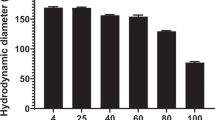
Abstract
Polyethylene glycol (PEG)ylated polyacridine peptides bind to plasmid DNA with high affinity to form unique polyplexes that possess a long circulatory half-life and are hydrodynamically (HD)-stimulated to produce efficient gene expression in the liver of mice. We previously demonstrated that acridine-modified lysine (Acr) in (Acr-Lys)6-Cys-PEG5kDa stabilizes a 1-μg pGL3 dose for up to 1 h in the circulation, resulting in HD-stimulated (saline only) gene expression in the liver, equivalent in magnitude to direct-HD dosing of 1 μg of pGL3. In this study, we report that increasing the spacing of Acr with either four or five Lys residues markedly increases the stability of PEGylated polyacridine peptide polyplexes in the circulation allowing maximal HD-stimulated expression for up to 5 h post DNA administration. Co-administration of a decoy dose of 9 μg of non-expressing DNA polyplex with 1 μg of pGL3 polyplex further extended the HD-stimulated expression to 9 h. This structure–activity relationship study defines the PEGylated polyacridine peptide requirements for maintaining fully transfection competent plasmid DNA in the circulation for 5 h and provides an understanding as to why polyplexes or lipoplexes prepared with polyethylenimine, chitosan or Lipofectamine are inactive within 5 min following intravenous dosing.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Fernandez CA, Rice KG . Engineered nanoscaled polyplex gene delivery systems. Mol Pharm 2009; 6: 1277–1289.
Merdan T, Kunath K, Petersen H, Bakowsky U, Voigt KH, Kopecek J et al. PEGylation of poly(ethylene imine) affects stability of complexes with plasmid DNA under in vivo conditions in a dose-dependent manner after intravenous injection into mice. Bioconj Chem 2005; 16: 785–792.
Cohen RN, van der Aa MA, Macaraeg N, Lee AP, Szoka FC . Quantification of plasmid DNA copies in the nucleus after lipoplex and polyplex transfection. J Cont Rel 2009; 135: 166–174.
Burke RS, Pun SH . Extracellular barriers to in vivo PEI and PEGylated PEI polyplex-mediated gene delivery to the liver. Bioconj Chem 2008; 19: 693–704.
Grigsby C, Leong K . Balancing protection and release of DNA: tools to address a bottleneck of non-viral gene delivery. J Royal Soc Interface 2010; 7: S67–S82.
Van der Aa MA, Koning GA, d’Oliveira C, Oosting RS, Wilschut KJ, Hennink WE et al. An NLS peptide covalently linked to linear DNA does not enhance transfection efficiency of cationic polymer based gene delivery systems. J Gene Med 2005; 7: 208–217.
Tanimoto M, Kamiya H, Minakawa N, Matsuda A, Harashima H . No enhancement of nuclear entry by direct conjugation of a nuclear localization signal peptide to linearized DNA. Bioconj Chem 2003; 14: 1197–1202.
Fernandez CA, Baumhover NJ, Duskey JT, Khargharia S, Kizzire K, Ericson MD et al. Metabolically stabilized long-circulating PEGylated polyacridine peptide polyplexes mediate hydrodynamically stimulated gene expression in liver. Gene Therapy 2011; 18: 23–37.
Chen CP, Kim JS, Liu D, Rettig GR, McAnuff MA, Martin ME et al. Synthetic PEGylated glycoproteins and their utility in gene delivery. Bioconj Chem 2007; 18: 371–378.
Boussif O, Lezoualc’h F, Zanta MA, Mergny MD, Scherman D, Demeneix B et al. A versatile vector for gene and oligonucleotide transfer into cells in culture and in vivo: polyethylenimine. PNAS USA 1995; 92: 7297–7301.
Felgner PL, Gadek TR, Holm M, Roman R, Chan HW, Wenz M et al. Lipofection: a highly efficient, lipid-mediated DNA-transfection procedure. PNAS USA 1987; 84: 7413–7417.
Liu F, Song Y, Liu D . Hydrodynamics-based transfection in animals by systemic administration of plasmid DNA. Gene Therapy 1999; 6: 1258–1266.
Rettig GR, Rice KG . Quantitative in vivo imaging of non-viral-mediated gene expression and rnai-mediated knockdown. Bioluminescence. In: Rich P, Douillet C, (eds). Methods in Molecular Biology, vol. 574. Humana Press: New York, 2009, pp 155–171.
Rettig GR, McAnuff M, Liu D, Kim JS, Rice KG . Quantitative bioluminscence imaging of transgene expression in vivo. Anal Biochem 2006; 335: 90–94.
Collard WT, Collard WT, Yang Y, Kwok KY, Park Y, Rice KG et al. Biodistribution, metabolism, and in vivo gene expression of low molecular weight glycopeptide polyethylene glycol peptide DNA co-condensates. J Pharm Sci 2000; 89: 499–512.
Tung C, Zhu T, Lackland H, Stein S . An acridine amino acid derivative for use in Fmoc peptide synthesis. Peptide Res 1992; 5: 115–118.
Ueyama H, Takagi M, Waki M, Takenaka S . DNA binding behavior of peptides carrying acridinyl units: First example of effective poly-intercalation. Nucleic Acids Symp Ser (Oxf) 2001; 1: 163–164.
Kwok KY, McKenzie DL, Evers DL, Rice KG . Formulation of highly soluble poly(ethylene glycol)-peptide DNA condensates. J Pharm Sci 1999; 88: 996–1003.
Adami RC, Rice KG . Metabolic stability of glutaraldehyde cross-linked peptide DNA condensates. J Pharm Sci 1999; 88: 739–746.
Mullen PM, Lollo CP, Phan QC, Amini A, Banaszczyk MG, Fabrycki JM et al. Strength of conjugate binding to plasmid DNA affects degradation rate and expression level in vivo. Biochim Biophys Acta 2000; 1523: 103–110.
Adami RC, Collard WT, Gupta SA, Kwok KY, Bonadio J, Rice KG et al. Stability of peptide-condensed plasmid dna formulations. J Pharm Sci 1998; 87: 678–683.
Ogris M, Brunner S, Schüller S, Kircheis R, Wagner E . PEGylated DNA/transferrin-PEI complexes: reduced interaction with blood components, extended circulation in blood and potential for systemic gene delivery. Gene Ther 1999; 6: 595–605.
Heyes J, Palmer L, Chan K, Giesbrecht C, Jeffs L, MacLachlan I . Lipid encapsulation enables the effective systemic delivery of polyplex plasmid DNA. Mol Ther 2007; 15: 713–720.
Fortune JA, Novobrantseva TI, Klibanov AM . Highly effective gene transfection in vivo by alkylated polyethylenimine. J Drug Deliv 2011; 2011: 204058.
Kim KS, Lei Y, Stolz DB, Liu D . Bifunctional compounds for targeted hepatic gene delivery. Gene Therapy 2007; 14: 704–708.
Harris T, Green JJ, Fung PW, Langer R, Anderson DG, Bhatia SN et al. Tissue-specific gene delviery via nanoparticle coating. Biomaterials 2010; 31: 998–1006.
Baumhover NJ, Anderson K, Fernandez CA, Rice KG . Synthesis and in vitro testing of new potent polyacridine-melittin gene delivery peptides. Bioconj Chem 2010; 21: 74–86.
Kawabata K, Takakura Y, Hashida M . The fate of plasmid DNA after intravenous injection in mice: involvement of scavenger receptors in its hepatic uptake. Pharm Res 1995; 12: 825–830.
Fernandez CA, Baumhover NJ, Anderson K, Rice KG . Discovery of metabolically stablized electronegative polyacridine-PEG peptide DNA open polyplexes. Bioconj Chem 2010; 21: 723–730.
Kunath K, von Harpe A, Petersen H, Fischer D, Voigt K, Kissel T et al. The Structure of peg-modified poly(ethylene imines) influences biodistribution and pharmacokinetics of their complexes with NF-κB decoy in Mice. Pharm Res 2002; 19: 810–817.
Li S-D, Chen Y-C, Hackett MJ, Huang L . Tumor-targeted delivery of siRNA by self-assembled nanoparticles. Mol Ther 2007; 16: 163–169.
Jaing X, Dai H, Leong KW, Goh SH, Mao HQ, Yang YY et al. Chitosan-g-PEG/DNA complexes deliver gene to the rat liver via intrabiliary and intraportal infusions. J Gene Med 2006; 8: 477–487.
Morille M, Montier T, Legras P, Carmoy N, Brodin P, Pitard B et al. Long-circulating DNA lipid nanocapsules as new vector for passive tumor targeting. Biomaterials 2010; 31: 321–329.
Mahato RI, Kawabata K, Nomura T, Takakura Y, Hashida M . Physicochemical and pharmacokinetic characteristics of plasmid DNA/cationic liposome complexes. J Pharm Sci 1995; 84: 1267–1271.
Mahato RI, Kawabata K, Takakura Y, Hashida M . In vivo disposition characteristics of plasmid DNA complexed with cationic liposomes. J Drug Target 1995; 3: 149–157.
Anderson K, Fernandez CA, Rice KG . N-glycan targeted gene delivery to the dendritic cell SIGN receptor. Bioconj Chem 2010; 21: 1479–1485.
Wadhwa MS, Collard WT, Adami RC, McKenzie DL, Rice KG . Peptide-mediated gene delivery: influence of peptide structure on gene expression. Bioconj Chem 1997; 8: 81–88.
Terebesi J, Kwok KY, Rice KG . Iodinated plasmid DNA as a tool for studying gene delivery. Anal Biochem 1998; 263: 120–123.
Zhang G, Budker V, Wolff JA . High levels of foreign gene expression in hepatocytes after tail vein injections of naked plasmid DNA. Hum Gene Ther 1999; 10: 1735–1737.
Acknowledgements
We acknowledge support from the NIH grant GM097093 and (KK) NIH T32GM067795.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kizzire, K., Khargharia, S. & Rice, K. High-affinity PEGylated polyacridine peptide polyplexes mediate potent in vivo gene expression. Gene Ther 20, 407–416 (2013). https://doi.org/10.1038/gt.2012.47
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2012.47
Keywords
This article is cited by
-
Heat-shrinking DNA nanoparticles for in vivo gene delivery
Gene Therapy (2020)
-
Design and development of a robust photo-responsive block copolymer framework for tunable nucleic acid delivery and efficient gene silencing
Polymer Journal (2018)
-
Efficient expression of stabilized mRNA PEG-peptide polyplexes in liver
Gene Therapy (2015)
-
The uptake mechanism of PEGylated DNA polyplexes by the liver influences gene expression
Gene Therapy (2014)