Abstract
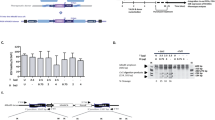
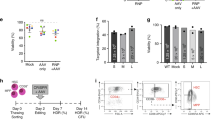
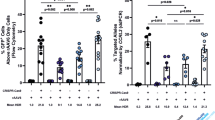
The correction of genetic mutations by homologous recombination is an attractive approach to gene therapy. We used the DNA double-strand breaks introduced by the site-specific endonuclease I-Sce1 as a means of increasing homologous recombination of an exogenous DNA template in murine hematopoietic stem cells (mHSCs). To develop this approach, we chose an Artemis knockout (Art−/−) mouse in which exon 12 of the Artemis gene had been replaced by an I-Sce1 recognition site. The I-Sce1 enzyme and the Artemis correction template were each delivered by a self-inactivating (SIN)-integrase-defective lentiviral vector (SIN-IDLV-CMV-ISce1 and SIN-IDLV-Art, respectively). Transduction of Art−/− mHSCs with the two vectors successfully reverted the Art−/− phenotype in 2 of our 10 experiments. Even though the potential for genotoxicity has yet to be evaluated, this new approach to gene editing appears to be promising. Improving the efficacy of this approach will require further technical work.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Fischer A, Hacein-Bey-Abina S, Cavazzana-Calvo M . 20 years of gene therapy for SCID. Nat Immunol 2010; 11: 457–460.
Porteus M . Homologous recombination-based gene therapy for the primary immunodeficiencies. Ann N Y Acad Sci 2011; 1246: 131–140.
Cornu TI, Cathomen T . Targeted genome modifications using integrase-deficient lentiviral vectors. Mol Ther J Am Soc Gene Ther 2007; 15: 2107–2113.
Tichy ED, Pillai R, Deng L, Liang L, Tischfield J, Schwemberger SJ et al. Mouse embryonic stem cells, but not somatic cells, predominantly use homologous recombination to repair double-strand DNA breaks. Stem Cells Dev 2010; 19: 1699–1711.
Choulika A, Perrin A, Dujon B, Nicolas JF . Induction of homologous recombination in mammalian chromosomes by using the I-SceI system of Saccharomyces cerevisiae. Mol Cell Biol 1995; 15: 1968–1973.
Benjelloun F, Garrigue A, Demerens-de Chappedelaine C, Soulas-Sprauel P, Malassis-Séris M, Stockholm D et al. Stable and functional lymphoid reconstitution in artemis-deficient mice following lentiviral artemis gene transfer into hematopoietic stem cells. Mol Ther J Am Soc Gene Ther 2008; 16: 1490–1499.
Moshous DCI, Moshous D, Callebaut I, de Chasseval R, Corneo B, Cavazzana-Calvo M et al. Artemis, a novel DNA double-strand break repair/V(D)J recombination protein, is mutated in human severe combined immune deficiency. Cell 2001; 105: 177–186.
Stephan V, Wahn V, Le Deist F, Dirksen U, Broker B, Müller-Fleckenstein I et al. Atypical X-linked severe combined immunodeficiency due to possible spontaneous reversion of the genetic defect in T cells. N Engl J Med 1996; 335: 1563–1567.
Urnov FD, Rebar EJ, Holmes MC, Zhang HS, Gregory PD . Genome editing with engineered zinc finger nucleases. Nat Rev Genet 2010; 11: 636–646.
Urnov FD, Miller JC, Lee Y-L, Beausejour CM, Rock JM, Augustus S et al. Highly efficient endogenous human gene correction using designed zinc-finger nucleases. Nature 2005; 435: 646–651.
Lombardo A, Genovese P, Beausejour CM, Colleoni S, Lee Y-L, Kim KA et al. Gene editing in human stem cells using zinc finger nucleasesand integrase-defective lentiviral vector delivery. Nat Biotechnol 2007; 25: 1298–1306.
Pattanayak V, Ramirez CL, Joung JK, Liu DR . Revealing off-target cleavage specificities of zinc-finger nucleases by in vitro selection. Nat Methods 2011; 8: 765–770.
Cornu TI, Cathomen T . Quantification of zinc finger nuclease-associated toxicity. Methods Mol Biol Clifton NJ 2010; 649: 237–245.
Santoni de Sio FR, Cascio P, Zingale A, Gasparini M, Naldini L . Proteasome activity restricts lentiviral gene transfer into hematopoietic stem cells and is down-regulated by cytokines that enhance transduction. Blood 2006; 107: 4257–4265.
Paulk NK, Loza LM, Finegold MJ, Grompe M . AAV-mediated gene targeting is significantly enhanced by transient inhibition of nonhomologous end joining or the proteasome in v ivo. Hum Gene Ther 2012; 23: 658–665.
Gaj T, Gersbach CA, Barbas CF 3rd . ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol 2013; 31: 397–405.
Rivera-Munoz P, Soulas-Sprauel P, Le Guyader G, Abramowski V, Bruneau S, Fischer A et al. Reduced immunoglobulin class switch recombination in the absence of Artemis. Blood 2009; 114: 3601–3609.
Yáñez-Muñoz RJ, Balaggan KS, MacNeil A, Howe SJ, Schmidt M, Smith AJ et al. Effective gene therapy with nonintegrating lentiviral vectors. Nat Med 2006; 12: 348–353.
Zufferey R, Nagy D, Mandel RJ, Naldini L, Trono D . Multiply attenuated lentiviral vector achieves efficient gene delivery in vivo. Nat Biotechnol 1997; 15: 871–875.
Yee JK, Miyanohara A, LaPorte P, Bouic K, Burns JC, Friedmann T . A general method for the generation of high-titer, pantropic retroviral vectors: highly efficient infection of primary hepatocytes. Proc Natl Acad Sci USA 1994; 91: 9564–9568.
Iglesias MC, Frenkiel M-P, Mollier K, Souque P, Despres P, Charneau P . A single immunization with a minute dose of a lentiviral vector-based vaccine is highly effective at eliciting protective humoral immunity against West Nile virus. J Gene Med 2006; 8: 265–274.
Six EM, Benjelloun F, Garrigue A, Bonhomme D, Morillon E, Rouiller J et al. Cytokines and culture medium have a major impact on human in vitro T-cell differentiation. Blood Cells Mol Dis 2011; 47: 72–78.
Acknowledgements
This work was part of the INGENIS project, which is funded by the French government (via the Direction Générale de la Compétitivité, de l’Industrie et des Services) and the Medicen cluster.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Rivière, J., Hauer, J., Poirot, L. et al. Variable correction of Artemis deficiency by I-Sce1-meganuclease-assisted homologous recombination in murine hematopoietic stem cells. Gene Ther 21, 529–532 (2014). https://doi.org/10.1038/gt.2014.20
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2014.20
This article is cited by
-
TALEN mediated gene editing in a mouse model of Fanconi anemia
Scientific Reports (2020)
-
Genome-editing Technologies for Gene and Cell Therapy
Molecular Therapy (2016)