Abstract
Background/Objectives:
To determine the impact of a not hydrolyzed fermented infant formula containing heat-killed Bifidobacterium breve C50 and Streptococcus thermophilus 065 (HKBBST) on the incidence of allergy-like events during the first 2 years of life in children at high risk of atopy.
Subjects/Methods:
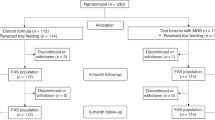
This multicenter, randomized, double-blind, controlled study included infants at high risk of atopy. Infants used HKBBST or a standard infant formula (SIF) since birth until 1 year of age, and were followed at 4, 12 and 24 months after birth. Skin prick tests (SPTs) for six foods and six aeroallergens were systematically performed and adverse events (AEs) were recorded. In case of potentially allergic AE (PAAE), allergy could be further tested by SPT, patch tests and quantification of specific IgEs. If cow's milk allergy (CMA) was suspected, an oral challenge could also be performed.
Results:
The study included 129 children, 63 were randomized to SIF, 66 to HKBBST. The use of HKBBST milk did not alter the proportion of CMA but decreased the proportion of positive SPT to cow's milk (1.7 vs 12.5%, P=0.03), and the incidence of digestive (39 vs 63%, P=0.01) and respiratory potentially allergic AEs (7 vs 21%, P=0.03) at 12 months, and that of respiratory PAAEs at 24 months (13 vs 35%, P=0.01).
Conclusions:
HKBBST decreased the incidence of PAAEs in children with family history of atopy, during the first months of life and after the formula was stopped. Oral tolerance to cow's milk in infants at high risk of atopy may therefore be improved using not hydrolyzed fermented formulae.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
AFSSA (2003). Report of the working group on ‘Infant foods and modification of the intestinal flora’.
Agostoni C, Axelsson I, Goulet O, Koletzko B, Michaelsen KF, Puntis JW et al. (2004). Prebiotic oligosaccharides in dietetic products for infants: a commentary by the ESPGHAN Committee on Nutrition. J Pediatr Gastroenterol Nutr 39, 465–473.
Agostoni C, Goulet O, Kolacek S, Koletzko B, Moreno L, Puntis J et al. (2007). Fermented infant formulae without live bacteria. J Pediatr Gastroenterol Nutr 44, 392–397.
Betsi GI, Papadavid E, Falagas ME (2008). Probiotics for the treatment or prevention of atopic dermatitis: a review of the evidence from randomized controlled trials. Am J Clin Dermatol 9, 93–103.
Boehm G, Moro G (2008). Structural and functional aspects of prebiotics used in infant nutrition. J Nutr 138, 1818S–1828S.
Dreborg S (1991). Skin test in diagnosis of food allergy. Allergy Proc 12, 251–254.
Exl BM, Fritsche R (2001). Cow's milk protein allergy and possible means for its prevention. Nutrition 17, 642–651.
FAO/WHO (2002). Report of a Joint FAO/WHO Working Group on Drafting Guidelines for the Evaluation of Probiotics in Food. London, Ontario, Canada.
Garcia-Ara C, Boyano-Martinez T, Diaz-Pena JM, Martin-Munoz F, Reche-Frutos M, Martin-Esteban M (2001). Specific IgE levels in the diagnosis of immediate hypersensitivity to cows’ milk protein in the infant. J Allergy Clin Immunol 107, 185–190.
Giovannini M, Agostoni C, Riva E, Salvini F, Ruscitto A, Zuccotti GV et al. (2007). A randomized prospective double blind controlled trial on effects of long-term consumption of fermented milk containing Lactobacillus casei in pre-school children with allergic asthma and/or rhinitis. Pediatr Res 62, 215–220.
Greer FR, Sicherer SH, Burks AW (2008). Effects of early nutritional interventions on the development of atopic disease in infants and children: the role of maternal dietary restriction, breastfeeding, timing of introduction of complementary foods, and hydrolyzed formulas. Pediatrics 121, 183–191.
Hoarau C, Lagaraine C, Martin L, Velge-Roussel F, Lebranchu Y (2006). Supernatant of Bifidobacterium breve induces dendritic cell maturation, activation, and survival through a Toll-like receptor 2 pathway. J Allergy Clin Immunol 117, 696–702.
Hol J, van Leer EH, Elink Schuurman BE, de Ruiter LF, Samsom JN, Hop W et al. (2008). The acquisition of tolerance toward cow's milk through probiotic supplementation: a randomized, controlled trial. J Allergy Clin Immunol 121, 1448–1454.
Host A, Halken S, Muraro A, Dreborg S, Niggemann B, Aalberse R et al. (2008). Dietary prevention of allergic diseases in infants and small children. Pediatr Allergy Immunol 19, 1–4.
Indrio F, Ladisa G, Mautone A, Montagna O (2007). Effect of a fermented formula on thymus size and stool pH in healthy term infants. Pediatr Res 62, 98–100.
Isolauri E, Arvola T, Sutas Y, Moilanen E, Salminen S (2000). Probiotics in the management of atopic eczema. Clin Exp Allergy 30, 1604–1610.
Kalliomaki M, Salminen S, Arvilommi H, Kero P, Koskinen P, Isolauri E (2001). Probiotics in primary prevention of atopic disease: a randomised placebo-controlled trial. Lancet 357, 1076–1079.
Kekki OM, Turjanmaa K, Isolauri E (1997). Differences in skin-prick and patch-test reactivity are related to the heterogeneity of atopic eczema in infants. Allergy 52, 755–759.
Kopp MV, Hennemuth I, Heinzmann A, Urbanek R (2008). Randomized, double-blind, placebo-controlled trial of probiotics for primary prevention: no clinical effects of Lactobacillus GG supplementation. Pediatrics 121, e850–e856.
Kukkonen K, Savilahti E, Haahtela T, Juntunen-Backman K, Korpela R, Poussa T et al. (2007). Probiotics and prebiotic galacto-oligosaccharides in the prevention of allergic diseases: a randomized, double-blind, placebo-controlled trial. J Allergy Clin Immunol 119, 192–198.
Kvenshagen B, Halvorsen R, Jacobsen M (2008). Adverse reactions to milk in infants. Acta Paediatr 97, 196–200.
Lee J, Seto D, Bielory L (2008). Meta-analysis of clinical trials of probiotics for prevention and treatment of pediatric atopic dermatitis. J Allergy Clin Immunol 121, 116–121. e111.
Menard S, Candalh C, Bambou JC, Terpend K, Cerf-Bensussan N, Heyman M (2004). Lactic acid bacteria secrete metabolites retaining anti-inflammatory properties after intestinal transport. Gut 53, 821–828.
Menard S, Candalh C, Ben Ahmed M, Rakotobe S, Gaboriau-Routhiau V, Cerf-Bensussan N et al. (2006). Stimulation of immunity without alteration of oral tolerance in mice fed with heat-treated fermented infant formula. J Pediatr Gastroenterol Nutr 43, 451–458.
Osborn DA, Sinn J (2006). Formulas containing hydrolysed protein for prevention of allergy and food intolerance in infants. Cochrane Database Syst Rev, CD003664.
Osborn DA, Sinn JK (2007). Probiotics in infants for prevention of allergic disease and food hypersensitivity. Cochrane Database Syst Rev, CD006475.
Romond MB, Ais A, Guillemot F, Bounouader R, Cortot A, Romond C (1998). Cell-free whey from milk fermented with Bifidobacterium breve C50 used to modify the colonic microflora of healthy subjects. J Dairy Sci 81, 1229–1235.
Romond MB, Ais A, Yazourh A, Romond C (1997). Cell-free wheys from bifidobacteria fermented milks exert a regulatory effect on the intestinal microflora of mice and humans. Anaerobe 3, 137–143.
Rosenfeldt V, Benfeldt E, Nielsen SD, Michaelsen KF, Jeppesen DL, Valerius NH et al. (2003). Effect of probiotic Lactobacillus strains in children with atopic dermatitis. J Allergy Clin Immunol 111, 389–395.
Saarinen KM, Juntunen-Backman K, Jarvenpaa AL, Kuitunen P, Lope L, Renlund M et al. (1999). Supplementary feeding in maternity hospitals and the risk of cow's milk allergy: a prospective study of 6209 infants. J Allergy Clin Immunol 104, 457–461.
Sashihara T, Sueki N, Ikegami S (2006). An analysis of the effectiveness of heat-killed lactic acid bacteria in alleviating allergic diseases. J Dairy Sci 89, 2846–2855.
Thibault H, Aubert-Jacquin C, Goulet O (2004). Effects of long-term consumption of a fermented infant formula (with Bifidobacterium breve c50 and Streptococcus thermophilus 065) on acute diarrhea in healthy infants. J Pediatr Gastroenterol Nutr 39, 147–152.
Turjanmaa K, Darsow U, Niggemann B, Rance F, Vanto T, Werfel T (2006). EAACI/GA2LEN position paper: present status of the atopy patch test. Allergy 61, 1377–1384.
Vandenplas Y, Koletzko S, Isolauri E, Hill D, Oranje AP, Brueton M et al. (2007). Guidelines for the diagnosis and management of cow's milk protein allergy in infants. Arch Dis Child 92, 902–908.
Viljanen M, Savilahti E, Haahtela T, Juntunen-Backman K, Korpela R, Poussa T et al. (2005). Probiotics in the treatment of atopic eczema/dermatitis syndrome in infants: a double-blind placebo-controlled trial. Allergy 60, 494–500.
von Berg A, Koletzko S, Filipiak-Pittroff B, Laubereau B, Grubl A, Wichmann HE et al. (2007). Certain hydrolyzed formulas reduce the incidence of atopic dermatitis but not that of asthma: three-year results of the German Infant Nutritional Intervention Study. J Allergy Clin Immunol 119, 718–725.
Wold AE (1998). The hygiene hypothesis revised: is the rising frequency of allergy due to changes in the intestinal flora? Allergy 53, 20–25.
Acknowledgements
This study was supported by Blédina, France (Danone group). We thank the families who kindly accepted to participate in the study, Ms Catherine Guitton for the dietetic follow-up and food products monitoring, Ms Valérie Brenas-Barrière for her help in the conduct of the study and Dr Guillaume Hébert from SC Partners who assisted in preparing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Cécile Aubert-Jacquin is the Danone employee. Martine Morisset, Denise-Anne Moneret-Vautrin and Christophe Dupont have received honoraria and/or compensation with regards to the study, as an investigator, coordinator or expert, in relation with the time spent on the study. The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Morisset, M., Aubert-Jacquin, C., Soulaines, P. et al. A non-hydrolyzed, fermented milk formula reduces digestive and respiratory events in infants at high risk of allergy. Eur J Clin Nutr 65, 175–183 (2011). https://doi.org/10.1038/ejcn.2010.250
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2010.250
Keywords
This article is cited by
-
Safety and efficacy of adding postbiotics in infant formula: a systematic review and meta-analysis
Pediatric Research (2024)
-
Early life microbial exposures and allergy risks: opportunities for prevention
Nature Reviews Immunology (2021)
-
Food Allergy: Our Evolving Understanding of Its Pathogenesis, Prevention, and Treatment
Current Allergy and Asthma Reports (2016)
-
Fermented infant formulas without live bacteria: a systematic review
European Journal of Pediatrics (2015)
-
World Allergy Organization-McMaster University Guidelines for Allergic Disease Prevention (GLAD-P): Probiotics
World Allergy Organization Journal (2015)