Abstract
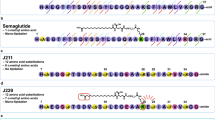
Glucagon-like peptide-1 (GLP-1) is a gastrointestinal hormone secreted from L cells in the distal small intestine and proximal colon after a meal that acts as an incretin to augment the insulin response, while also inhibiting glucagon and slowing gastric emptying. These characteristics of GLP-1, as well as its ability to reduce islet beta cell apoptosis and expand beta cell mass and its cardioprotective and neuroprotective effects, provide a broad spectrum of actions potentially useful for the management of type-2 diabetes mellitus. GLP-1 also has the added advantage of having its incretin effects dependent on the level of serum glucose, only acting in the presence of hyperglycaemia, and thereby preventing hypoglycemic responses. Although natural GLP-1 has a very short half-life, limiting its therapeutic usefulness, a variety of analogues and formulations have been developed to provide extended actions and to limit side effects. However, all of these peptides require parenteral administration. Potentially orally active small-molecule agonists acting at the GLP-1 receptor are also being developed, but have not yet been approved for clinical use. Recent insights into the molecular nature of the class B G-protein-coupled GLP-1 receptor has provided insights into the modes of binding these types of ligands, as well as providing opportunities for rational enhancement. The advantages and disadvantages of each of these agents and their possible clinical utility will be explored.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Hu FB . Globalization of diabetes: the role of diet, lifestyle, and genes. Diabetes Care 2011; 34: 1249–1257.
Creutzfeldt W . The incretin concept today. Diabetologia 1979; 16: 75–85.
Elrick H, Stimmler L, Hlad Jr CJ, Arai Y . Plasma insulin response to oral and intravenous glucose administration. J Clin Endocrinol Metab 1964; 24: 1076–1082.
Nauck M, Stockmann F, Ebert R, Creutzfeldt W . Reduced incretin effect in type 2 (non-insulin-dependent) diabetes. Diabetologia 1986; 29: 46–52.
Adelhorst K, Hedegaard BB, Knudsen LB, Kirk O . Structure-activity studies of glucagon-like peptide-1. J Biol Chem 1994; 269: 6275–6278.
Neidigh JW, Fesinmeyer RM, Prickett KS, Andersen NH . Exendin-4 and glucagon-like-peptide-1: NMR structural comparisons in the solution and micelle-associated states. Biochemistry 2001; 40: 13188–13200.
Hoare SR . Mechanisms of peptide and nonpeptide ligand binding to class B G-protein-coupled receptors. Drug Discov Today 2005; 10: 417–427.
Dong M, Lam PC, Pinon DI, Hosohata K, Orry A, Sexton PM et al. Molecular basis of secretin docking to its intact receptor using multiple photolabile probes distributed throughout the pharmacophore. J Biol Chem 2011; 286: 23888–23899.
White JW, Saunders GF . Structure of the human glucagon gene. Nucleic Acids Res 1986; 14: 4719–4730.
Mortensen K, Christensen LL, Holst JJ, Orskov C . GLP-1 and GIP are colocalized in a subset of endocrine cells in the small intestine. Regul Pept 2003; 114: 189–196.
Mayo KE, Miller LJ, Bataille D, Dalle S, Goke B, Thorens B et al. International union of pharmacology. XXXV. The glucagon receptor family. Pharmacol Rev 2003; 55: 167–194.
Shoichet BK, Kobilka BK . Structure-based drug screening for G-protein-coupled receptors. Trends Pharmacol Sci 2012; 33: 268–272.
Parthier C, Reedtz-Runge S, Rudolph R, Stubbs MT . Passing the baton in class B GPCRs: peptide hormone activation via helix induction? Trends Biochem Sci 2009; 34: 303–310.
Lisenbee CS, Dong M, Miller LJ . Paired cysteine mutagenesis to establish the pattern of disulfide bonds in the functional intact secretin receptor. J Biol Chem 2005; 280: 12330–12338.
Miras AD, Jackson RN, Jackson SN, Goldstone AP, Olbers T, Hackenberg T et al. Gastric bypass surgery for obesity decreases the reward value of a sweet-fat stimulus as assessed in a progressive ratio task. Am J Clin Nutr 2012; 96: 467–473.
Eng J, Kleinman WA, Singh L, Singh G, Raufman JP . Isolation and characterization of exendin-4, an exendin-3 analogue, from Heloderma suspectum venom. Further evidence for an exendin receptor on dispersed acini from guinea pig pancreas. J Biol Chem 1992; 267: 7402–7405.
Donnelly D . The structure and function of the glucagon-like peptide-1 receptor and its ligands. Br J Pharmacol 2012; 166: 27–41.
Kendall DM, Riddle MC, Rosenstock J, Zhuang D, Kim DD, Fineman MS et al. Effects of exenatide (exendin-4) on glycemic control over 30 weeks in patients with type 2 diabetes treated with metformin and a sulfonylurea. Diabetes Care 2005; 28: 1083–1091.
Buse JB, Klonoff DC, Nielsen LL, Guan X, Bowlus CL, Holcombe JH et al. Metabolic effects of two years of exenatide treatment on diabetes, obesity, and hepatic biomarkers in patients with type 2 diabetes: an interim analysis of data from the open-label, uncontrolled extension of three double-blind, placebo-controlled trials. Clin Ther 2007; 29: 139–153.
Christensen M, Knop FK, Holst JJ, Vilsboll T . Lixisenatide, a novel GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus. IDrugs 2009; 12: 503–513.
Knudsen LB . Liraglutide: the therapeutic promise from animal models. Int J Clin Pract Suppl 2010; 64: 4–11.
Nauck MA, Meier JJ . Glucagon-like peptide 1 and its derivatives in the treatment of diabetes. Regul Pept 2005; 128: 135–148.
Meier JJ . GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus. Nat Rev Endocrinol 2012; 8: 728–742.
Raz I, Hanefeld M, Xu L, Caria C, Williams-Herman D, Khatami H . Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy in patients with type 2 diabetes mellitus. Diabetologia 2006; 49: 2564–2571.
Baggio LL, Huang Q, Brown TJ, Drucker DJ . Oxyntomodulin and glucagon-like peptide-1 differentially regulate murine food intake and energy expenditure. Gastroenterology 2004; 127: 546–558.
Chen D, Liao J, Li N, Zhou C, Liu Q, Wang G et al. A nonpeptidic agonist of glucagon-like peptide 1 receptors with efficacy in diabetic db/db mice. Proc Natl Acad Sci USA 2007; 104: 943–948.
Irwin N, Flatt PR, Patterson S, Green BD . Insulin-releasing and metabolic effects of small molecule GLP-1 receptor agonist 6,7-dichloro-2-methylsulfonyl-3-N-tert-butylaminoquinoxaline. Eur J Pharmacol 2010; 628: 268–273.
Knudsen LB, Kiel D, Teng M, Behrens C, Bhumralkar D, Kodra JT et al. Small-molecule agonists for the glucagon-like peptide 1 receptor. Proc Natl Acad Sci USA 2007; 104: 937–942.
Sloop KW, Willard FS, Brenner MB, Ficorilli J, Valasek K, Showalter AD et al. Novel small molecule glucagon-like peptide-1 receptor agonist stimulates insulin secretion in rodents and from human islets. Diabetes 2010; 59: 3099–3107.
Su H, He M, Li H, Liu Q, Wang J, Wang Y et al. Boc5, a non-peptidic glucagon-like Peptide-1 receptor agonist, invokes sustained glycemic control and weight loss in diabetic mice. PLoS One 2008; 3: e2892.
Willard FS, Bueno AB, Sloop KW . Small molecule drug discovery at the glucagon-like peptide-1 receptor. Exp Diabetes Res 2012; 2012: 709893.
Koole C, Wootten D, Simms J, Miller LJ, Christopoulos A, Sexton PM . Second extracellular loop of human glucagon-like peptide-1 receptor (GLP-1R) has a critical role in GLP-1 peptide binding and receptor activation. J Biol Chem 2012; 287: 3642–3658.
Koole C, Wootten D, Simms J, Savage EE, Miller LJ, Christopoulos A et al. Second extracellular loop of human glucagon-like peptide-1 receptor (GLP-1R) differentially regulates orthosteric but not allosteric agonist binding and function. J Biol Chem 2012; 287: 3659–3673.
Koole C, Wootten D, Simms J, Valant C, Sridhar R, Woodman OL et al. Allosteric ligands of the glucagon-like peptide 1 receptor (GLP-1R) differentially modulate endogenous and exogenous peptide responses in a pathway-selective manner: implications for drug screening. Mol Pharmacol 2010; 78: 456–465.
Harikumar KG, Wootten D, Pinon DI, Koole C, Ball AM, Furness SGB et al. Glucagon-like peptide-1 receptor dimerization differentially regulates agonist signalling but does not affect small molecule allostery. Proc Natl Acad Sci USA 2012; 109: 18607–18612.
Gao F, Harikumar KG, Dong M, Lam PC, Sexton PM, Christopoulos A et al. Functional importance of a structurally distinct homodimeric complex of the family B G protein- coupled secretin receptor. Mol Pharmacol 2009; 76: 264–274.
Acknowledgements
This work was supported by a grant from the National Institutes of Health (DK046577). PS has received an NHMRC Program grant (#519461) and an NHMRC Project grant (#1002180).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This article is published as part of a supplement sponsored by the Université Laval's Research Chair in Obesity in an effort to inform the public on the causes, consequences, treatments, and prevention of obesity.
Rights and permissions
About this article
Cite this article
Miller, L., Sexton, P., Dong, M. et al. The class B G-protein-coupled GLP-1 receptor: an important target for the treatment of type-2 diabetes mellitus. Int J Obes Supp 4 (Suppl 1), S9–S13 (2014). https://doi.org/10.1038/ijosup.2014.4
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijosup.2014.4