Abstract
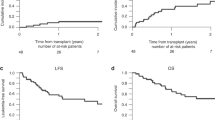
We investigated the administration of i.v. BU combined with melphalan (Mel) in patients with ALL undergoing allogeneic hematopoietic SCT. Forty-seven patients with a median age of 33 years (range 20–61) received a matched sibling (n=27) or matched unrelated donor transplant (n=20) for ALL in first CR (n=26), second CR (n=13), or with more advanced disease (n=8). BU was infused daily for 4 days, either at a fixed dose of 130 mg/m2 (5 patients) or using pharmacokinetic (PK) dose adjustment (42 patients), to target an average daily area-under-the-curve (AUC) of 5000 μmol/min, determined by a test dose of i.v. BU at 32 mg/m2. This was followed by a rest day, then two daily doses of Mel at 70 mg/m2. Stem cells were infused on the following day. The 2-year OS, PFS and non-relapse mortality (NRM) rates were 35% (95% confidence interval (CI), 23–51%), 31% (95% CI, 21–48%) and 37% (95% CI, 23–50%), respectively. Acute NRM at 100 days was favorable at 12% (95% CI, 5–24%); however, the 2-year NRM was significantly higher for patients older than 40 years, 58% vs 20%, mainly due to GVHD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Oliansky DM, Larson RA, Weisdorf D, Dillon H, Ratko TA, Wall D et al. The role of cytotoxic therapy with hematopoietic stem cell transplantation in the treatment of adult acute lymphoblastic leukemia: update of the 2006 evidence-based review. Biol Blood Marrow Transplant 2012; 18: 505–522.
Sutton L, Kuentz M, Cordonnier C, Blaise D, Devergie A, Guyotat D et al. Allogeneic bone marrow transplantation for adult acute lymphoblastic leukemia in first complete remission: factors predictive of transplant-related mortality and influence of total body irradiation modalities. Bone Marrow Transplant 1993; 12: 583–589.
Goldstone AH, Richards SM, Lazarus HM, Tallman MS, Buck G, Fielding AK et al. In adults with standard-risk acute lymphoblastic leukemia, the greatest benefit is achieved from a matched sibling allogeneic transplantation in first complete remission, and an autologous transplantation is less effective than conventional consolidation/maintenance chemotherapy in all patients: final results of the International ALL Trial (MRC UKALL XII/ECOG E2993). Blood 2008; 111: 1827–1833.
Kelsey CR, Horwitz ME, Chino JP, Craciunescu O, Steffey B, Folz RJ et al. Severe pulmonary toxicity after myeloablative conditioning using total body irradiation: an assessment of risk factors. Int J Radiat Oncol Biol Phys 2011; 81: 812–818.
Cohen A, van Lint MT, Uderzo C, Rovelli A, Lavagetto A, Vitale V et al. Growth in patients after allogeneic bone marrow transplant for hematological diseases in childhood. Bone Marrow Transplant 1995; 15: 343–348.
Liesner RJ, Leiper AD, Hann IM, Chessells JM . Late effects of intensive treatment for acute myeloid leukemia and myelodysplasia in childhood. J Clin Oncol 1994; 12: 916–924.
Rizzo JD, Curtis RE, Socie G, Sobocinski KA, Gilbert E, Landgren O et al. Solid cancers after allogeneic hematopoietic cell transplantation. Blood 2009; 113: 1175–1183.
Hassan M, Ljungman P, Bolme P, Ringden O, Syruckova Z, Bekassy A et al. Busulfan bioavailability. Blood 1994; 84: 2144–2150.
Jones RB . Clinical pharmacology of melphalan and its implications for clinical resistance to anticancer agents. Cancer Treat Res 2002; 112: 305–322.
Madden T, de Lima M, Thapar N, Nguyen J, Roberson S, Couriel D et al. Pharmacokinetics of once-daily IV busulfan as part of pretransplantation preparative regimens: a comparison with an every 6-hour dosing schedule. Biol Blood Marrow Transplant 2007; 13: 56–64.
Balis FM, Poplack DG . Central nervous system pharmacology of antileukemic drugs. Am J Pediatr Hematol Oncol 1989; 11: 74–86.
Cony-Makhoul P, Marit G, Boiron JM, Puntous M, Reiffers J . Busulphan and melphalan prior to autologous transplantation for myeloid malignancies. Bone Marrow Transplant 1995; 16: 69–70.
Meloni G, Capria S, Vignetti M, Alimena G, de Fabritiis P, Montefusco E et al. Ten-year follow-up of a single center prospective trial of unmanipulated peripheral blood stem cell autograft and interferon-alpha in early phase chronic myeloyd leukemia. Haematologica 2001; 86: 596–601.
Kebriaei P, Madden T, Kazerooni R, Wang X, Thall PF, Ledesma C et al. Intravenous busulfan plus melphalan is a highly effective, well-tolerated preparative regimen for autologous stem cell transplantation in patients with advanced lymphoid malignancies. Biol Blood Marrow Transplant 2011; 17: 412–420.
Blanes M, de la Rubia J, Lahuerta JJ, Gonzalez JD, Ribas P, Solano C et al. Single daily dose of intravenous busulfan and melphalan as a conditioning regimen for patients with multiple myeloma undergoing autologous stem cell transplantation: a phase II trial. Leuk Lymphoma 2009; 50: 216–222.
Vey N, De Prijck B, Faucher C, Stoppa AM, Sainty D, Lafage M et al. A pilot study of busulfan and melphalan as preparatory regimen prior to allogeneic bone marrow transplantation in refractory or relapsed hematological malignancies. Bone Marrow Transplant 1996; 18: 495–499.
Small TN, Young JW, Castro-Malaspina H, Prockop S, Wilton A, Heller G et al. Intravenous busulfan and melphalan, tacrolimus, and short-course methotrexate followed by unmodified HLA-matched related or unrelated hematopoietic stem cell transplantation for the treatment of advanced hematologic malignancies. Biol Blood Marrow Transplant 2007; 13: 235–244.
Watanabe T, Kajiume T, Abe T, Kawano Y, Iwai A, Iwai T et al. Allogeneic peripheral blood stem cell transplantation in children with hematologic malignancies from HLA-matched siblings. Med Pediatr Oncol 2000; 34: 171–176.
Matsuyama T, Kojima S, Kato K . Allogeneic bone marrow transplantation for childhood leukemia following a busulfan and melphalan preparative regimen. Bone Marrow Transplant 1998; 22: 21–26.
Martino R, Badell I, Brunet S, Sureda A, Torras A, Cubells J et al. High-dose busulfan and melphalan before bone marrow transplantation for acute nonlymphoblastic leukemia. Bone Marrow Transplant 1995; 16: 209–212.
Russell JA, Tran HT, Quinlan D, Chaudhry A, Duggan P, Brown C et al. Once-daily intravenous busulfan given with fludarabine as conditioning for allogeneic stem cell transplantation: study of pharmacokinetics and early clinical outcomes. Biol Blood Marrow Transplant 2002; 8: 468–476.
Andersson BS, Thall PF, Madden T, Couriel D, Wang X, Tran HT et al. Busulfan systemic exposure relative to regimen-related toxicity and acute graft-versus-host disease: defining a therapeutic window for i.v. BuCy2 in chronic myelogenous leukemia. Biol Blood Marrow Transplant 2002; 8: 477–485.
de Lima M, Couriel D, Thall PF, Wang X, Madden T, Jones R et al. Once-daily intravenous busulfan and fludarabine: clinical and pharmacokinetic results of a myeloablative, reduced-toxicity conditioning regimen for allogeneic stem cell transplantation in AML and MDS. Blood 2004; 104: 857–864.
D'Argenio DZ, Schumitzky A . ADAPT II User’s Guide: Pharmacokinetic/Pharmacodynamic Systems Analysis Software.. Los Angeles: Biomedical Simulations Resource, 1997.
Andersson BS, Madden T, Tran HT, Hu WW, Blume KG, Chow DS et al. Acute safety and pharmacokinetics of intravenous busulfan when used with oral busulfan and cyclophosphamide as pretransplantation conditioning therapy: a phase I study. Biol Blood Marrow Transplant 2000; 6: 548–554.
Kebriaei P, Saliba R, Rondon G, Chiattone A, Luthra R, Anderlini P et al. Long-term follow-up of allogeneic hematopoietic stem cell transplantation for patients with philadelphia chromosome-positive acute lymphoblastic leukemia: impact of tyrosine kinase inhibitors on treatment outcomes. Biol Blood Marrow Transplant 2012; 18: 584–592.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J et al. 1994 consensus conference on acute GVHD grading. Bone Marrow Transplant 1995; 15: 825–828.
Sullivan KM, Shulman HM, Storb R, Weiden PL, Witherspoon RP, McDonald GB et al. Chronic graft-versus-host disease in 52 patients: adverse natural course and successful treatment with combination immunosuppression. Blood 1981; 57: 267–276.
Thall PF, Wathen JK . Covariate-adjusted adaptive randomization in a sarcoma trial with multi-stage treatments. Stat Med 2005; 24: 1947–1964.
Meier P, Kaplan EL . Non-parametric estimation from incomplete observations. J Am Statist Assoc 1958; 53: 457–481.
Mantel N . Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemotherapy Reports 1966; 50: 163–170.
Jones RJ, Lee KS, Beschorner WE, Vogel VG, Grochow LB, Braine HG et al. Venoocclusive disease of the liver following bone marrow transplantation. Transplantation 1987; 44: 778–783.
Lahuerta JJ, Mateos MV, Martinez-Lopez J, Grande C, de la Rubia J, Rosinol L et al. Busulfan 12 mg/kg plus melphalan 140 mg/m2 versus melphalan 200 mg/m2 as conditioning regimens for autologous transplantation in newly diagnosed multiple myeloma patients included in the PETHEMA/GEM2000 study. Haematologica 2010; 95: 1913–1920.
Grigg AP, Stone J, Milner AD, Schwarer AP, Wolf M, Prince HM et al. Phase II study of autologous stem cell transplant using busulfan-melphalan chemotherapy-only conditioning followed by interferon for relapsed poor prognosis follicular non-Hodgkin lymphoma. Leuk Lymphoma 2010; 51: 641–649.
Lahuerta JJ, Martinez-Lopez J, Grande C, Blade J, de la Serna J, Alegre A et al. Conditioning regimens in autologous stem cell transplantation for multiple myeloma: a comparative study of efficacy and toxicity from the Spanish Registry for Transplantation in Multiple Myeloma. Br J Haematol 2000; 109: 138–147.
Marks DI, Wang T, Perez WS, Antin JH, Copelan E, Gale RP et al. The outcome of full-intensity and reduced-intensity conditioning matched sibling or unrelated donor transplantation in adults with Philadelphia chromosome-negative acute lymphoblastic leukemia in first and second complete remission. Blood 2010; 116: 366–374.
Tang W, Wang L, Zhao WL, Chen YB, Shen ZX, Hu J . Intravenous busulfan-cyclophosphamide as a preparative regimen before allogeneic hematopoietic stem cell transplantation for adult patients with acute lymphoblastic leukemia. Biol Blood Marrow Transplant 2011; 17: 1555–1561.
Valdez BC, Li Y, Murray D, Champlin RE, Andersson BS . The synergistic cytotoxicity of clofarabine, fludarabine and busulfan in AML cells involves ATM pathway activation and chromatin remodeling. Biochemical Pharmacology 2011; 81: 222–232.
Andersson BS, Valdez BC, de Lima M, Wang X, Thall PF, Worth LL et al. Clofarabine +/- fludarabine with once daily i.v. busulfan as pretransplant conditioning therapy for advanced myeloid leukemia and MDS. Biol Blood Marrow Transplant 2011; 17: 893–900.
Santarone S, Pidala J, Di Nicola M, Field T, Alsina M, Ayala E et al. Fludarabine and pharmacokinetic-targeted busulfan before allografting for adults with acute lymphoid leukemia. Biol Blood Marrow Transplant 2011; 17: 1505–1511.
O'Connor D, Sibson K, Caswell M, Connor P, Cummins M, Mitchell C et al. Early UK experience in the use of clofarabine in the treatment of relapsed and refractory paediatric acute lymphoblastic leukaemia. Br J Haematol 2011; 154: 482–485.
Jeha S, Gandhi V, Chan KW, McDonald L, Ramirez I, Madden R et al. Clofarabine, a novel nucleoside analog, is active in pediatric patients with advanced leukemia. Blood 2004; 103: 784–789.
Kebriaei P, L C, Culotta K, Shpall E, Ciurea S, Alousi A et al. (eds). Clofarabine (Clo) and Busulfan as a Novel Reduced Toxicity Conditioning Regimen for Allogeneic Hematopoietic Cell Transplantation (HCT) in Patients (pts) with Acute Lymphoblastic Leukemia (ALL), Blood, American Society of Hematology Orlando, Florida, 2010.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kebriaei, P., Madden, T., Wang, X. et al. Intravenous BU plus Mel: an effective, chemotherapy-only transplant conditioning regimen in patients with ALL. Bone Marrow Transplant 48, 26–31 (2013). https://doi.org/10.1038/bmt.2012.114
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2012.114
Keywords
This article is cited by
-
Effects of combined test dose and therapeutic drug monitoring strategy in exposure-directed busulfan
Annals of Hematology (2023)
-
Adding melphalan to fludarabine and a myeloablative dose of busulfan improved survival after allogeneic hematopoietic stem cell transplantation in a propensity score-matched cohort of hematological malignancies
Bone Marrow Transplantation (2021)
-
Fludarabine with pharmacokinetically guided IV busulfan is superior to fixed-dose delivery in pretransplant conditioning of AML/MDS patients
Bone Marrow Transplantation (2017)