Article
|
Open Access
Featured
-
-
Article |
 Structural basis of mitochondrial protein import by the TIM23 complex
Structural basis of mitochondrial protein import by the TIM23 complex
The cryo-electron microscopy structure of the mitochondrial TIM23 complex from Saccharomyces cerevisiae shows that Tim17 forms a protein translocation path.
- Sue Im Sim
- , Yuanyuan Chen
- & Eunyong Park
-
Article |
 Peroxisome biogenesis initiated by protein phase separation
Peroxisome biogenesis initiated by protein phase separation
A study presents evidence to support a model in which liquid–liquid phase separation of components of the transport machinery mediates formation of transient protein transport channels on peroxisomes.
- Rini Ravindran
- , Isabel O. L. Bacellar
- & Stephen W. Michnick
-
Article |
 Cryptochrome–Timeless structure reveals circadian clock timing mechanisms
Cryptochrome–Timeless structure reveals circadian clock timing mechanisms
Structural analysis of a protein complex in the circadian clock of Drosophila reveals how a light-sensing cryptochrome recognizes and engages its target.
- Changfan Lin
- , Shi Feng
- & Brian R. Crane
-
Article |
 Architecture of chloroplast TOC–TIC translocon supercomplex
Architecture of chloroplast TOC–TIC translocon supercomplex
A cryo-electron microscopy analysis of the Chlamydomonas reinhardtii TOC–TIC supercomplex reveals that Tic214 traverses the chloroplast inner membrane, the intermembrane space and the outer membrane, connecting the TOC complex with the TIC proteins.
- Hao Liu
- , Anjie Li
- & Zhenfeng Liu
-
Article
| Open Access Mitochondrial complexome reveals quality-control pathways of protein import
Mitochondrial complexome reveals quality-control pathways of protein import
An analysis of MitCOM—a comprehensive resource for the identification, organization and interaction of mitochondrial machineries and pathways in yeast—identifies a constitutive pathway for the removal of preproteins.
- Uwe Schulte
- , Fabian den Brave
- & Thomas Becker
-
Article
| Open Access Substrate-driven assembly of a translocon for multipass membrane proteins
Substrate-driven assembly of a translocon for multipass membrane proteins
Biochemical reconstitution and functional analysis reveal how newly synthesized multipass membrane proteins dynamically remodel the translocon to facilitate their successful biogenesis.
- Arunkumar Sundaram
- , Melvin Yamsek
- & Robert J. Keenan
-
Article
| Open Access A peroxisomal ubiquitin ligase complex forms a retrotranslocation channel
A peroxisomal ubiquitin ligase complex forms a retrotranslocation channel
The cryo-electron microscopy structure of the membrane-embedded ubiquitin ligase complex reveals its function as a retrotranslocation channel for shuttling mobile receptors out of peroxisomes.
- Peiqiang Feng
- , Xudong Wu
- & Tom A. Rapoport
-
Article |
 AKIRIN2 controls the nuclear import of proteasomes in vertebrates
AKIRIN2 controls the nuclear import of proteasomes in vertebrates
Using time-controlled CRISPR screens, the authors identify AKIRIN2 as a factor involved in the nuclear import of the proteasome.
- Melanie de Almeida
- , Matthias Hinterndorfer
- & Johannes Zuber
-
Article |
 Mitochondrial sorting and assembly machinery operates by β-barrel switching
Mitochondrial sorting and assembly machinery operates by β-barrel switching
Proteins are inserted into the outer mitochondrial membrane by the mitochondrial sorting and assembly machinery, two structural forms of which are presented here, suggesting the mechanism involved.
- Hironori Takeda
- , Akihisa Tsutsumi
- & Toshiya Endo
-
Article |
 Structure of the ER membrane complex, a transmembrane-domain insertase
Structure of the ER membrane complex, a transmembrane-domain insertase
The cryo-electron microscopy structure of the ER membrane complex provides insight into its overall architecture, evolution and function in co-translational protein insertion.
- Lin Bai
- , Qinglong You
- & Huilin Li
-
Article |
 Structure of the mitochondrial import gate reveals distinct preprotein paths
Structure of the mitochondrial import gate reveals distinct preprotein paths
The high-resolution cryo-electron microscopy structure of the yeast translocase of the outer mitochondrial membrane reveals key features of mitochondrial protein import that are conserved in all eukaryotes.
- Yuhei Araiso
- , Akihisa Tsutsumi
- & Toshiya Endo
-
Article |
 Mitochondrial protein translocation-associated degradation
Mitochondrial protein translocation-associated degradation
In Saccharomyces cerevisiae, Ubx2 promotes clearing trapped precursor proteins from the channel of the translocase of the outer membrane, in a translocation-associated degradation pathway that maintains the protein import capacity of mitochondria.
- Christoph U. Mårtensson
- , Chantal Priesnitz
- & Thomas Becker
-
Article |
 Innervation of thermogenic adipose tissue via a calsyntenin 3β–S100b axis
Innervation of thermogenic adipose tissue via a calsyntenin 3β–S100b axis
The newly identified calsyntenin 3β protein has a role in the innervation of thermogenic fat through a mechanism of communication—which is unique to mammals—between thermogenic adipocytes and sympathetic neurons.
- Xing Zeng
- , Mengchen Ye
- & Bruce M. Spiegelman
-
Letter |
 Structure of the post-translational protein translocation machinery of the ER membrane
Structure of the post-translational protein translocation machinery of the ER membrane
The cryo-EM structure of the post-translational protein translocation machinery of the endoplasmic reticulum membrane shows that Sec63 opens the channel, enabling insertion of low-hydrophobicity signal sequences into the lipid phase.
- Xudong Wu
- , Cerrone Cabanos
- & Tom A. Rapoport
-
Letter |
 TIC236 links the outer and inner membrane translocons of the chloroplast
TIC236 links the outer and inner membrane translocons of the chloroplast
TIC236 is an inner-membrane protein that binds to the outer-membrane channel TOC75 to create a long, stable bridge that enables the transport of proteins into chloroplasts.
- Yih-Lin Chen
- , Lih-Jen Chen
- & Hsou-min Li
-
Article |
 Malaria parasite translocon structure and mechanism of effector export
Malaria parasite translocon structure and mechanism of effector export
Cryo-electron microscopy analysis of the purified Plasmodium translocon of exported proteins (PTEX) reveals two distinct resolved states, suggesting a mechanism by which Plasmodium falciparum exports malarial effector proteins into erythrocytes.
- Chi-Min Ho
- , Josh R. Beck
- & Z. Hong Zhou
-
Letter |
 Mfsd2b is essential for the sphingosine-1-phosphate export in erythrocytes and platelets
Mfsd2b is essential for the sphingosine-1-phosphate export in erythrocytes and platelets
Identification of a transmembrane protein, Mfsd2b, that is essential for the export of the signalling molecule sphingosine-1-phosphate (S1P) from red blood cells and platelets.
- Thiet M. Vu
- , Ayako-Nakamura Ishizu
- & Long N. Nguyen
-
Letter |
 Cryo-EM structure of the protein-conducting ERAD channel Hrd1 in complex with Hrd3
Cryo-EM structure of the protein-conducting ERAD channel Hrd1 in complex with Hrd3
The structure of yeast Hrd1 in complex with Hrd3 shows that Hrd1 forms an aqueous cavity with a lateral seal within the endoplasmic reticulum membrane, shedding light on how misfolded proteins are transported out of the endoplasmic reticulum.
- Stefan Schoebel
- , Wei Mi
- & Maofu Liao
-
Letter |
 The SND proteins constitute an alternative targeting route to the endoplasmic reticulum
The SND proteins constitute an alternative targeting route to the endoplasmic reticulum
Experiments in yeast cells show that three proteins—Snd1, Snd2 and Snd3—provide an alternative pathway for targeting of cellular proteins to the endoplasmic reticulum.
- Naama Aviram
- , Tslil Ast
- & Maya Schuldiner
-
Letter |
 Cotranslational signal-independent SRP preloading during membrane targeting
Cotranslational signal-independent SRP preloading during membrane targeting
The signal recognition particle (SRP) preferentially binds peptides destined for secretion before peptide-targeting signals are translated through recognition of elements in their mRNA, including non-coding sequences.
- Justin W. Chartron
- , Katherine C. L. Hunt
- & Judith Frydman
-
Letter |
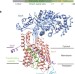 Crystal structure of a substrate-engaged SecY protein-translocation channel
Crystal structure of a substrate-engaged SecY protein-translocation channel
The crystal structure of a substrate-engaged SecY channel and the SecA ATPase, which provides molecular insight into the process of protein translocation across membranes.
- Long Li
- , Eunyong Park
- & Tom A. Rapoport
-
Letter |
 Mistargeted mitochondrial proteins activate a proteostatic response in the cytosol
Mistargeted mitochondrial proteins activate a proteostatic response in the cytosol
Mitochondrial dysfunction and cellular protein homeostasis failure are hallmarks of many diseases and age-associated pathologies; this study shows that the mitochondrial import defect of nuclear-encoded proteins triggers a cellular pathway, termed unfolded protein response activated by mistargeting of proteins (UPRam), that acts to minimize the stress caused by non-imported mitochondrial precursor proteins in order to sustain cellular protein homeostasis and organismal fitness.
- Lidia Wrobel
- , Ulrike Topf
- & Agnieszka Chacinska
-
Letter |
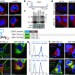 Alternative 3′ UTRs act as scaffolds to regulate membrane protein localization
Alternative 3′ UTRs act as scaffolds to regulate membrane protein localization
Many human genes undergo alternative cleavage and polyadenylation to generate messenger RNA transcripts with different lengths at the 3' untranslated regions (3' UTRs) but that encode the same protein; now it is shown that these alternative 3' UTRs regulate protein localization.
- Binyamin D. Berkovits
- & Christine Mayr
-
Letter |
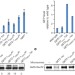 The Get1/2 transmembrane complex is an endoplasmic-reticulum membrane protein insertase
The Get1/2 transmembrane complex is an endoplasmic-reticulum membrane protein insertase
The receptor for the cytoplasmic factor that targets tail-anchored proteins to the endoplasmic reticulum is an enzyme that enables a facilitated insertion path into the lipid bilayer.
- Fei Wang
- , Charlene Chan
- & Vladimir Denic
-
Letter |
 PTEX component HSP101 mediates export of diverse malaria effectors into host erythrocytes
PTEX component HSP101 mediates export of diverse malaria effectors into host erythrocytes
Plasmodium parasites, the causative agent of malaria, infect and remodel red blood cells by exporting hundreds of proteins into the red blood cell cytosol, a topological conundrum given that the parasite resides in a compartment known as the parasitophorous vacuole; here a dihydrofolate-reductase-based destabilization domain approach is used to inactivate HSP101, part of the Plasmodium translocon of exported proteins, and to demonstrate that it is required for the secretion of all classes of exported Plasmodium proteins.
- Josh R. Beck
- , Vasant Muralidharan
- & Daniel E. Goldberg
-
Letter |
 Activated GTPase movement on an RNA scaffold drives co-translational protein targeting
Activated GTPase movement on an RNA scaffold drives co-translational protein targeting
Single-molecule fluorescence microscopy techniques are used to elucidate features of the highly conserved protein-targeting machinery known as the signal recognition particle (SRP); the long SRP RNA is shown to be crucial for correct timing and precision of cargo handover to the protein-translocation machinery, a finding that could help to explain how other ribonucleosome complexes function during complex cellular processes.
- Kuang Shen
- , Sinan Arslan
- & Shu-ou Shan
-
Article |
 Structure of the TatC core of the twin-arginine protein transport system
Structure of the TatC core of the twin-arginine protein transport system
The twin-arginine translocation (Tat) pathway transports folded proteins across membranes in bacteria and plant chloroplasts; the crystal structure of TatC, an integral membrane protein and core component of this complex, is now presented.
- Sarah E. Rollauer
- , Michael J. Tarry
- & Susan M. Lea
-
Letter |
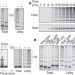 Protein targeting and degradation are coupled for elimination of mislocalized proteins
Protein targeting and degradation are coupled for elimination of mislocalized proteins
- Tara Hessa
- , Ajay Sharma
- & Ramanujan S. Hegde
-
Letter |
 Structure and function of a membrane component SecDF that enhances protein export
Structure and function of a membrane component SecDF that enhances protein export
- Tomoya Tsukazaki
- , Hiroyuki Mori
- & Osamu Nureki
-
Letter |
 Preserving the membrane barrier for small molecules during bacterial protein translocation
Preserving the membrane barrier for small molecules during bacterial protein translocation
- Eunyong Park
- & Tom A. Rapoport
-
News & Views |
 Translocation in slow motion
Translocation in slow motion
Time-resolved electron microscopy can capture structural changes in active macromolecular complexes, but detailed imaging is essential. The dynamics of one step in protein synthesis has been deduced from two million images.
- Måns Ehrenberg
-
Article |
 Plasmepsin V licenses Plasmodium proteins for export into the host erythrocyte
Plasmepsin V licenses Plasmodium proteins for export into the host erythrocyte
To survive and evade host responses, malaria parasites export several hundred proteins into the host cell on infection. A feature of these proteins is a conserved, pentameric motif that is cleaved by an unknown protease before export. This is one of two independent studies revealing the identity of the protease as plasmepsin V, an aspartic acid protease located in the endoplasmic reticulum. This enzyme is essential for parasite viability and is an attractive candidate for drug development.
- Ilaria Russo
- , Shalon Babbitt
- & Daniel E. Goldberg
-
Article |
 An aspartyl protease directs malaria effector proteins to the host cell
An aspartyl protease directs malaria effector proteins to the host cell
To survive and evade host responses, malaria parasites export several hundred proteins into the host cell on infection. A feature of these proteins is a conserved, pentameric motif that is cleaved by an unknown protease before export. This is one of two independent studies revealing the identity of the protease as plasmepsin V, an aspartic acid protease located in the endoplasmic reticulum. This enzyme is essential for parasite viability and is an attractive candidate for drug development.
- Justin A. Boddey
- , Anthony N. Hodder
- & Alan F. Cowman

 Central role of Tim17 in mitochondrial presequence protein translocation
Central role of Tim17 in mitochondrial presequence protein translocation