Abstract
Duchenne muscular dystrophy (DMD), a progressive muscle-wasting disease, is mostly caused by exon deletion mutations in the DMD gene. The reading frame rule explains that out-of-frame deletions lead to muscle dystrophin deficiency in DMD. In outliers to this rule, deletion junction sequences have never previously been explored as splicing modulators. In a Japanese case, we identified a single exon 45 deletion in the patient’s DMD gene, indicating out-of-frame mutation. However, immunohistochemical examination disclosed weak dystrophin signals in his muscle. Reverse transcription-PCR amplification of DMD exons 42 to 47 revealed a major normally spliced product with exon 45 deletion and an additional in-frame product with deletion of both exons 44 and 45, indicating upstream exon 44 skipping. We considered the latter to underlie the observed dystrophin expression. Remarkably, the junction sequence cloned by PCR walking abolished the splicing enhancer activity of the upstream intron in a chimeric doublesex gene pre-mRNA in vitro splicing. Furthermore, antisense oligonucleotides directed against the junction site counteracted this effect. These indicated that the junction sequence was a splicing silencer that induced upstream exon 44 skipping. It was strongly suggested that creation of splicing regulator is a modifier of dystrophinopathy.
Similar content being viewed by others
Introduction
Duchenne muscular dystrophy (DMD) and Becker muscular dystrophy (BMD) are the most common forms of inherited myopathy. DMD is a rapidly progressive muscle-wasting disease that usually causes loss of ambulation before the age of 12 years and results in the death of patients in their twenties. In contrast, BMD is a clinically less severe form of the disease and often has only a slightly debilitating effect. DMD/BMD are caused by mutations in the DMD gene that consists of 79 exons spanning 2500 kb on the X chromosome. Deletion of one or more exons is the most frequent mutation. The difference between DMD and BMD can be explained by the reading frame rule: out-of-frame mutations that generate a premature stop codon in the coding sequence of DMD mRNA usually result in the DMD phenotype, whereas in-frame mutations that maintain the original reading frame cause the milder BMD phenotype.1 The reading frame rule has been reported to be upheld for >90% of deletions.2, 3 However, there remains an unanswered question regarding this rule: there are some exceptions in which out-of-frame mutations result in BMD and vice versa.4, 5
There are two deletion hot spots in the DMD gene, and the region encompassing exons 44 to 55 is the major hot spot.6 In this region, many deletion types differing in their number or location of deleted exons have been identified in both DMD and BMD. Deletion of the single exon 45 is the most common deletion type identified in the DMD gene worldwide.3, 6, 7, 8 Because exon 45 is 176 bp long, its deletion is calculated to produce out-of-frame mRNA and is expected to result in DMD. However, as outliers from the reading frame rule, BMD cases with a single exon 45 deletion have been reported.6, 8 The reason why the BMD phenotype develops in the presence of an out-of-frame deletion mutation is not yet clear.
Splicing, which removes intron sequences from pre-mRNA, is a highly regulated process that assembles the ordered array of exons in mature transcripts.9, 10 Splicing depends on the correct identification of exons that must be recognized within the pre-mRNA despite being relatively short compared with introns. The presence of well-defined cis-elements, namely the splice donor and acceptor sites and the branch point, are necessary but not sufficient to define intron–exon boundaries.11 In addition, splicing is regulated by sequences present within exons or introns. Sequences that promote exon recognition are known as splicing enhancers, whereas those that suppress recognition are known as splicing silencers.12 It is possible that a novel deletion junction in the DMD gene might acquire splicing regulatory activity, thereby producing alternatively spliced mRNA.
In this study, we revealed the natural occurrence of exon 44 skipping in a dystrophinopathy patient with an exon 45 deletion. The cloned junction sequence suppressed upstream intron splicing in an in vitro splicing assay using a chimeric doublesex gene pre-mRNA. We propose that the novel junction causes in vivo exon skipping.
Materials and methods
Case
A Japanese boy was referred to Kobe University Hospital (KUCG#565) at the age of 10 years because of muscle weakness. He was born to healthy Japanese parents. He started to walk independently from 1.5 years and could run. At the age of 5 years, walking disability was noticed. At age 10 years, he showed the Gowers’ sign and his serum creatine kinase was 9271 IU l−1 (normal <200 IU l−1). He was clinically diagnosed as DMD. He maintained independent ambulation beyond the age of 12 years but became wheelchair-bound at age 18 years. To confirm the diagnosis, mutation analysis of the DMD gene and dystrophin immunohistochemical examination on skeletal muscle were conducted. The following study was conducted after obtaining informed consent. The study protocol was approved by the ethics committee of Kobe University School of Medicine (no. 28).
Immunohistochemical examination of skeletal muscle
Skeletal muscle samples were obtained from the biceps by biopsy. The muscle biopsy sample was examined histologically. Indirect immunofluorescence analysis was performed using a monoclonal antibody that recognizes the C-terminal domain of dystrophin (NCL-Dys2, recognizing an epitope in exons 77–79; Novocastra Laboratories, Burlingame, CA, USA). Normal and DMD skeletal muscle tissues were simultaneously stained with the antibody. The conditions for immunostaining have been described previously.13
RT-PCR analysis of DMD mRNA
DMD mRNA in skeletal muscle was analyzed as described previously.14, 15 A fragment spanning exons 42 to 47 was amplified by reverse transcription (RT)-PCR using a set of primers (c42f, 5′-GCTCCTGACCTCTGTGCT-3′; and c47r, 5′-GCAGGGGCAACTCTTCCAC-3′) using conditions described before.16 The amplified products were separated by 2% agarose gel electrophoresis.
Quantitative real-time RT-PCR
To quantify exon 44 skipping, real-time PCR amplification was performed using a 7500 Fast Real-time PCR System (Applied Biosystems, Foster City, CA, USA). The sequences of the primers and probes are shown in Table 1. Spectral data were captured and analyzed using 7500 Real-time Analysis Software version 2.0.3 (Applied Biosystems). All samples were run in triplicate. Desmin mRNA was analyzed as an endogenous RNA reference gene for normalization using a TaqMan Gene Expression Assay kit (Life Technologies, Carlsbad, CA, USA).
Genomic DNA analysis
Exon examination
Genomic DNA was extracted from blood of the index case and a control, and exons of the DMD gene were amplified from genomic DNA as described before.17 Exon 44 and its flanking introns were examined by PCR amplification and direct sequencing on an ABI 310 (Applied Biosystems).
Cloning of the junction sequence
Introns 44 and 45 were extended by PCR walking in the patient. The non-repeat sequence regions of both were amplified by PCR, walking step-wise inwards from the 5′ to the 3′ end of intron 44 and from the 3′ to the 5′ end of intron 45, until no amplification product was obtained. After narrowing down the deletion junction, the junction region could be PCR amplified using a forward primer in intron 44 (F8int44, 5′-TAACCACTTGGAGCCCTGTTCTTCC-3′) and a reverse primer in intron 45 (R4int45, 5′-TGGAATCTGTTTCCCCACAGTGTGT-3′). The product was purified and sequenced on an ABI 310 (Applied Biosystems) after subcloning into the vector pT7Blne-T (Novagen, Merck KGaA, Darmstadt, Germany).
Examination of splicing silencer activity
The splicing silencer activity of the junction sequences was examined in vitro by analyzing splicing of the heterologous intron of the Drosophila melanogaster doublesex (dsx) gene pre-mRNA in HeLa cell nuclear extracts as described before.18, 19 To evaluate splicing regulatory activity, various sequences were inserted between the BamH1 and XhoI restriction enzyme recognition sites located downstream of exon 4. In this in vitro system, splicing of chimeric dsx pre-mRNA has been reported to be dependent on the downstream sequence of avian sarcoma leukosis virus (ASLV).18 A construct containing ASLV (dsxASLV; provided by Professor Inoue, Kobe University, Kobe, Japan) was used as a positive control. The 270-bp sequence containing the junction sequence (DJ) amplified using a set of primers (forward primer dsx.565F_BamHI, 5′-GCGGATCCCAGCAAGGAAGAAGAG-3′; reverse primer, dsx.565R_XhoI, 5′-GCCTCGAGAAACACAGGTGGGAAG-3′) was inserted into the predigested dsx plasmid to generate a hybrid minigene (dsxDJ). In addition, the DJ fragment was inserted downstream of the ASLV sequence using two PCR-amplified products obtained by two sets of primers (dsx.aslv_565F_BamHI, 5′-GCGGATCCTCTAGACTCCAAGAAGAAGCCG-3′; dsx.aslv_565R, 5′-CATCTCTTCTTCCTTGCTGTCCTTCTTGCTTGTTGCTG-3′; dsx.aslv_565F, 5′-CAGCAACAAGCAAGAAGGACAGCAAGGAAGAAGAGATG-3′; and dsx.aslv_565R_XhoI, 5′-GCCTCGAGAAACACAGGTGGGAAGAT-3′, restriction enzyme recognition sites underlined). The two amplified products were used for overlapped PCR using primers (dsx.aslv_565F_BamHI and dsx.aslv_565R_XhoI) to incorporate ASLV and DJ. The amplified product was inserted into the predigested dsx plasmid (dsxASLV/DJ). The sequences of the hybrid minigenes were confirmed by DNA sequencing on an ABI 3130 genetic analyzer (Applied Biosystems).
A region of the hybrid minigene was PCR amplified using forward (T7, 5′-TAATACGACTCACTATAGG-3′) and reverse (SP6, 5′-ATTTAGGTGACACTATAG-3′) primers and then purified using a MinElute PCR Purification kit (Qiagen, Hilden, Germany). The purified template DNAs were subjected to in vitro transcription using an mMESSAGE mMACHINE High Yield Capped RNA Transcription kit (Ambion, Austin, TX, USA). The resultant RNAs were purified on a G-50 Quick Spin Column (Roche Applied Science, Indianapolis, IN, USA) and used as a substrate for the splicing reaction.
The in vitro splicing reaction was carried out at 30 °C for 1 h in a volume of 20 μl containing 30 ng pre-mRNA, 50% (v/v) HeLa cell nuclear extract (Lot No. 4168HNE; Computer Cell Culture Center, Mons, Belgium), 1.6 mM MgCl2, 0.5 mM ATP, 20 mM creatine phosphate and 20 U of RNaseOUT (Invitrogen, Carlsbad, CA, USA).20 In some experiments, antisense oligonucleotides (AOs) were added directly to the reaction mixture at 200 nM. The resulting RNA was RT- PCR amplified using a set of primers (DSXex3-F, 5′-GCCAAGACGTTTTCCTAGACT-3′ and DSXex4-R, 5′-CGGGAGTACTCATTCACAACG-3′) to check the intron 3 splicing status. Each amplified product was semiquantified by measuring the peak areas of high-resolution capillary electrophoresis using an Agilent 2100 Bioanalyzer with a DNA1000 LabChip kit (Agilent Technologies, Santa Clara, CA, USA). PCR size determination and quantification were automatically performed using 2100 Expert software (Agilent Technologies). The sequences of all the detected bands were confirmed by subcloning and sequencing as previously described.21
Antisense oligonucleotides
Three phosphorothioate AOs were synthesized using phosphorothioate monomers by Gene Design (Osaka, Japan). AOJ (5′-AACACCAAATAAGTCATGTCCAT-3′), 5′ AOJ (5′-TCAAAAGAATGGCTAAGATGAGATT-3′) and 3′ AOJ (5′-TGCCTAGTATAAAGGCTAATTAGCA-3′) were complementary to the junction site and its flanking 5′ and 3′ sequences, respectively.
Motif analysis
SpliceAid (http://www.introni.it/splicing.html)22 was used for motif analysis of splicing regulatory proteins.
Results
In the index case, no product could be PCR amplified from the exon 45-encompassing region of the DMD gene, whereas neighboring exons 44 and 46 were unaffected (Figure 1). We concluded that his genomic DNA contained a deletion of the single exon 45. The patient’s phenotype was DMD, as expected from the reading frame rule, because the deletion removed the 176 bp of exon 45, producing an out-of-frame mRNA. However, he only became wheelchair bound at the age of 18 years, indicating an outlier from the reading frame rule. It was suggested that the index case could possibly express dystrophin. Consistently, immunohistochemical examination of his skeletal muscle using an antidystrophin monoclonal antibody disclosed weak signals that were patchy and discontinuous (Figure 2a).
PCR amplification of DMD exons. (a) Schematic depiction of the gene structure of DMD exons 44–46. Boxes and bars represent exons and introns, respectively. The shaded area indicates the deletion region. Numbers in and over the boxes indicate nucleotide and exon numbers, respectively. The approximate intron size is described above the line. Brackets indicate the extent of the rearrangement events. Arrows indicate the location and direction of primers used to clone the deletion junction. (b) Genomic PCR products from the exon 44-, 45- and 46-encompassing regions. In the index case (P), expected-size products were obtained from exons 44 and 46, but not from exon 45. All three regions could be amplified from a control (C).
Examination of skeletal muscle. (a) Immunostaining for dystrophin. Immunohistochemical examination of skeletal muscle was done using a monoclonal antibody against the C-terminal region of dystrophin. In the control (C), dystrophin was clearly present along the membranes. In contrast, no signal for dystrophin was observed in a Duchenne muscular dystrophy (DMD) case (DMD). However, staining in the index case (P) revealed weak dystrophin signals with a discontinuous and patchy pattern. (b) Reverse transcription (RT)-PCR amplification of DMD mRNA. RT-PCR products from dystrophin mRNA are shown. A fragment encompassing exons 42 to 47 of DMD mRNA was RT-PCR amplified and analyzed by agarose gel electrophoresis. C, control; P, patient. The exon content of each product is described schematically on the right (Δ45dys, Δ44/45dys). The boxes and number within indicate exons and exon number, respectively. Part of the sequence directly joining exon 43 or 44 to exon 46 in the patient is shown to the right. (c) Quantification of splicing products. Quantification of two dystrophin mRNAs (Δ45dys and Δ44/45dys) is shown. Real-time RT-PCR was performed using probes at the junction of exons 43 and 46 (Δ44/45dys) or exons 44 and 46 (Δ45dys). A full color version of this figure is available at the Journal of Human Genetics journal online.
How was dystrophin synthesized from out-of-frame mRNA in his muscle? We hypothesized that the splicing of DMD pre-mRNA was modified to produce in-frame mRNA. To test this, we amplified DMD mRNA from exons 42 to 47 from muscle RNA by RT-PCR. Remarkably, we obtained two amplified products (Figure 2b). The large and dense band was the expected-size product; sequencing revealed five exons from 42 to 47, lacking 45 (Δ45dys). The smaller, trace-amount product contained an additional deletion of the 148-bp exon 44 sequence (Δ44/45dys), implicating exon 44 skipping. The Δ44/45dys was found to maintain the original dystrophin reading frame, because the 324 bp deletion became in-frame. To examine the level of exon 44 skipping, we quantified Δ45dys and Δ44/45dys in the patient’s muscle by real-time RT–PCR. By designing probes at the exon junctions, either between exons 44 and 46 or exons 43 and 46, we could specifically amplify and quantify both Δ45dys and Δ44/45dys (Figure 2c). The relative quantity of each product was obtained by normalizing with desmin mRNA. The Δ44/45dys accounted for 6% of the total amplified product (Δ45dys+Δ44/45dys). We concluded that partial exon 44 skipping was the mechanism that led to positive dystrophin signals in the patient’s skeletal muscle (Figure 2a).
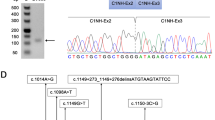
We then investigated why the genomic DNA of the index case underwent partial exon 44 skipping. We first proposed that there was an unidentified nucleotide change around exon 44 that disturbed its splicing. However, sequencing of the genomic region encompassing exon 44 and its flanking introns (187 bp of intron 43 and 92 bp of intron 44) revealed no such nucleotide change (data not shown). Next, we supposed that the created deletion junction acquired a role to repress splicing of the upstream exon 44. To test this, we cloned the deletion junction sequence. Because introns 44 and 45 are so huge (248.4 and 36.1 kb, respectively), we narrowed down the junction site by PCR walking. We sequentially PCR amplified the nonrepeat regions, working inwards toward the deletion junctions until we reached regions that could not be amplified (data not shown). Ultimately, we were able to amplify the region containing the junction using a forward primer in intron 44 (F8 int 44; 175 kb downstream of exon 44) and a reverse primer in intron 45 (R4 int 45; 21 kb upstream of exon 46) (Figures 1a and 3a). Sequencing of this product revealed that the first part (779 bp) completely matched intron 44 sequence, and the last part (1129 bp) completely matched intron 45 sequence, joining intron 44 directly to intron 45 (Figure 3b). However, the exact deletion site was still not determined, because 4 bp at the junction site matched both introns 44 and 45 (Figure 3b). Thus, there were five candidate junction sites. The junction was localized 175.8 kb downstream of exon 44 and 21.8 kb upstream of exon 46 (Figure 1a). As a result, the deletion removed 72.6 kb of the 3′ end of intron 44, 176 bp of exon 45 and 14.3 kb of the 5′ end of intron 45, making the deletion 87 kb long (Figure 1a).
Characterization of the deletion junction. (a) PCR amplification of the deletion junction. PCR amplification product of the deletion junction is shown. The junction fragment was PCR amplified using primers in introns 44 and 45. One clear product was obtained in the patient (P). No product was obtained from the control (C). M indicates the DNA size marker. (b) Junction sequence. Part of the junction sequence is described, showing 33 bp of the 3′ end of intron 44 and 31 bp of the 5′ end of intron 45. Four base pairs, TGAC, occur in both intron sequences. (c) Splicing motif analysis. Result of SpliceAid splicing motif analysis is shown. The names of splicing enhancer and silencer protein-binding sequences are described above and below the sequence, respectively. No known motif was created at the candidate junction sites. A full color version of this figure is available at the Journal of Human Genetics journal online.
To examine the role of the cloned junction sequence in splicing, we searched for splicing regulatory protein motifs using SpliceAid. Although we supposed that the novel junction site would create a splicing silencer protein binding site, no known motifs were created at any candidate junction site (Figure 3c).
Next, we examined the role of the junction sequence in splicing, with an in vitro splicing system using chimeric dsx pre-mRNA (Figure 4a). We tested the 270-bp junction sequence (DJ) containing the junction site in its middle (Figure 4b) for splicing regulatory activity by inserting it downstream of the ASLV enhancer sequence (dsxASLV/DJ), or where ASLV was replaced with the junction sequence (dsxDJ) (Figure 4b). Synthesized pre-mRNA was subjected to splicing by HeLa cell nuclear extract and the resultant mRNA was analyzed by RT–PCR. From dsxASLV we obtained a mature product consisting of exons 3 and 4 (Figure 4c). This indicated that the in vitro splicing of intron 3 of dsxASLV proceeded well. However, dsxDJ did not produce a mature product. This indicated that ASLV was a splicing enhancer whereas junction sequence (DJ) had no effect. Because partial exon 44 skipping was observed in the index case, we supposed that the junction sequence suppresses splicing of intron 3. We examined the junction sequence for splicing regulatory activity by inserting it directly downstream of ASLV (dsxASLV/DJ). Remarkably, no mature product was obtained (Figure 4c). This indicated that the inserted junction sequence inhibited splicing of the upstream intron 3 in this in vitro splicing assay by abolishing splicing enhancer function.
Analysis of the splicing silencer activity of the junction sequence. (a) Schematic representation of Drosophila melanogaster doublesex (dsx) chimeric plasmid. The chimeric plasmid consists of the entire third exon (168 bp), the third intron (114 bp) and 30 bp of the 5′ portion of the fourth exon of the dsx gene plus a linker sequence derived from the pSP72 vector (8 bp). The exons and introns are shown as boxes and lines, respectively. The insertion site for the test sequence is indicated by parallel slanted lines. B and X indicate BamHI and XhoI restriction enzyme recognition sites, respectively. (b) The 270-bp sequence of the junction. The 270 bp sequence of the junction is described. This sequence was inserted into the test sequence site and examined for splicing regulatory activity. The four nucleotides that occur in both introns 44 and 45 are shown in bold. Locations that were targeted by antisense oligonucleotides (AOs) are marked by a thin underline (5′ AOJ), a box (AOJ) or a thick underline (3′ AOJ). Dots indicate bases that match with an intronic splicing silencer in the FGFR2 gene. (c) In vitro splicing products. In vitro splicing products are shown. In vitro-spliced chimeric dsx pre-mRNA was reverse transcription (RT)-PCR amplified using primers from the plasmid; amplified products were analyzed by microcapillary electrophoresis. The structure of each band is depicted schematically on the right. The dsx pre-mRNA harboring ASLV was well spliced to produce the mature mRNA (dsxASLV). However, no mature product was obtained when the junction sequence was replaced with the ASLV splicing enhancer sequence (dsxDJ). Production of mature mRNA from ASLV was abolished by inserting the junction sequence downstream of ASLV (ASLV/DJ). The abolition of mature mRNA production was suppressed by adding AOs to the reaction mixture (ASLV/DJ+AOJ). Other AOs (5′ AOJ and 3′ AOJ) weakly suppressed this abolition (ASLV/DJ+5′ AOJ and ASLV/DJ+3′ AOJ). A full color version of this figure is available at the Journal of Human Genetics journal online.
AOs have been used to localize splicing cis-elements; thus, we employed this strategy to show a role for the junction sequence in in vitro splicing. We designed a 23-bp phosphorothioate oligonucleotide complementary to the junction sequence (AOJ) (Figure 4b) and added it to the in vitro splicing reaction. Upon addition of AOJ, production of mature mRNA from dsxASLV/DJ recovered to the same level as that of dsxASLV. Furthermore, we showed that inhibition of splicing was dependent on the dose of AOJ (data not shown). To determine the sequence specificities, we synthesized two other AOs that were complementary to the 5′ or 3′ part of the junction sequence (5′ AOJ and 3′ AOJ, respectively) (Figure 4b) and added them to the in vitro splicing system of dsxASLV/DJ. Both 5′ AOJ and 3′ AOJ treatment produced a very weak band corresponding to the mature mRNA in addition to a large amount of unspliced pre-mRNA. These results indicated that AOJ exactly complementary to the junction site inhibited the splicing silencer activity of the junction sequence in a sequence-specific manner. This strongly suggested that the junction created by the deletion mutation functions as a splicing silencer, producing partial exon 44 skipping in the index case.
Discussion
Genomic mutation analysis of the index case disclosed deletion of the single exon 45 of the DMD gene. Exon 45 deletion is the most common exon deletion identified in DMD patients, who are generally characterized by dystrophin deficiency. In the index case, in contrast, we observed weak dystrophin staining in his skeletal muscle (Figure 2). This finding did not agree with the reading frame rule, because exon 45 deletion was expected to create a premature stop codon in DMD mRNA. Therefore, we explored the underlying mechanism by analyzing the patient’s skeletal muscle dystrophin mRNA. Remarkably, we identified partial exon 44 skipping, producing Δ44/45dys. Because the Δ44/45dys product was in-frame, it was able to produce internally truncated dystrophin. We considered this to be the mechanism underlying the dystrophin positivity of the patient’s skeletal muscle (Figure 2a).
The Δ44/45dys transcript accounted for 6% of total dystrophin mRNA (Figure 2c). This level of in-frame mRNA in skeletal muscle was probably sufficient to elongate the independent ambulation period to 18 years. This is the first report of conversion of DMD to a milder phenotype by exon 44 skipping.
We searched the literature for other examples of outliers to the reading frame rule with a single exon 45 deletion. In France and the United States, exon 45 deletion has been reported 60 and 13 times, respectively; all were identified in DMD patients, with none in BMD patients.3, 7 In the Leiden database (http://www.dmd.nl/), there are 50 cases with exon 45 deletion in not only DMD/BMD but also intermediate cases.8 This illustrates the clinical heterogeneity of exon 45 deletion. In our Japanese data, 10 cases with exon 45 deletion were classified as either DMD (8 cases) or BMD (2 cases).6 Combining these data, 13 out of 133 cases with a single exon 45 deletion were reported in cases milder than DMD. However, the molecular mechanisms have remained unexplained. Here, we showed that partial exon 44 skipping could modify DMD to a milder phenotype. The variance of phenotypes among the reported patients with an exon 45 deletion could be explained by their levels of exon 44 skipping; mRNA analysis of these patients is necessary. Although the production of Δ44/45dys has been reported in a case with a single exon 45 deletion,23 the level was very low and the authors of the study concluded that the tiny amount of in-frame mRNA (<1% of normal levels) caused the revertant fibers.23 In this case, the level of exon 44 skipping was insufficient to change the phenotype from DMD to a milder form.
Currently, the production of in-frame mRNA by exon skipping with AOs is the primary way to express dystrophin.24, 25 A crucial issue in developing DMD therapy is to determine the minimal level of DMD mRNA needed for alleviation of clinical symptoms. However, the necessary target levels of exon skipping by AO-mediated therapy are still unclear.26, 27 In our case, 6% in-frame dystrophin mRNA probably had a role in prolonging his independent walking period to the age of 18 years. This sheds light on the restoration levels needed in exon skipping therapy. In other words, at least 6% exon skipping should be induced in AO-mediated therapies.
Splicing is strictly regulated to produce proper transcripts at the proper time in the proper tissues. Perhaps unsurprisingly, errors of splicing have been reported in nearly half of genetic diseases,28 mostly mutations affecting conserved splicing regulatory elements. In the DMD gene, 16 alternative splicing transcripts were identified in the region encompassing exons 17–58.29 Although a single skipping of exons 39, 41 and 42 was identified, no single exon 44 skipping event has been described. Exon 44 skipping has been reported in BMD patients with deletion of exons 45–47 or 45–48.30 The phenotype of these patients, however, did not change. Furthermore, no evident explanation of the splicing pattern could be derived by analysis of the breakpoint locations. Instead, it was proposed that CUG-binding protein 2 plays a role in DMD splicing regulation, because a binding site for this protein is located close to the intron–exon boundary.30 In our previous study, we categorized the DMD exons into five groups based on their strength of exon recognition. Exon 44 was categorized as group C, a group comprising the exons that are not so strong.31 On the basis of this classification, it is difficult to explain why exon 44 skipping was induced in the index case.
The deletion junction of the DMD gene has been clarified in more than 50 recombination events.32, 33 In these studies, the mechanisms favoring deletion were discussed but the effect on splicing of the newly created junctions has never been studied. We hypothesized that the deletion junction unique to our index case plays a role in regulating splicing of the upstream exon 44, and tested it using the Drosophila sex determination system—a classic example of how splicing factors exert specific control of splicing.34 This system has been used to determine the splicing regulatory activity of target sequences.19 We showed that the junction sequence suppressed splicing of the upstream intron of dsxASLV (Figure 4). It has been reported that AO could be applied to unravel the role of intronic motifs that function through long-distance interactions35 and, indeed, AOs against the junction site restored the production of mature mRNA from dsxASLV/DJ (Figure 4c). These data indicated that the junction sequence, a novel sequence joining introns 44 and 45, creates a splicing silencer (Figure 4). Several intronic splicing silencers have been characterized: CA-rich sequences bound by hnRNPL,36 specific octamers (CUCGGUGC) flanking exon b of the FGFR2 gene37 and an element in intron 7 of SMN2 (hnRNPA1-binding site).38 Comparing these sequences with our junction sequence, we noticed that part of our junction sequence was similar to an octamer of FGFR2 (UUUGGUGU; underlining indicates match with the octamer). This sequence might act as the splicing silencer, although more studies are needed to clarify this.
Our results strongly suggest that the junction sequence suppresses exon 44 splicing in vivo remotely (175.8 kb away from the 5′ end of intron 44). It is thought that a complex combinatorial interplay of RNA structure and trans-acting factors determines the outcome of splicing.39, 40 Intron length has a major influence on the mode of action of the spliceosome and the choice between competing splice sites.41 Secondary RNA structure is also a factor regulating splicing.42, 43 Our case had a 87.1-kb deletion. We suggest that this changes the secondary RNA structure, positioning the junction site near the splice site. This should be examined in the future.
References
Monaco, A. P., Bertelson, C. J., Liechti-Gallati, S., Moser, H. & Kunkel, L. M. An explanation for the phenotypic differences between patients bearing partial deletions of the DMD locus. Genomics 2, 90–95 (1988).
Beggs, A. H., Koenig, M., Boyce, F. M. & Kunkel, L. M. Detection of 98% of DMD/BMD gene deletions by polymerase chain reaction. Hum. Genet. 86, 45–48 (1990).
Tuffery-Giraud, S., Beroud, C., Leturcq, F., Yaou, R. B., Hamroun, D., Michel-Calemard, L. et al. Genotype-phenotype analysis in 2,405 patients with a dystrophinopathy using the UMD-DMD database: a model of nationwide knowledgebase. Hum. Mutat. 30, 934–945 (2009).
Koenig, M., Beggs, A. H., Moyer, M., Scherpf, S., Heindrich, K., Bettecken, T. et al. The molecular basis for Duchenne versus Becker muscular dystrophy: correlation of severity with type of deletion. Am. J. Hum. Genet. 45, 498–506 (1989).
Kesari, A., Pirra, L., Bremadesam, L., McIntyre, O., Gordon, E., Dubrovsky, A. et al. Integrated DNA, cDNA, and protein studies in Becker muscular dystrophy show high exception to the reading frame rule. Hum. Mutat. 17, 728–737 (2008).
Takeshima, Y., Yagi, M., Okizuka, Y., Awano, H., Zhang, Z., Yamauchi, Y. et al. Mutation spectrum of the dystrophin gene in 442 Duchenne/Becker muscular dystrophy cases from one Japanese referral center. J. Hum. Genet. 55, 379–388 (2010).
Cunniff, C., Andrews, J., Meaney, F. J., Mathews, K. D., Matthews, D., Ciafaloni, E. et al. Mutation analysis in a population-based cohort of boys with Duchenne or Becker muscular dystrophy. J. Child Neurol. 24, 425–430 (2009).
Aartsma-Rus, A., Van Deutekom, J. C., Fokkema, I. F., Van Ommen, G. J. & Den Dunnen, J. T. Entries in the Leiden Duchenne muscular dystrophy mutation database: an overview of mutation types and paradoxical cases that confirm the reading-frame rule. Muscle Nerve 34, 135–144 (2006).
Keren, H., Lev-Maor, G. & Ast, G. Alternative splicing and evolution: diversification, exon definition and function. Nat. Rev. Genet. 11, 345–355 (2010).
Licatalosi, D. D. & Darnell, R. B. RNA processing and its regulation: global insights into biological networks. Nat. Rev. Genet. 11, 75–87 (2010).
Senapathy, P., Shapiro, M. B. & Harris, N. L. Splice junctions, branch point sites, and exons: sequence statistics, identification, and applications to genome project. Methods Enzymol. 183, 252–278 (1990).
Wang, Z. & Burge, C. B. Splicing regulation: from a parts list of regulatory elements to an integrated splicing code. RNA 14, 802–813 (2008).
Adachi, K., Takeshima, Y., Wada, H., Yagi, M., Nakamura, H. & Matsuo, M. Heterogous dystrophin mRNAs produced by a novel splice acceptor site mutation in intermediate dystrophinopathy. Pediatr. Res. 53, 125–131 (2003).
Roberts, R. G., Barby, T. F., Manners, E., Bobrow, M. & Bentley, D. R. Direct detection of dystrophin gene rearrangements by analysis of dystrophin mRNA in peripheral blood lymphocytes. Am. J. Hum. Genet. 49, 298–310 (1991).
Surono, A., Takeshima, Y., Wibawa, T., Ikezawa, M., Nonaka, I. & Matsuo, M. Circular dystrophin RNAs consisting of exons that were skipped by alternative splicing. Hum. Mol. Genet. 8, 493–500 (1999).
Habara, Y., Takeshima, Y., Awano, H., Okizuka, Y., Zhang, Z., Saiki, K. et al. In vitro splicing analysis showed that availability of a cryptic splice site is not a determinant for alternative splicing patterns caused by +1G–>A mutations in introns of the dystrophin gene. J. Med. Genet. 46, 542–547 (2009).
Matsuo, M., Masumura, T., Nakajima, T., Kitoh, Y., Takumi, T., Nishio, H. et al. A very small frame-shifting deletion within exon 19 of the Duchenne muscular dystrophy gene. Biochem. Biophys. Res. Commun. 170, 963–967 (1990).
Tanaka, K., Watakabe, A. & Shimura, Y. Polypurine sequences within a downstream exon function as a splicing enhancer. Mol. Cell. Biol. 14, 1347–1354 (1994).
Shiga, N., Takeshima, Y., Sakamoto, H., Inoue, K., Yokota, Y., Yokoyama, M. et al. Disruption of the splicing enhancer sequence within exon 27 of the dystrophin gene by a nonsense mutation induces partial skipping of the exon and is responsible for Becker muscular dystrophy. J. Clin. Invest. 100, 2204–2210 (1997).
Habara, Y., Doshita, M., Hirozawa, S., Yokono, Y., Yagi, M., Takeshima, Y. et al. A strong exonic splicing enhancer in dystrophin exon 19 achieve proper splicing without an upstream polypyrimidine tract. J. Biochem. 143, 303–310 (2008).
Tran, V. K., Takeshima, Y., Zhang, Z., Yagi, M., Nishiyama, A., Habara, Y. et al. Splicing analysis disclosed a determinant single nucleotide for exon skipping caused by a novel intra-exonic four-nucleotide deletion in the dystrophin gene. J. Med. Genet. 43, 924–930 (2006).
Piva, F., Giulietti, M., Nocchi, L. & Principato, G. SpliceAid: a database of experimental RNA target motifs bound by splicing proteins in humans. Bioinformatics 25, 1211–1213 (2009).
Prior, T. W., Bartolo, C., Papp, A. C., Snyder, P. J., Sedra, M. S., Burghes, A. H. et al. Dystrophin expression in a Duchenne muscular dystrophy patient with a frame shift deletion. Neurology 48, 486–488 (1997).
Cirak, S., Arechavala-Gomeza, V., Guglieri, M., Feng, L., Torelli, S., Anthony, K. et al. Exon skipping and dystrophin restoration in patients with Duchenne muscular dystrophy after systemic phosphorodiamidate morpholino oligomer treatment: an open-label, phase 2, dose-escalation study. Lancet 378, 595–605 (2011).
Goemans, N. M., Tulinius, M., van den Akker, J. T., Burm, B. E., Ekhart, P. F., Heuvelmans, N. et al. Systemic administration of PRO051 in Duchenne’s muscular dystrophy. N. Engl. J. Med. 364, 1513–1522 (2011).
Moulton, H. M. & Moulton, J. D. Morpholinos and their peptide conjugates: therapeutic promise and challenge for Duchenne muscular dystrophy. Biochim. Biophys. Acta 1798, 2296–2303 (2010).
Anthony, K., Cirak, S., Torelli, S., Tasca, G., Feng, L., Arechavala-Gomeza, V. et al. Dystrophin quantification and clinical correlations in Becker muscular dystrophy: implications for clinical trials. Brain 134, 3547–3559 (2011).
Ward, A. J. & Cooper, T. A. The pathobiology of splicing. J. Pathol. 220, 152–163 (2010).
Sironi, M., Cagliani, R., Pozzoli, U., Bardoni, A., Comi, G. P., Giorda, R. et al. The dystrophin gene is alternatively spliced throughout its coding sequence. FEBS Lett. 517, 163–166 (2002).
Sironi, M., Cagliani, R., Comi, G. P., Pozzoli, U., Bardoni, A., Giorda, R. et al. Trans-acting factors may cause dystrophin splicing misregulation in BMD skeletal muscles. FEBS Lett. 537, 30–34 (2003).
Malueka, R. G., Takaoka, Y., Yagi, M., Awano, H., Lee, T., Dwianingsih, E. K. et al. Categorization of 77 dystrophin exons into 5 groups by a decision tree using indexes of splicing regulatory factors as decision markers. BMC Genet 13, 23 (2012).
Toffolatti, L., Cardazzo, B., Nobile, C., Danieli, G. A., Gualandi, F., Muntoni, F. et al. Investigating the mechanism of chromosomal deletion: characterization of 39 deletion breakpoints in introns 47 and 48 of the human dystrophin gene. Genomics 80, 523–530 (2002).
Oshima, J., Magner, D. B., Lee, J. A., Breman, A. M., Schmitt, E.S., White, L.D. et al. Regional genomic instability predisposes to complex dystrophin gene rearrangements. Hum. Genet. 126, 411–423 (2009).
Qi, J., Su, S. & Mattox, W. The doublesex splicing enhancer components Tra2 and Rbp1 also repress splicing through an intronic silencer. Mol. Cell. Biol. 27, 699–708 (2007).
Singh, N. N., Hollinger, K., Bhattacharya, D. & Singh, R. N. An antisense microwalk reveals critical role of an intronic position linked to a unique long-distance interaction in pre-mRNA splicing. RNA 16, 1167–1181 (2010).
Hui, J., Hung, L. H., Heiner, M., Schreiner, S., Neumuller, N., Reither, G. et al. Intronic CA-repeat and CA-rich elements: a new class of regulators of mammalian alternative splicing. EMBO J 24, 1988–1998 (2005).
Wagner, E.J., Baraniak, A. P., Sessions, O. M., Mauger, D., Moskowitz, E. & Garcia-Blanco, M. A. Characterization of the intronic splicing silencers flanking FGFR2 exon IIIb. J. Biol. Chem. 280, 14017–14027 (2005).
Kashima, T., Rao, N. & Manley, J. L. An intronic element contributes to splicing repression in spinal muscular atrophy. Proc. Natl Acad. Sci. USA 104, 3426–3431 (2007).
Hertel, K. Combinatorial control of exon recognition. J. Biol. Chem. 283, 1211–1215 (2008).
Dhir, A., Buratti, E., van Santen, M. A., Luhrmann, R. & Baralle, F. E. The intronic splicing code: multiple factors involved in ATM pseudoexon definition. EMBO J. 29, 749–760 (2010).
Farlow, A., Dolezal, M., Hua, L. & Schlotterer, C. The genomic signature of splicing-coupled selection differs between long and short introns. Mol. Biol. Evol. 29, 21–24 (2012).
Shepard, P. J. & Hertel, K. J. Conserved RNA secondary structures promote alternative splicing. RNA 14, 1463–1469 (2008).
McManus, C. J. & Graveley, B. R. RNA structure and the mechanisms of alternative splicing. Curr. Opin. Genet. Dev. 21, 373–379 (2011).
Acknowledgements
We thank Professor Kunio Inoue, Kobe University, for providing a plasmid. This work was supported by grants from the JSPS KAKENHI, a Health and Labour Sciences Research Grant for Research on Psychiatric and Neurological Diseases and Mental Health and a research grant for Nervous and Mental Disorders from the Ministry of Health, Labour, and Welfare, Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest. The study sponsors played no role in the study design; in the collection, analysis and interpretation of data; in the writing of the report; or in the decision to submit the manuscript for publication.
Additional information
Author contributions
EKD wrote the first draft of the manuscript. RGM and AN participated in analysis of mutation and in vitro splicing analysis. TL, MY, KI and YT participated to collect clinical samples and data. MM supervised the study and edited the manuscript. No honoraria, grants or other forms of payment were given to anyone to produce the manuscript.
Rights and permissions
About this article
Cite this article
Dwianingsih, E., Malueka, R., Nishida, A. et al. A novel splicing silencer generated by DMD exon 45 deletion junction could explain upstream exon 44 skipping that modifies dystrophinopathy. J Hum Genet 59, 423–429 (2014). https://doi.org/10.1038/jhg.2014.36
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jhg.2014.36
This article is cited by
-
Is it time for genetic modifiers to predict prognosis in Duchenne muscular dystrophy?
Nature Reviews Neurology (2023)
-
Genotype characterization and delayed loss of ambulation by glucocorticoids in a large cohort of patients with Duchenne muscular dystrophy
Orphanet Journal of Rare Diseases (2021)
-
Normal and altered pre-mRNA processing in the DMD gene
Human Genetics (2017)
-
A commentary on a novel splicing silencer generated by DMD exon 45 deletion junction could explain upstream exon 44 skipping that modifies dystrophinopathy
Journal of Human Genetics (2014)