Abstract
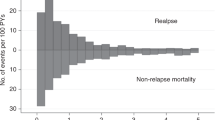
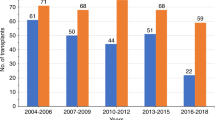
The impact of extramedullary disease (EMD) in AML on the outcomes of allogeneic hematopoietic cell transplantation (alloHCT) is unknown. Using data from the Center for International Blood and Marrow Transplant Research, we compared the outcomes of patients who had EMD of AML at any time before transplant, with a cohort of AML patients without EMD. We reviewed data from 9797 AML patients including 814 with EMD from 310 reporting centers and 44 different countries, who underwent alloHCT between and 1995 and 2010. The primary outcome was overall survival (OS) after alloHCT. Secondary outcomes included leukemia-free survival (LFS), relapse rate and treatment-related mortality (TRM). In a multivariate analysis, the presence of EMD did not affect either OS (hazard ratio 1.00, 95% confidence interval (CI) 0.91–1.09), LFS (0.98, 0.89–1.09), TRM (relative risk 0.92, 95% CI 0.80–1.16, P=0.23) or relapse (relative risk=1.03, 95% CI, 0.92–1.16; P=0.62). Furthermore, the outcome of patients with EMD was not influenced by the location, timing of EMD, or intensity of conditioning regimen. The presence of EMD in AML does not affect transplant outcomes and should not be viewed as an independent adverse prognostic feature.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Byrd JC, Edenfield WJ, Shields DJ, Dawson NA . Extramedullary myeloid cell tumors in acute nonlymphocytic leukemia: a clinical review. J Clin Oncol 1995; 13: 1800–1816.
Chang H, Brandwein J, Yi QL, Chun K, Patterson B, Brien B . Extramedullary infiltrates of AML are associated with CD56 expression, 11q23 abnormalities and inferior clinical outcome. Leuk Res 2004; 28: 1007–1011.
Byrd JC, Weiss RB, Arthur DC, Lawrence D, Baer MR, Davey F et al. Extramedullary leukemia adversely affects hematologic complete remission rate and overall survival in patients with t(8;21)(q22;q22): results from Cancer and Leukemia Group B 8461. J Clin Oncol 1997; 15: 466–475.
Shimizu H, Saitoh T, Hatsumi N, Takada S, Yokohama A, Handa H et al. Clinical significance of granulocytic sarcoma in adult patients with acute myeloid leukemia. Cancer Sci 2012; 103: 1513–1517.
Chevallier P, Mohty M, Lioure B, Michel G, Contentin N, Deconinck E et al. Allogeneic hematopoietic stem-cell transplantation for myeloid sarcoma: a retrospective study from the SFGM-TC. J Clin Oncol 2008; 26: 4940–4943.
Harris AC, Kitko CL, Couriel DR, Braun TM, Choi SW, Magenau J et al. Extramedullary relapse of acute myeloid leukemia following allogeneic hematopoietic stem cell transplantation: incidence, risk factors and outcomes. Haematologica 2013; 98: 179–184.
Craddock C, Nagra S, Peniket A, Brookes C, Buckley L, Nikolousis E et al. Factors predicting long-term survival after T-cell depleted reduced intensity allogeneic stem cell transplantation for acute myeloid leukemia. Haematologica 2010; 95: 989–995.
Schmid C, Schleuning M, Schwerdtfeger R, Hertenstein B, Mischak-Weissinger E, Bunjes D et al. Long-term survival in refractory acute myeloid leukemia after sequential treatment with chemotherapy and reduced-intensity conditioning for allogeneic stem cell transplantation. Blood 2006; 108: 1092–1099.
Lee KH, Lee JH, Choi SJ, Kim S, Seol M, Lee YS et al. Bone marrow vs extramedullary relapse of acute leukemia after allogeneic hematopoietic cell transplantation: risk factors and clinical course. Bone Marrow Transplant 2003; 32: 835–842.
Kogut N, Tsai NC, Thomas SH, Palmer J, Paris T, Murata-Collins J et al. Extramedullary relapse following reduced intensity allogeneic hematopoietic cell transplant for adult acute myelogenous leukemia. Leuk Lymphoma 2013; 54: 665–668.
Slovak ML, Kopecky KJ, Cassileth PA, Harrington DH, Theil KS, Mohamed A et al. Karyotypic analysis predicts outcome of preremission and postremission therapy in adult acute myeloid leukemia: a Southwest Oncology Group/Eastern Cooperative Oncology Group Study. Blood 2000; 96: 4075–4083.
Giralt S, Ballen K, Rizzo D, Bacigalupo A, Horowitz M, Pasquini M et al. Reduced-intensity conditioning regimen workshop: defining the dose spectrum. Report of a workshop convened by the center for international blood and marrow transplant research. Biol Blood Marrow Transplant 2009; 15: 367–369.
Champlin R, Khouri I, Shimoni A, Gajewski J, Kornblau S, Molldrem J et al. Harnessing graft-versus-malignancy: non-myeloablative preparative regimens for allogeneic haematopoietic transplantation, an evolving strategy for adoptive immunotherapy. Br J Haematol 2000; 111: 18–29.
Gooley TA, Leisenring W, Crowley J, Storer BE . Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med 1999; 18: 695–706.
Fang M, Storer B, Estey E, Othus M, Zhang L, Sandmaier BM et al. Outcome of patients with acute myeloid leukemia with monosomal karyotype who undergo hematopoietic cell transplantation. Blood 2011; 118: 1490–1494.
Brunet S, Labopin M, Esteve J, Cornelissen J, Socie G, Iori AP et al. Impact of FLT3 internal tandem duplication on the outcome of related and unrelated hematopoietic transplantation for adult acute myeloid leukemia in first remission: a retrospective analysis. J Clin Oncol 2012; 30: 735–741.
Falini B, Lenze D, Hasserjian R, Coupland S, Jaehne D, Soupir C et al. Cytoplasmic mutated nucleophosmin (NPM) defines the molecular status of a significant fraction of myeloid sarcomas. Leukemia 2007; 21: 1566–1570.
Acknowledgements
We acknowledge the following contributing co-authors for their contributions to this manuscript: Kirk R Schultz, Mark R Litzow, Philip L McCarthy, Mahmoud D Aljurf, Mitchell S Cairo, William A Wood, Celalettin Ustun, Thomas R Klumpp, Edwin M. Horwitz, Jyotishankar Raychaudhuri, Bruce M Camitta, Yi-Bin Chen, Peter H Wiernik, Tsiporah B Shore, Selina M Luger, Ashish Bajel, Harry C Schouten, Grace H Ku, Maxim Norkin, Faiz Anwer, Asmita Mishra, Attaphol Pawarode, Amelia Langston, Mitchell Sabloff, Ann E Woolfrey, Hans-Jochem Kolb, Edmund K Waller, Usama Gergis, John Koreth, Reinhold Munker, Joseph McGuirk, William R Drobyski, Bita Jalilizeinali and H Jean Khoury. Geoffrey L Uy’s contribution is supported by NCI grant K23 CA140707. The CIBMTR is supported by Public Health Service Grant/Cooperative Agreement U24-CA076518 from the National Cancer Institute (NCI), the National Heart, Lung and Blood Institute (NHLBI) and the National Institute of Allergy and Infectious Diseases (NIAID); a Grant/Cooperative Agreement 5U10HL069294 from NHLBI and NCI; a contract HHSH250201200016C with Health Resources and Services Administration (HRSA/DHHS); two grants N00014-12-1-0142 and N00014-13-1-0039 from the Office of Naval Research; and grants from *Actinium Pharmaceuticals, Allos Therapeutics, Inc. and *Amgen, Inc. Anonymous donation to the Medical College of Wisconsin, Ariad, Be the Match Foundation, *Blue Cross and Blue Shield Association, *Celgene Corporation, Chimerix, Inc., Fred Hutchinson Cancer Research Center, Fresenius-Biotech North America, Inc., *Gamida Cell Teva Joint Venture Ltd., Genentech, Inc.,*Gentium SpA, Genzyme Corporation, GlaxoSmithKline, Health Research, Inc., Roswell Park Cancer Institute, HistoGenetics, Inc., Incyte Corporation, Jeff Gordon Children’s Foundation, Kiadis Pharma, The Leukemia & Lymphoma Society, Medac GmbH, The Medical College of Wisconsin, Merck & Co, Inc., Millennium: The Takeda Oncology Co., *Milliman USA, Inc., *Miltenyi Biotec, Inc., National Marrow Donor Program, Onyx Pharmaceuticals, Optum Healthcare Solutions, Inc., Osiris Therapeutics, Inc., Otsuka America Pharmaceutical, Inc., Perkin Elmer, Inc., *Remedy Informatics, *Sanofi US, Seattle Genetics, Sigma-Tau Pharmaceuticals, Soligenix, Inc., St Baldrick’s Foundation, StemCyte, A Global Cord Blood Therapeutics Co., Stemsoft Software, Inc., Swedish Orphan Biovitrum, *Tarix Pharmaceuticals, *TerumoBCT, *Teva Neuroscience, Inc., *THERAKOS, Inc., University of Minnesota, University of Utah and *Wellpoint, Inc. *Corporate members. The views expressed in this article do not reflect the official policy or position of the National Institute of Health, the Department of the Navy, the Department of Defense, Health Resources and Services Administration (HRSA) or any other agency of the U.S. Government.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Presented in part at the 2014 BMT Tandem Meetings, Grapevine, TX, 2 March 2014.
Supplementary Information accompanies this paper on Bone Marrow Transplantation website
Rights and permissions
About this article
Cite this article
Goyal, S., Zhang, MJ., Wang, HL. et al. Allogeneic hematopoietic cell transplant for AML: no impact of pre-transplant extramedullary disease on outcome. Bone Marrow Transplant 50, 1057–1062 (2015). https://doi.org/10.1038/bmt.2015.82
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2015.82
This article is cited by
-
Salvage hematopoietic stem cell transplantation for patients with higher leukemia burden in relapsed or refractory acute myeloid leukemia: a ten-year study
Annals of Hematology (2023)
-
Myeloid sarcoma: more and less than a distinct entity
Annals of Hematology (2023)
-
Allogeneic hematopoietic stem cell transplantation for adults with therapy-related acute myeloid leukaemia: a retrospective multicentre study on behalf of the SFGM-TC
Bone Marrow Transplantation (2023)
-
Effect of extramedullary disease on allogeneic hematopoietic cell transplantation for pediatric acute myeloid leukemia: a nationwide retrospective study
Bone Marrow Transplantation (2021)
-
Better disease control before allogeneic stem cell transplantation is crucial to improve the outcomes of transplantation for acute myeloid leukemia patients with extramedullary disease
Bone Marrow Transplantation (2020)