer in terms of features such as TP53 and KRAS mutation, microsatellite instability and the CpG island methylator phenotype. Future work should also examine whether other putative cancer stem cells markers such as CD166,35 EpCAM,36 CD44,15 Musashi-1,37 Lgr538 or OLFM439 have predictive value for chemoresistance in colorectal cancer and in other tumor types. It may be that combinations of such markers will show the strongest predictive value for response to chemotherapy. The cut-off for defining CD133+ tumors in this study was set at 10% based on the discriminating value of this threshold for prognostic significance (Table 1). However, for optimal discrimination of predictive significance, other threshold levels may be more suitable. Exploratory investigations indicated that a 5% cut-off value for CD133+ retained strong predictive value (results not shown).
One of the limitations of this study was the relatively small sample size, particularly for stage IV colorectal cancer cases (n=44). Another was that the use of adjuvant chemotherapy for stage III patients was not randomized, thus introducing possible confounding factors. All patient subgroups defined by cancer stem cells expression and that received chemotherapy showed better survival compared with corresponding patients treated by surgery alone, except for those with CD133+ tumors (Figure 2). Nevertheless, this observation requires confirmation in larger, randomized patient cohorts before CD133 expression can be used in a clinical setting.
In summary, tumor expression of the two cancer stem cells markers CD133 and Oct-4 was associated with worse survival of colorectal cancer patients. Our findings also show for the first time that CD133+ colorectal cancer tumors are more resistant to 5-fluorouracil-based chemotherapy. Confirmation of this observation in independent colorectal cancer cohorts could lead to better targeting of conventional cytotoxic therapies toward patient subgroups that are most likely to benefit.
References
Boman BM, Wicha MS, Fields JZ, et al. Symmetric division of cancer stem cells—a key mechanism in tumor growth that should be targeted in future therapeutic approaches. Clin Pharmacol Ther 2007;81:893–898.
Dalerba P, Cho RW, Clarke MF . Cancer stem cells: models and concepts. Annu Rev Med 2007;58:267–284.
Reya T, Morrison SJ, Clarke MF, et al. Stem cells, cancer, and cancer stem cells. Nature 2001;414:105–111.
Boman BM, Huang E . Human colon cancer stem cells: a new paradigm in gastrointestinal oncology. J Clin Oncol 2008;26:2828–2838.
Horst D, Kriegl L, Engel J, et al. Prognostic significance of the cancer stem cell markers CD133, CD44, and CD166 in colorectal cancer. Cancer Invest 2009;27:844–850.
O’Brien CA, Pollett A, Gallinger S, et al. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature 2007;445:106–110.
Ricci-Vitiani L, Lombardi DG, Pilozzi E, et al. Identification and expansion of human colon-cancer-initiating cells. Nature 2007;445:111–115.
Horst D, Kriegl L, Engel J, et al. CD133 expression is an independent prognostic marker for low survival in colorectal cancer. Br J Cancer 2008;99:1285–1289.
Horst D, Kriegl L, Engel J, et al. CD133 and nuclear beta-catenin: the marker combination to detect high risk cases of low stage colorectal cancer. Eur J Cancer 2009;45:2034–2040.
Smith LM, Nesterova A, Ryan MC, et al. CD133/prominin-1 is a potential therapeutic target for antibody-drug conjugates in hepatocellular and gastric cancers. Br J Cancer 2008;99:100–109.
Liu G, Yuan X, Zeng Z, et al. Analysis of gene expression and chemoresistance of CD133+ cancer stem cells in glioblastoma. Mol Cancer 2006;5:67.
Ma S, Lee TK, Zheng BJ, et al. CD133+ HCC cancer stem cells confer chemoresistance by preferential expression of the Akt/PKB survival pathway. Oncogene 2008;27:1749–1758.
Chen YC, Hsu HS, Chen YW, et al. Oct-4 expression maintained cancer stem-like properties in lung cancer-derived CD133-positive cells. PLoS ONE 2008;3:e2637.
Kim JB, Zaehres H, Wu G, et al. Pluripotent stem cells induced from adult neural stem cells by reprogramming with two factors. Nature 2008;454:646–650.
Dallas NA, Xia L, Fan F, et al. Chemoresistant colorectal cancer cells, the cancer stem cell phenotype, and increased sensitivity to insulin-like growth factor-I receptor inhibition. Cancer Res 2009;69:1951–1957.
Greene FL, American Joint Committee on Cancer. American Cancer Society. AJCC Cancer Staging Manual 6th edn Springer, SpVerlag: New York, 2002.
Therasse P, Arbuck SG, Eisenhauer EA, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 2000;92:205–216.
Das K, Mohd Omar MF, Ong CW, et al. TRARESA: a tissue microarray-based hospital system for biomarker validation and discovery. Pathology 2008;40:441–449.
Salto-Tellez M, Nga ME, Han HC, et al. Tissue microarrays characterise the clinical significance of a VEGF-A protein expression signature in gastrointestinal stromal tumours. Br J Cancer 2007;96:776–782.
Saigusa S, Tanaka K, Toiyama Y, et al. Correlation of CD133, OCT4, and SOX2 in rectal cancer and their association with distant recurrence after chemoradiotherapy. Ann Surg Oncol 2009;16:3488–3498.
Hattab EM, Tu PH, Wilson JD, et al. OCT4 immunohistochemistry is superior to placental alkaline phosphatase (PLAP) in the diagnosis of central nervous system germinoma. Am J Surg Pathol 2005;29:368–371.
Ong CW, Kim LG, Kong HH, et al. Computer-assisted pathological immunohistochemistry scoring is more time-effective than conventional scoring, but provides no analytical advantage. Histopathology 2009, in press.
Zlobec I, Steele R, Terracciano L, et al. Selecting immunohistochemical cut-off scores for novel biomarkers of progression and survival in colorectal cancer. J Clin Pathol 2007;60:1112–1116.
Presnell SC, Petersen B, Heidaran M . Stem cells in adult tissues. Semin Cell Dev Biol 2002;13:369–376.
Baba T, Convery PA, Matsumura N, et al. Epigenetic regulation of CD133 and tumorigenicity of CD133+ ovarian cancer cells. Oncogene 2009;28:209–218.
Todaro M, Alea MP, Di Stefano AB, et al. Colon cancer stem cells dictate tumor growth and resist cell death by production of interleukin-4. Cell Stem Cell 2007;1:389–402.
Kojima M, Ishii G, Atsumi N, et al. Immunohistochemical detection of CD133 expression in colorectal cancer: a clinicopathological study. Cancer Sci 2008;99:1578–1583.
Zlobec I, Minoo P, Baumhoer D, et al. Multimarker phenotype predicts adverse survival in patients with lymph node-negative colorectal cancer. Cancer 2008;112:495–502.
Suetsugu A, Nagaki M, Aoki H, et al. Characterization of CD133+ hepatocellular carcinoma cells as cancer stem/progenitor cells. Biochem Biophys Res Commun 2006;351:820–824.
Yin S, Li J, Hu C, et al. CD133 positive hepatocellular carcinoma cells possess high capacity for tumorigenicity. Int J Cancer 2007;120:1444–1450.
Hermann PC, Huber SL, Herrler T, et al. Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell 2007;1:313–323.
Collins AT, Berry PA, Hyde C, et al. Prospective identification of tumorigenic prostate cancer stem cells. Cancer Res 2005;65:10946–10951.
Singh SK, Hawkins C, Clarke ID, et al. Identification of human brain tumour initiating cells. Nature 2004;432:396–401.
Eramo A, Lotti F, Sette G, et al. Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death Differ 2008;15:504–514.
Botchkina IL, Rowehl RA, Rivadeneira DE, et al. Phenotypic subpopulations of metastatic colon cancer stem cells: genomic analysis. Cancer Genomics Proteomics 2009;6:19–29.
Fields AL, Keller A, Schwartzberg L, et al. Adjuvant therapy with the monoclonal antibody Edrecolomab plus fluorouracil-based therapy does not improve overall survival of patients with stage III colon cancer. J Clin Oncol 2009;27:1941–1947.
Johannessen TC, Wang J, Skaftnesmo KO, et al. Highly infiltrative brain tumours show reduced chemosensitivity associated with a stem cell-like phenotype. Neuropathol Appl Neurobiol 2009;35:380–393.
Vermeulen L, Todaro M, de Sousa Mello F, et al. Single-cell cloning of colon cancer stem cells reveals a multi-lineage differentiation capacity. Proc Natl Acad Sci USA 2008;105:13427–13432.
Liu W, Liu Y, Zhu J, et al. Reduced hGC-1 protein expression is associated with malignant progression of colon carcinoma. Clin Cancer Res 2008;14:1041–1049.
Acknowledgements
This study is funded by Singapore Cancer Syndicate, Agency for Science, Technology and Research, Singapore, and the Grants numbers are MN005, MN005R and MN077.
Author information
Authors and Affiliations
Corresponding author
Additional information
Disclosure/conflict of interest
The authors declare no conflict of interest.
Supplementary Information accompanies the paper on Modern Pathology website (http://www.nature.com/modpathol)
Supplementary information
Rights and permissions
About this article
Cite this article
Ong, C., Kim, L., Kong, H. et al. CD133 expression predicts for non-response to chemotherapy in colorectal cancer. Mod Pathol 23, 450–457 (2010). https://doi.org/10.1038/modpathol.2009.181
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2009.181
Keywords
This article is cited by
-
Treatment with decitabine induces the expression of stemness markers, PD-L1 and NY-ESO-1 in colorectal cancer: potential for combined chemoimmunotherapy
Journal of Translational Medicine (2023)
-
PCGF1 promotes epigenetic activation of stemness markers and colorectal cancer stem cell enrichment
Cell Death & Disease (2021)
-
An integrated magneto-electrochemical device for the rapid profiling of tumour extracellular vesicles from blood plasma
Nature Biomedical Engineering (2021)
-
Analysis of the Correlation between CD133 Expression on Human Colorectal Adenocarcinoma Cells HT-29 and Their Resistance to Chemotherapeutic Drugs
Bulletin of Experimental Biology and Medicine (2021)
-
Stroma-normalised vessel density predicts benefit from adjuvant fluorouracil-based chemotherapy in patients with stage II/III colon cancer
British Journal of Cancer (2019)