Abstract
Aspirin may reduce the risk of cancer at some sites but its effect at the lung is unclear. We prospectively examined associations between aspirin use and risk of lung cancer in 109 348 women in the Nurses' Health study from 1980 to 2004. During this time, 1360 lung cancers were documented in participants 36–82 years of age. Aspirin use and smoking were assessed every 2 years. Risk of lung cancer was a non-significant 16% lower for regular aspirin users of one or two tablets per week and a significant 55% higher for users of 15 or more tablets per week compared with women who never regularly used aspirin. Results were similar when limited to never smokers. For both the low and high quantity aspirin users, risk of lung cancer did not decline or increase with longer durations of use, and associations attenuated as the latency period between aspirin assessment and lung cancer diagnosis was lengthened. Our findings, together with those from previous clinical trials and prospective studies, do not provide consistent evidence that aspirin influences the development of lung cancer and further investigation is required with adjustment for smoking.
Similar content being viewed by others
Main
Regular use of aspirin reduces the risk of colorectal adenoma and cancer (Benamouzig et al, 2005), most likely through the inhibition of cyclooxygenase enzymes, restoration of normal apoptosis, and reduction of angiogenesis (Zha et al, 2004). However, the influence of aspirin at other tumour sites is less clear. For lung cancer, several meta-analyses with different coverage of published results have offered varying results and interpretations. Of the two that focused solely on lung cancer, aspirin users were found to have a significant 27% lower risk in one (Khuder et al, 2005) and a non-significant 9% lower risk in the other (Hernandez-Diaz and Rodriguez, 2007). In two other meta-analyses in which lung cancer was embedded in a wider review of many cancers, the first reported a 16% lower risk for aspirin users that was compatible with no effect or a slightly reduced risk (Gonzalez-Perez et al, 2003), and the other, limited to cohort studies, reported no association (Bosetti et al, 2006). In general, these meta-analyses concluded that a chemopreventive value of aspirin for lung cancer should be interpreted with caution owing to the limitations of the available studies and the heterogeneity of study designs and results. The authors called for larger studies with better exposure characterisation of dose–response measures and detailed adjustment for smoking. In an attempt to resolve these uncertainties, we examined relations between regular aspirin use and risk of invasive lung cancer among women in the Nurses' Health Study (NHS) cohort, taking into consideration quantity, frequency, and duration of use and latency between exposure and diagnosis, while controlling for detailed smoking characteristics. We placed our results within a wider context by conducting a literature review focused on clinical trials and prospective studies that, like our study, had a quantitative measure of aspirin use and controlled for smoking in analyses.
Methods
Study population
The NHS was established in 1976 when 121 700 female registered nurses, 30–55 years of age, returned a mailed questionnaire. The NHS was approved by the Institutional Review Board of the Brigham and Women's Hospital in Boston. Using mailed questionnaires, participants provided a disease history and information about their personal characteristics and behaviours at baseline, and every 2 years they have updated and extended these data and reported newly diagnosed diseases on follow-up questionnaires. Deaths are commonly reported by families or the postal service and are confirmed through the National Death Index.
For this investigation, analysis began in 1980 when aspirin use was first ascertained, and follow-up for incident lung cancer continued until June 2004. The baseline study population consisted of the women who had not reported a diagnosis of cancer (except non-melanoma skin cancer) and provided information on their smoking status and aspirin use. A total of 109 348 women, 34–82 years of age over the follow-up period, contributed approximately 1.9 million person-years. The follow-up rate for this study population was 95%.
Lung cancer cases
When lung cancer was reported by a cohort member or identified from a death report, we sought medical records for confirmation and date of diagnosis. Between 1980 and 2004, 1446 incident lung cancers were reported among the women in this study population, of which 1360 were confirmed as cases, with a median age at diagnosis of 65 years (range 36–82 years). The majority of lung cancers were adenocarcinomas (46%), small cell (17%), or squamous cell (15%) tumours. The 86 unconfirmed lung cancers were either metastatic from another site or lacked a follow-up response from the participant and were censored at time of self-report.
Aspirin assessment
Aspirin use was first assessed in 1980, when participants were asked if they used aspirin in most weeks, and if so, to write the number of years of use and the number of aspirin tablets consumed per week. On subsequent biennial questionnaires, participants were asked if they were a regular aspirin user over the past 2 years and tablets per week and/or days per week of use were assessed with categorical responses. In 2000 and 2002, participants were additionally asked whether they took standard dose (325 mg or more) or low-dose (100 mg or less) aspirin. We converted reported tablets per week of low-dose aspirin into the equivalent tablets of standard dose.
In this analysis, we reclassified participants in each 2-year follow-up cycle by status and quantity (1–2, 3–5, 6–14, ⩾15 tablets per week) of aspirin use. A participant was classified as a current aspirin user if she reported at least 1 tablet per week or 1 day per week of regular use for the last 2 years; a past user if she did not qualify as a current user but had been previously classified as such; or a never user if she had never been classified as a current user during cohort follow-up. Duration was calculated for current users in each follow-up cycle as years of continuous aspirin use.
To determine reasons for aspirin use in this cohort, we had queried a sample of 100 women who reported taking 1–6 tablets per week in 1980, 1982, and 1984 (90% response) and 100 women who reported taking 7 or more tablets per week in those years (92% response). In both groups, the major reasons for aspirin use were headache, arthritis, and/or musculoskeletal pain. Less than 10% of both the lower and higher quantity users cited cardiovascular disease prevention.
Smoking and other covariates
On the initial NHS questionnaire, participants reported whether they were a current smoker or had ever smoked in the past, the age at which they began to smoke, the number of cigarettes typically smoked in a day, and the age at which they stopped smoking. On each subsequent biennial questionnaire, participants again reported whether they currently smoked and their daily quantity of cigarette use. In this analysis, we reclassified participants in each 2-year follow-up cycle by smoking status, cigarettes smoked per day among current smokers, time since quitting among past smokers, and age at start of smoking among ever smokers. Pack-years were also calculated in each cycle as the product of years of smoking and packs of cigarettes smoked per day.
Height was reported in 1976 and body mass index (BMI in kg m−2) was calculated with each biennial report of body weight. Menopausal status and use of oestrogen replacement therapy were updated every 2 years. The total number of hours per week of physical activity was ascertained in 1980 and on eight subsequent questionnaires. Diet was assessed with a food frequency questionnaire six times beginning in 1980 and daily intakes of fruits, vegetables, α-carotene, lycopene, vitamin C, vitamin E, total fat, and alcohol were calculated.
Statistical analysis
Participants contributed person-time from the return date of their 1980 questionnaire until a report of any other cancer except non-melanoma skin cancer, death, or end of follow-up on 1 June 2004. The median follow-up time per participant was 23.7 years. The most recent data on aspirin use and all covariates were used to allocate person-time to the appropriate category for each variable at the beginning of every 2-year follow-up cycle. Participants did not contribute person-time in any follow-up cycle in which they were missing aspirin or smoking status.
Age-adjusted incidence rates of lung cancer were determined within categories of aspirin use and relative risks (RRs) were calculated as the ratio of the rate in each category of use compared with the rate in never users. We used Cox proportional hazards models to adjust simultaneously for age, smoking characteristics, and the other potential confounders. To assess dose–response effects for duration of aspirin use, P-values for linear trend were calculated using medians per category.
Literature review
Relevant papers were sought electronically and by hand. The PubMed database was searched without limits for ‘aspirin and lung and cancer’. Reference lists of all articles of interest were scanned for additional manuscripts and all papers citing two of the earliest relevant publications (Peto et al, 1988; Thun et al, 1993) were tracked through Science Citation Index. We reviewed all papers with original data that assessed the relation between aspirin use and lung cancer incidence or mortality. We excluded retrospective analyses (Rosenberg 1995; Harris et al, 2002; Moysich et al, 2002; Muscat et al, 2003) because of potential biases in aspirin recall and selection of controls and the observed heterogeneity of results from case-control and cohort studies in previous reviews. We also excluded prospective studies that did not account for smoking (Paganini-Hill et al, 1989; Friis et al, 2003; Sorensen et al, 2003) or had no quantitative measure of aspirin use (Shreinemachers and Everson, 1994; Ratnasinghe et al, 2004) because these factors likely influence the association between aspirin and lung cancer. After exclusions, we identified nine studies that met our criteria for inclusion in the review (see Table 4). Confidence intervals (CIs) were missing from one paper (Peto et al, 1988) for which we calculated approximate intervals around the RR.
Results
The characteristics of the women in the study population are shown in Table 1 by status and quantity of regular aspirin use over the 1980–2004 follow-up period. Current and past aspirin users were older than those who never used aspirin on a regular basis (57 and 60 vs 53 years, respectively). After adjusting for age differences, the percent of women who smoked varied little between the never (21%) and current (18%) aspirin users, though among the current users, the likelihood of smoking and daily cigarette use increased with increasing aspirin tablets per week. The highest quantity aspirin users had the highest BMI and the post-menopausal women who never used aspirin regularly were least likely to use oestrogen replacement therapy. No differences by aspirin use were detected for the dietary variables nor for the single assessment of exposure to second-hand smoke in 1982 (ie number of adult years spent living with someone who smoked regularly, and hours per week exposed to cigarette smoke from people at home or work) (data not shown).
In the age-adjusted analysis, current aspirin users had a modestly lower risk of lung cancer when compared with women who never used aspirin regularly, and the RR was attenuated and became null after the smoking factors were added to the model (Table 2). However, when we examined the influence of aspirin according to quantity consumed, we observed a non-significant 16% lower risk for current users of 1 or 2 tablets per week (low quantity), a significant 55% increased risk for current users of 15 or more tablets per week (high quantity), and no risk or benefit for women using between 3 and 14 tablets per week. Results were essentially unchanged when all covariates were added to the model or when pack-years were used to control for smoking instead of age at start, years since quit, and cigarettes per day (data not shown). Frequency of aspirin use exhibited an association with lung cancer that was similar to that seen for quantity, with a lower risk for use on 1 day per week (RR=0.85, 95% CI 0.69–1.04) and an increased risk for use on 6 or 7 days per week (RR=1.20, 95% CI 1.00–1.40).
Associations between aspirin use and lung cancer did not differ by smoking status. Even among the never smokers with 128 lung cancers, the association was positive for high quantity users (RR=1.70, 95% CI 0.67–4.31) and inverse for low quantity aspirin users (RR=0.71, 95% CI 0.39–1.30). However, the inverse association was only evident for adenocarcinoma tumours (RR=0.83, 95% CI 0.62–1.10) and in women less than 70 years of age (RR=0.77, 95% CI 0.62–0.95).
Duration of aspirin use was unrelated to lung cancer risk (Table 3). With longer durations, risk neither notably decreased for low quantity users nor increased for high quantity users. We also examined timing of exposure to determine whether earlier use was more relevant than current use for lung cancer aetiology. However, for both low and high quantity users, the strongest associations were those observed for current users (diagnosis within the 2 years between aspirin assessments) and attenuated with increasing latency between aspirin assessment and lung cancer diagnosis.
The clinical trials and prospective studies of aspirin and lung cancer that were included in our review are described in Table 4. Despite the focus on research that included a quantitative measure of aspirin use and accounted for smoking in analyses, results remain inconsistent. The lowest RRs were reported from two studies (Peto et al, 1988; Akhmedkhanov et al, 2002) that had few lung cancer cases and very imprecise results. The only significant inverse association was the 27% lower risk in women in the Cancer Prevention Study II cohort who used aspirin 1–15 times per month for at least 1 year (Thun et al, 1993). Five other studies reported non-significant risk reductions. In the Women's Health Study clinical trial (Cook et al, 2005), a 22% lower risk of lung cancer was reported for the treatment group receiving 100 mg of aspirin every other day for 10 years, an amount similar to the 325 mg of aspirin once or twice a week in our NHS women. Fewer studies observed an increased risk of lung cancer for aspirin users. The only one with a significant result was a nested case-control study using the UK Health Improvement Network database in which a 53% increased risk of lung cancer was reported for men and women who received prescriptions for 150 mg or more per day of aspirin for at least 1 year (Hernandez-Diaz and Rodriguez, 2007). This increased risk is similar to what we observed for high quantity aspirin users in our cohort but the aspirin intake on which it was based is much lower.
Discussion
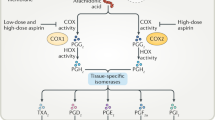
In this prospective study of women, we observed a 16% lower risk of lung cancer among regular users of one or two standard dose aspirin tablets per week and a 55% increased risk among those using 15 or more tablets per week for the past 2 years. It is difficult to interpret these diverse findings and to conclude whether one or both may be real effects or whether they are due to chance or residual confounding by smoking. The observed benefit from low quantity aspirin use may have been biased if women with heart disease were prescribed aspirin and also told to quit smoking. However, our results were unchanged when we excluded women who reported angina or a myocardial infarction. If aspirin is indeed beneficial in preventing lung cancer, it is anticipated to operate through anti-inflammatory pathways mediated by COX-2, the inducible form of the cyclooxygenase enzymes (Coussens and Werb, 2002). However, this requires higher doses of aspirin (Thun, 2000), as shown for colorectal cancer in several previous studies (Thun et al, 1993, Collet et al, 1999), including one within this NHS cohort (Chan et al, 2005). In this investigation, a lower risk of lung cancer was only observed for a quantity of aspirin use that was too low to affect COX-2. The lack of an increasing benefit with longer durations of aspirin use was also in contrast to the previous studies of colorectal cancer. If low-dose aspirin is indeed beneficial, it may act through pathways that do not involve inflammation and COX inhibition (Hanif et al, 1996).
No biologic mechanism is evident for our observed increased risk of lung cancer for women using 15 or more aspirin tablets per week, and it is likely at least partially the result of residual confounding, given the major impact of smoking on lung cancer and the positive association between smoking and aspirin use in this cohort. It is also possible that higher quantities of aspirin were used just prior to diagnosis to treat preclinical symptoms, though we did not find evidence to support this. Among the high quantity aspirin users, 12% of both the lung cancer cases and the non-cases had switched from no use or low quantity aspirin use in the previous assessment. The small size of the high quantity aspirin group prevented a more thorough examination of confounding by indication.
The prospective design of our study minimised the recall bias inherent in retrospective investigations, and the 24 years of biennial data collection provided updated information for accurate assessment of aspirin use, including status, quantity, frequency, and duration of use, and latency between aspirin exposure and lung cancer diagnosis. Self-reported aspirin use is prone to error, but is more likely to be accurate among the educated health professionals in this cohort. Moreover, such error would be random and would only have attenuated our results towards the null. The repeated biennial data collections also provided detailed and updated assessment of smoking, the primary confounder in lung cancer analyses. The importance of this confounding is demonstrated by the attenuation of the RR from 0.73 to 0.84 for women using 1–2 aspirin tablets per week when age at start of smoking, current cigarettes per day, and time since quit were added to the model. We acknowledge that measurement error remains in our smoking assessment, and it is possible that a more perfect assessment could further attenuate or nullify this association. On the other hand, we observed a lower risk of lung cancer for low quantity aspirin use even among the never smoking women in this cohort.
Our findings, together with those from previous clinical trials and prospective studies, do not offer consistent evidence that aspirin use is independently associated with risk of lung cancer. If there is indeed any effect of aspirin, future studies will require careful assessment of quantity and frequency of aspirin use, along with detailed histories of smoking and comorbidities.
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Akhmedkhanov A, Toniolo P, Zeleniuch-Jacquotte A, Koenig KL, Shore RE (2002) Aspirin and lung cancer in women. Br J Cancer 78: 49–53
Benamouzig R, Uzzan B, Little J, Chaussade S (2005) Low dose aspirin, Cox-inhibition and chemoprevention of colorectal cancer. Curr Top Med Chem 5: 493–503
Bosetti C, Gallus S, La Vecchia C (2006) Aspirin and cancer risk: an updated quantitative review to 2005. Cancer Causes Control 17: 871–888
Chan AT, Giovannucci EL, Meyerhardt JA, Schernhammer ES, Curhan GC, Fuchs CS (2005) Long-term use of aspirin and nonsteroidal anti-inflammatory drugs and risk of colorectal cancer. JAMA 294: 914–923
Collet JP, Sharpe C, Belzile E, Boivin JF, Hanley J, Abenhaim L (1999) Colorectal cancer prevention by non-steroidal anti-inflammatory drugs: effects of dosage and timing. Br J Cancer 81: 62–68
Cook NR, Lee IM, Gaziano JM, Gordon D, Ridker PM, Manson JE, Hennekens CH, Buring JE (2005) Low-dose aspirin in the primary prevention of cancer. JAMA 294: 47–55
Coussens LM, Werb Z (2002) Inflammation and cancer. Nature 420: 860–867
Friis S, Sorensen HT, McLaughlin JK, Johnsen SP, Biot WJ, Olsen JH (2003) A population-based cohort study of the risk of colorectal and other cancers among users of low-dose aspirin. Br J Cancer 88: 684–688
Gonzalez-Perez A, Rodriguez LAG, Lopez-Ridaura R (2003) Effects of non-steroidal anti-inflammatory drugs on cancer sites other than the colon and rectum: a meta-analysis. BMC Cancer 3: 28–39
Hanif R, Pittas A, Feng Y, Koutos MI, Qiao L, Staiano-Coico L, Shiff SI, Rigas B (1996) Effects of nonsteroidal anti-inflammatory drugs on proliferation and on induction of apoptosis in colon cancer cells by a prostaglandin-independent pathway. Biochem Pharmacol 52: 237–245
Harris RE, Beebe-Donk J, Schuller HM (2002) Chemoprevention of lung cancer by non-steroidal anti-inflammatory drugs among cigarette smokers. Oncol Rep 9: 693–695
Hayes JK, Anderson KE, Folsom AR (2006) Association between nonsteroidal anti-inflammatory drug use and the incidence of lung cancer in the Iowa Women's Health Study. Cancer Epidemiol Biomarkers Prev 15: 2226–2231
Hernandez-Diaz S, Rodriguez LAG (2007) Nonsteroidal anti-inflammatory drugs and risk of lung cancer. Int J Cancer 120: 1565–1572
Holick CN, Michaud DS, Leitzmann MF, Willett WC, Giovannucci E (2003) Aspirin use and lung cancer in men. Br J Cancer 89: 1705–1708
Khuder SA, Herial NA, Mutgi AB, Federman DJ (2005) Nonsteroidal antiinflammatory drug use and lung cancer. Chest 127: 748–754
Langman MJS, Cheng KK, Gilman EA, Lancashire RJ (2000) Effect of anti-inflammatory drugs on overall risk of common cancer: case-control study in general practice research database. BMJ 320: 1642–1646
Lee IM, Manson JE, Hennekens CH, Buring JE (1994) Low-dose aspirin and risk of cancer: the Physician's Health Study (abstract). Am J Epidemiol 141: S28
Moysich KB, Menezes RJ, Ronsani A, Swede H, Reid ME, Cummings KM, Falkner KL, Loewen GM, Bepler G (2002) Regular aspirin use and lung cancer risk. BMC Cancer 2: 31–37
Muscat JE, Chen S, Richie JP, Altorki NK, Citron M, Olson S, Neugut AI, Stellman SD (2003) Risk of lung carcinoma among users of nonsteroidal antiinflammatory drugs. Cancer 97: 1732–1736
Paganini-Hill A, Chao A, Ross RK, Henderson BE (1989) Aspirin use and chronic diseases: a cohort study of the elderly. BMJ 299: 1247–1250
Peto R, Gray R, Collins R, Wheatley K, Hennekens C, Jamrozik K, Warlow C, Hafner B, Thompson E, Norton S, Gilliland J, Doll R (1988) Randomised trial of prophylactic daily aspirin in British male doctors. BMJ 296: 313–316
Ratnasinghe LD, Graubard BI, Kahle L, Tangrea JA, Taylor PR, Hawk E (2004) Aspirin use and mortality from cancer in a prospective cohort study. Anticancer Res 24: 3177–3184
Rosenberg L (1995) Nonsteroidal anti-inflammatory drugs and cancer. Prev Med 24: 107–109
Shreinemachers DM, Everson RB (1994) Aspirin use and lung, colon, and breast cancer incidence in a prospective study. Epidemiology 5: 138–146
Sorensen HT, Friis S, Norgard B, Mellemkjaer L, Biot WJ, McLaughlin JK, Ekbom A, Baron JA (2003) Risk of cancer in a large cohort of nonaspirin NSAID users: a population-based study. Br J Cancer 88: 1687–1692
Thun MJ, Namboodiri MM, Calle EE, Flanders WD, Heath CW (1993) Aspirin use and risk of fatal cancer. Cancer Res 53: 1322–1327
Thun MJ (2000) Beyond willow bark: aspirin in the prevention of chronic disease. Epidemiology 11: 371–374
Zha S, Yegnasubramanian V, Nelson WG, Isaacs WB, De Marzo AM (2004) Cyclooxygenases in cancer: progress and perspective. Cancer Lett 215: 1–20
Acknowledgements
The research was supported by the Conquer Cancer Coalition of Massachusetts and by grant CA087969 from the US National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Feskanich, D., Bain, C., Chan, A. et al. Aspirin and lung cancer risk in a cohort study of women: dosage, duration and latency. Br J Cancer 97, 1295–1299 (2007). https://doi.org/10.1038/sj.bjc.6603996
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6603996
Keywords
This article is cited by
-
Use of glucosamine and chondroitin and lung cancer risk in the VITamins And Lifestyle (VITAL) cohort
Cancer Causes & Control (2011)
-
Aspirin and NSAID use and lung cancer risk: a pooled analysis in the International Lung Cancer Consortium (ILCCO)
Cancer Causes & Control (2011)